 |
 |
 |
| |
Preclinical Characteristics of ITMN-191, an Orally Active Inhibitor of the HCV NS3/4A Protease Nominated for Preclinical Development
|
| |
| |
Reported by Jules Levin
DDW, May 2006, Los Angeles
Scott Seiwert1, Steven W. Andrews2, Hong-Woon Yang2, Hua Tan1, Benedict Marafino1, Robert Rieger2, Ronald B. Franklin2, Jed Pheneger2, Patricia A. Lee2, Yutong Jiang2, April L. Kennedy2, Steven M. Wenglowsky2, Machender R. Madduru2, George A. Doherty2, Kevin R. Condroski2, Christine Lemieux2, Lisa Pieti Opie2, Francis Sullivan2, Nickolas Neitzel2, Gary P. Hingorani2, Jennifer Otten2, Barbara J. Brandhuber2, Guy Vigers2, John A. Josey2, Lawrence M. Blatt1
1InterMune, Inc., Brisbane, CA, USA
ABSTRACT
Current treatment options for chronic HCV infections provide sustained virologic response rates of ~50%, indicating that novel therapeutic approaches are needed. We therefore embarked on a rational drug design campaign to produce inhibitors of the HCV NS3/4A protease. ITMN-191 emerged from this discovery effort and was nominated as a preclinical candidate. Here we describe the preclinical characterization of ITMN-191.
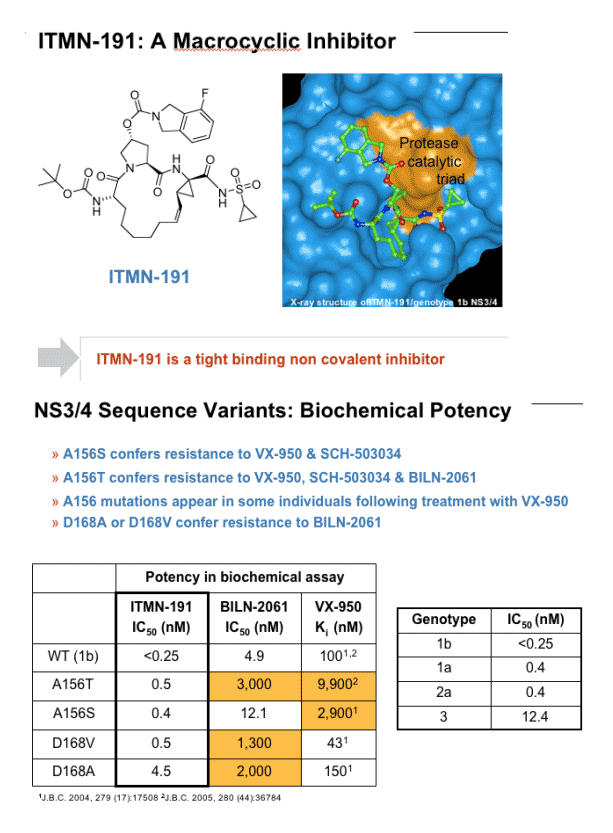
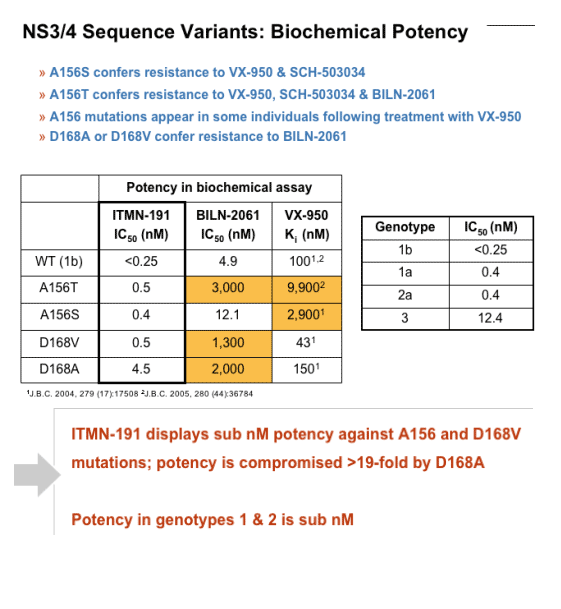
In biochemical assays using HCV NS3/4A protease domains derived from genotypes 1b, 1a, 2, or 3, the EC50 value of ITMN-191 is <300 pM, 400 pM, 400 pM and 12.4 nM, respectively. Its EC50 value against full-length genotype-1b protease is 900 pM. ITMN-191 retains subnanomolar to low-nanomolar potency against the NS3/4A variants at positions A156 and D168 that confer resistance to other experimental NS3/4A inhibitors.
