 |
 |
 |
| |
Further reduction of ribavirin dose in HCV genotype 2/3 patients receiving peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS)
|
| |
| |
Interim results of a randomised controlled trial
P. Ferenci*, H. Brunner, H. Laferl, U. Bergholz,
M. Rosenbeiger, R. Stauber, A. Maieron, G. Fischer,
M. Bischof, P. Steindl-Munda
*Univ. Klinik für Innere Medizin IV, Vienna, Austria
Reported by Jules Levin
EASL, April 29, 2006, Vienna, Austria
Note from Jules Levin: there was quite a bit of controversy regarding the findings of this study, that 400 mg of ribavirin showed equal efficacy to 800 mg in this study' Higher ribavirin dosing has been considered important to SVR with higher dosing leading to better response rates. Dose modifications and use of growth factors are not reported in the slides.
BACKGROUND
The current standard of care in chronic hepatitis C patients infected with HCV genotype 2 or 3 is 24 weeks of a pegylated interferon plus ribavirin 800 mg/day1,2
In HCV genotype 2 or 3 patients, similar SVR rates ( 80%) are seen with peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS)
1000/1200 mg/day or 800 mg/day3,4
1Strader DB, et al. Hepatology 2004; 40: 1147-71
2NIH Consens State Sci Statements 2002; 19: 1-46
3Hadziyannis S, et al. Ann Intern Med 2004; 140: 346-55
4Fried MW, et al. NEJM 2002; 347; 975-82
Further decreasing the dose of ribavirin may avoid unnecessary treatment side effects, including anaemia, and thus improve adherence reduce treatment costs.
Therefore, we prospectively assessed the efficacy and safety of treatment with peginterferon alfa-2a (40KD) plus ribavirin 400 mg/day.
PATIENTS
- Treatment naive
- Age >/=18 years
- Quantifiable HCV RNA (>600 IU/mL) in serum
- HCV genotype 2 or 3 infection
- Evidence of liver disease consistent with a diagnosis of chronic hepatitis C
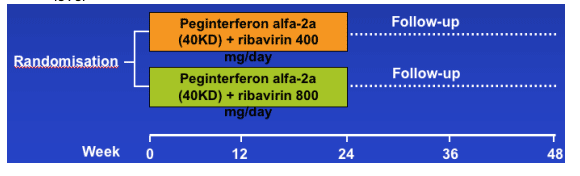
STUDY DESIGN
- Open-label study conducted in 22 centres in Austria
- Patients were randomised to peginterferon alfa-2a (40KD) 180 mg/week plus ribavirin 400 or 800 mg/day for 24 weeks
- Randomisation was stratified according to HCV genotype and HCV RNA level
CURRENT STATUS
- Original recruitment target of 196 patients
