 |
 |
 |
| |
Initial Results of a 14-Day Study of the Hepatitis C Virus Protease Inhibitor VX-950, In Combination with Peginterferon-alfa-2a
|
| |
| |
Reported by Jules Levin
EASL Conference
April 26-30, Vienna, Austria
The Late Breakers oral session just ended literally minutes ago and I wanted to report to you the most interesting data presented. First, in addition to the VX-950 data reported here, Roche reported for the first time 14-day monotherapy data on their new HCV polymerase inhibitor, an orally administered HCV drug which showed a mean -1.2 log reduction in HCV viral load. Albuferon administered once every two weeks showed at least equivalent antiviral efficacy & safety as peginterferon in preliminary results without a problem in immungenicity so far. The major story from EASL is the data reported on VX-950. At EASL two days ago Vertex presented resistance data from the Vx-950 monotherapy study and reported drug resistance emerged during the 14-day VX-950 monotherapy study and 3-7 months later wild-type virus was mostly present but with a percentage of resistant virus as well.
HW Reesnik (Academic medical center, Amsterdam) reported the study results.
AUTHOR SUMMARY
VX05-950-103 Summary
Peg-IFN alone group
--Response consistent with literature
VX-950 alone group
- about 3-log initial reduction in median HCV RNA
- 4.0-log reduction in median HCV RNA by Day 14
- Rebound or plateau in 4/8 patients
- Results consistent with prior VX04-950-101 study
VX-950 and Peg-IFN group
- 5.5-log reduction in median HCV RNA at Day 14
- All patients had continuous HCV RNA decline
- All patients have undetectable HCV RNA 12 weeks after starting follow-on standard therapy
AUTHORCONCLUSION & FUTURE DIRECTIONS
Addition of Peg-IFN to VX-950 increases antiviral response and was not associated with viral breakthrough.
Dosing has been completed in a 28-day study of VX-950/Peg-IFN/RBV, in 12 genotype 1 HCV patients.
Data to be presented at DDW in May 2006.
A Phase 2 study which will include evaluation of a 12-week VX-950-based treatment regimen is now being initiated.
BACKGROUND
- VX-950 is an orally-bioavailable inhibitor of the Hepatitis C NS 3A4 protease.
- It is one of a new class of Specifically Targeted Antiviral Therapies
for HCV (STAT-C)
- In in vitro studies it has demonstrated reversible and tight binding to the HCV protease active site, forming a stable enzyme-inhibitor complex
- Data previously published and presented has shown that VX-950 is active in replicon assays, replication assays, and in animal models, both as a single agent and in several different combinations.
- In a 14-day study in chronic hepatitis C patients, VX-950 was well tolerated and showed rapid and dramatic antiviral activity. This data was presented at AASLD in 2005
VX05-950-103 Clinical Study Design
Objectives:
- The primary objective of this study was the assessment of viral kinetics and antiviral activity of
VX-950 with Peg-IFN
- The secondary objective was the assessment of clinical safety and pharmacokinetics
Study dosing period was 14 days
20 patients, treatment-naïve, genotype 1
- VX-950 (750 mg q8h) plus Peg-IFN weekly (n=8)
- VX-950 (750 mg q8h) (n=8)
- Peg-IFN weekly (n=4)
VX-950 was dosed as a tablet
At the completion of study dosing, standard therapy (Peg-IFN and RBV) was provided for all subjects
Data presented today are based on an end-of-dosing interim analysis but includes clinical follow-up data available at this time--
--Includes clinical safety and antiviral data
--Pharmacokinetic analyses and viral sequencing still in progress
Baseline Status for Patients
The table immediately below shows baseline patient data. The VX-950 alone group had more females, had a higher median baseline weight by about 10 kg, and a median age of about 10 years older.
This group also had a slightly lower median HCV RNA.
The total study group had a median baseline viral load of 6.64 IU/mL.