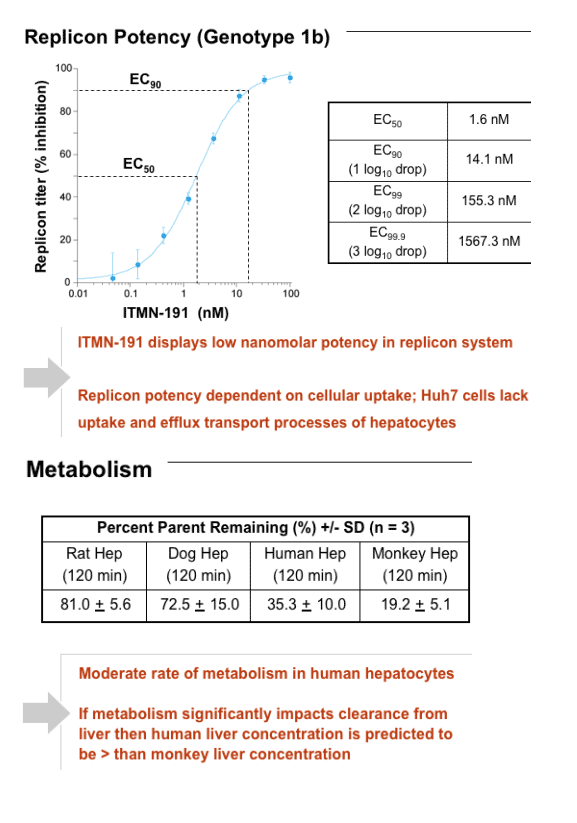
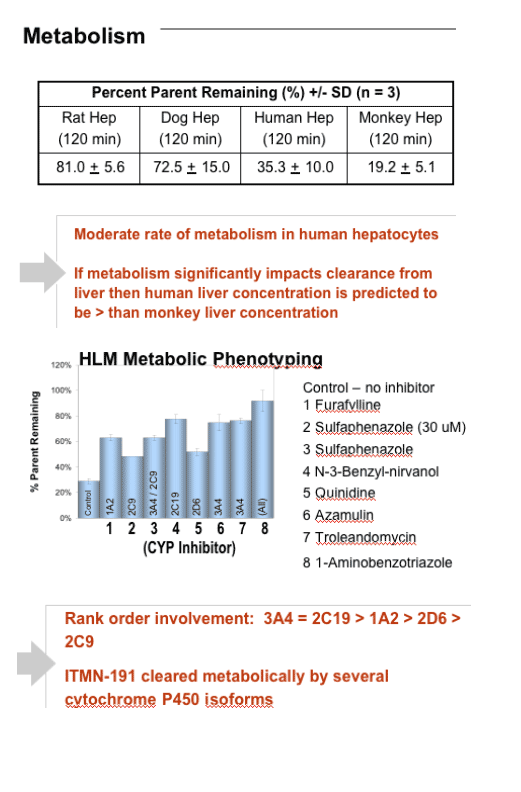
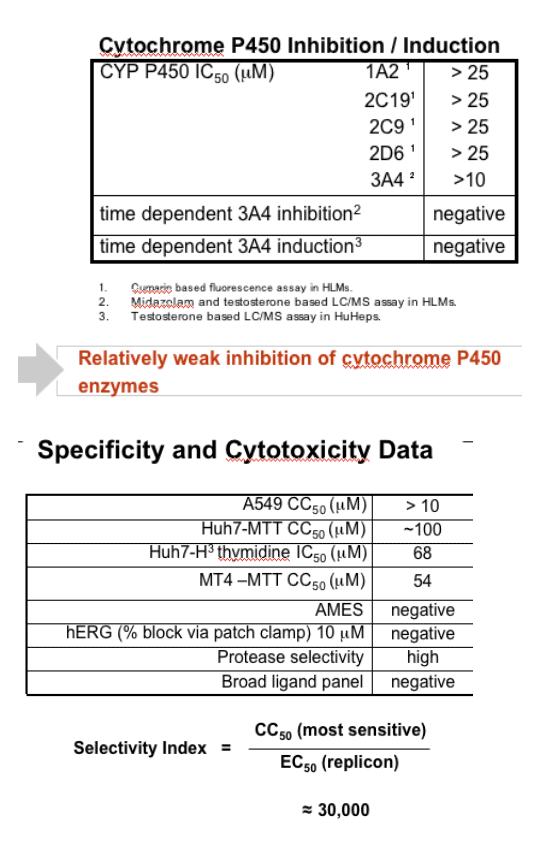
In a genotype-1b replicon system EC50 = 1.6 nM and EC90 = 14.1 nM. ITMN-191 is 97.9% bound by human plasma protein, and its replicon potency is shifted modestly by human serum. ITMN-191 does not inhibit a panel of selected serine proteases, a broader ligand panel, or hERG ion channel. Its CC50 value is >50 mM. ITMN-191 does not inhibit CYP450 isoforms 1A2, 2C19, 2C9, 2D6 and 3A4 at IC50s > 10 mM in human liver microsomes (HLMs). Use of CYP450 inhibitors show ITMN-191 to be metabolized by multiple CYP450 isoforms.