- Target revised in order to separately examine outcomes in genotype 2 and 3 patients1 (1. Zeuzem S, et al. J Hepatol 2004; 40: 993-9)
- Revised target of 196 genotype 3 patients, with ongoing recruitment of HCV genotype 2 patients as available
- Recruitment complete in December 2005, with
282 patients randomised to treatment
- Genotype 2 (n=69)
- Genotype 3 (n=213)
- 215 patients have now completed treatment and follow up (final analysis of the initial cohort)
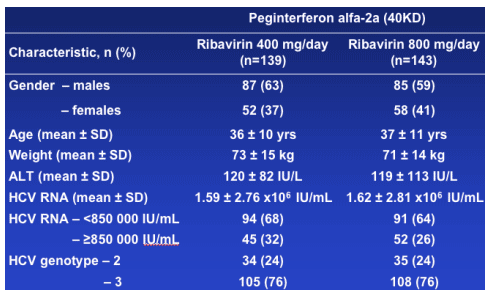
BASELINE CHARACTERISTICS
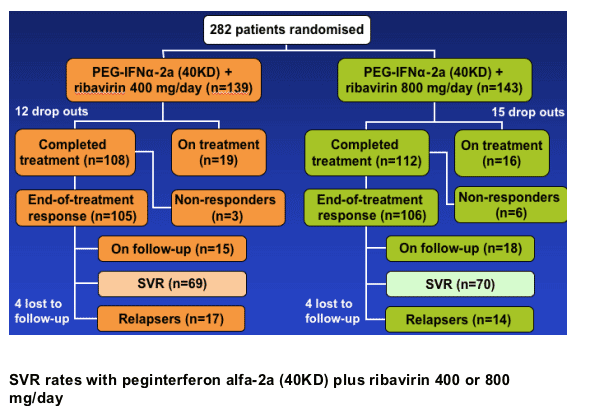
INTERIM ANALYSIS
Analysis population
ITT population - all patients recruited to the study who have reached an endpoint
PP population - all patients recruited to the study who have completed treatment and follow-up
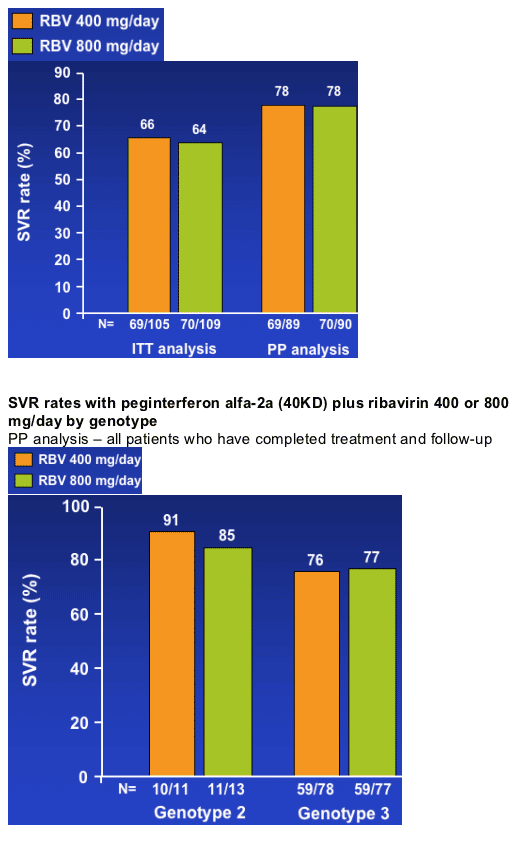
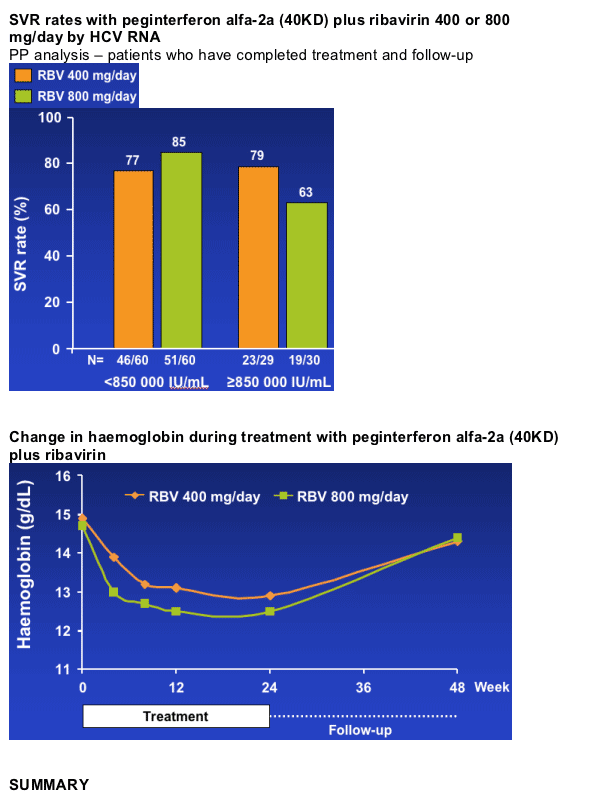
In patients with HCV genotype 2/3, low-dose ribavirin (COPEGUS) (400 mg/day) plus peginterferon alfa-2a (40KD) (PEGASYS) for 24 weeks produced an SVR rate similar to that seen with the standard regimen of peginterferon alfa-2a (40KD) plus ribavirin 800 mg/day:
- SVR rate 78% vs 78%
The rate of SVR was slightly higher in patients infected with HCV genotype 2 compared with genotype 3:
- Baseline HCV RNA also had an impact on SVR rate
The lower ribavirin dose resulted in a less profound reduction in haemoglobin levels
CONCLUSIONS
- Attempts to reduce treatment from 24 to 12-16 weeks has resulted in increased rates of relapse, even when the ribavirin dose was adjusted according to bodyweight1-3
- The large ACCELERATE study presented at this meeting has confirmed (see study results) that a shorter treatment duration results in lower SVR rates as a result of higher rates of relapse4
- Our study indicates that it may be possible to simplify treatment in patients with HCV genotype 2/3 infection by reducing the ribavirin dose to 400 mg/day when given with peginterferon alfa-2a (40KD) for 24 weeks
1Dalgard O, et al. Hepatology 2004; 40: 1260-5
2Mangia A, et al. NEJM 2005; 353: 2609-17
3von Wagner M, et al. Gastroenterology 2005; 129: 522-7
4Shiffman ML, et al. EASL 2006. Oral late-breaker presentation (abstract 734)
|
| |
|
 |
 |
|
|