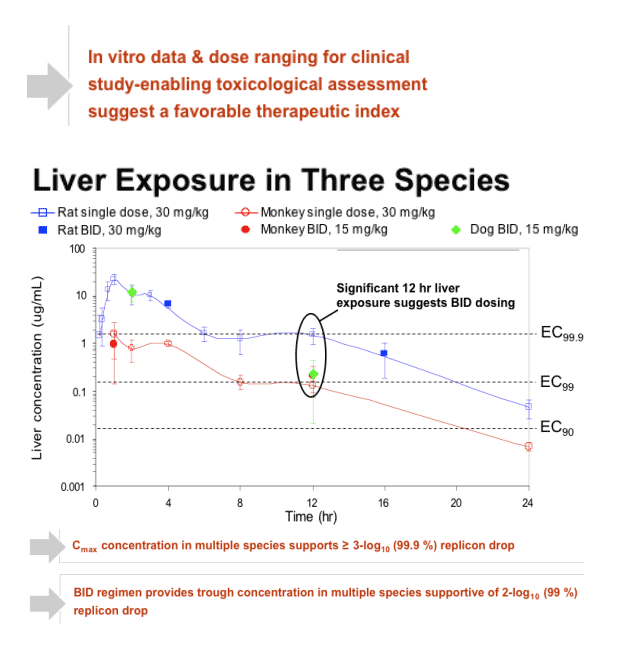
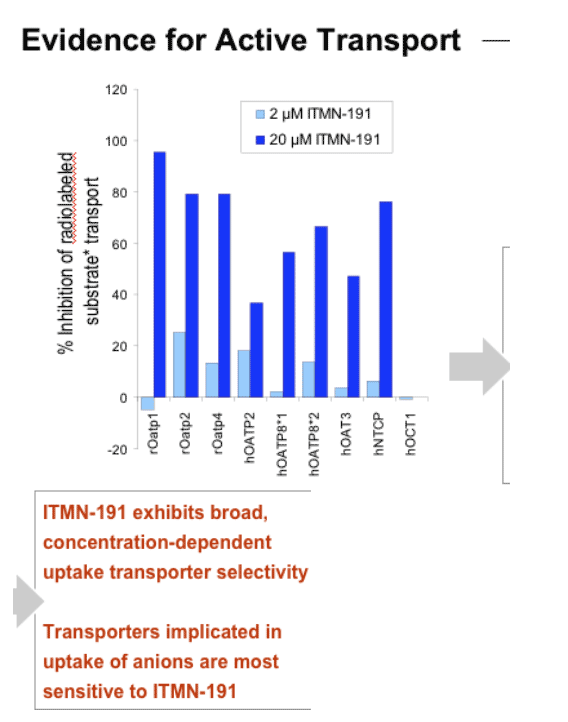
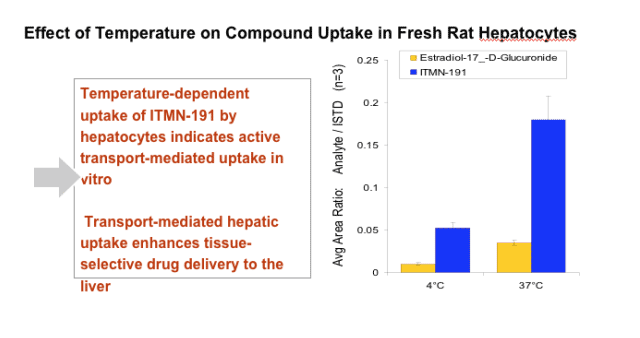
After 120 min incubation with rat, dog, human, or monkey hepatocytes, 81%, 73%, 35%, or 19% of input compound remained intact, respectively. ITMN-191 interacts with a broad spectrum of hepatic organic anion transporter proteins, which likely contributes to its ability to target the liver. After a regimen in which a human equivalent dose of 290 mg BID PO was administered for 7 days, compound trough liver levels in rats and cynomolgus monkeys were 390-fold and 52-fold the EC90 value, respectively, after the last dose in the study. Plasma exposure was linear with dose in both species to at least 300 mg/kg.
In conclusion, ITMN-191 is a highly potent, orally absorbed inhibitor of the NS3/4A protease found in the liver of rats and cynomolgus monkeys at levels predictive of human efficacy. Based on these data, ITMN-191 has been nominated for preclinical development and is currently undergoing IND-enabling toxicological assessment.
Author Summary & Conclusions
- Potent inhibitor of the HCV NS3/4A protease
- Active against proteases derived from genotypes 1, 2, and 3
- Active against A156 and D168 variants of NS3/4A
- Resistance characterization reported in a DDW oral presentation
- Balanced oxidative metabolism
- Weak inhibitor of CYP3A4
- Targets the liver through interactions with hepatic uptake transporters
- Displays high liver concentration in three species, including primate
- Liver trough concentrations support exploration of BID dosing in humans
- Dose-ranging studies for GLP toxicology and in vitro safety data suggest favorable therapeutic index
- European CTA expected in 3Q 2006
|
| |
|
 |
 |
|
|