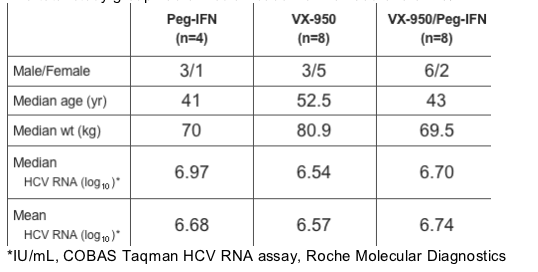
*IU/mL, COBAS Taqman HCV RNA assay, Roche Molecular Diagnostics
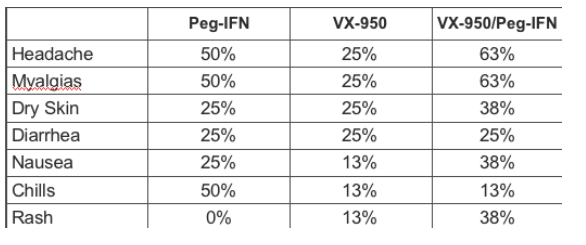
VX-950 was Well Tolerated with and without Peg-IFN
No SAEs, no treatment discontinuations
All patients received 100% of the intended doses.
There were no events that delayed or prevented dosing.
There were 4 episodes of rash, and they all resolved during dosing without treatment, and lasted no more than 5 days
Most common Adverse Events:
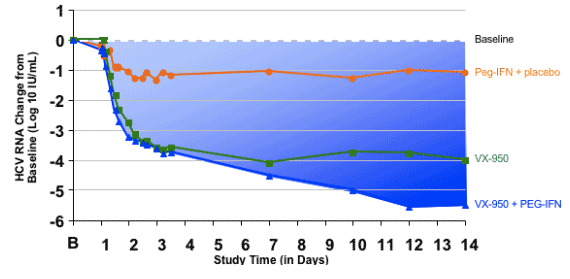
Median Change from Baseline HCV RNA: VX-950/Peg-IFN
This figure shows the median HCV RNA curve for the 8 patients who got VX-950 and Peg-IFN.
In the first 3 days the patterns of response are similar to the VX-950 alone group.
At day 14 of dosing 5.5 log reduction in median HCV-RNA was observed for the Peg-IFN+VX950 combination group.
Different from the VX-950 alone group, all patients had a continuous HCV-RNA decline.
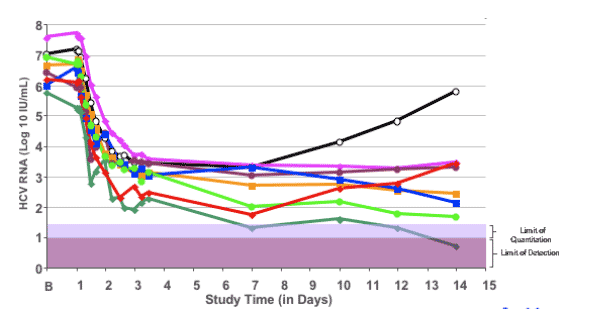
Individual HCV RNA curves: VX-950 group
In this slide the individual curves of the 8 patients receiving VX-950 alone are shown.
A rebound or plateau in viral response was observed in 4 of 8 patients.
This is consistent with our previous study in which the selection of viral variants with decreased sensitivity to VX-950 was found to be responsible for this rebound phenomenon.
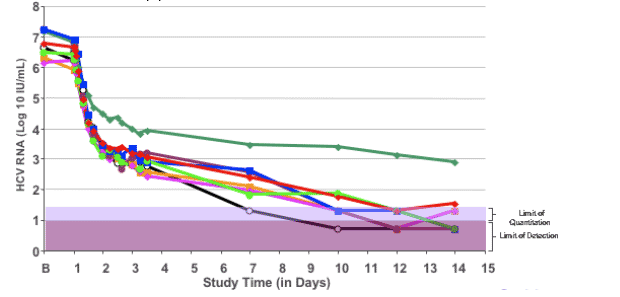
Individual HCV RNA curves: VX-950/Peg-IFN group
In this slide the individual curves of the 8 patients receiving VX-950 and Peg-IFN are depicted.
All patients had continuous HCV-RNA decline during the 14 day study period.
One subject in this group had a slower response than the rest of the group, still reaching a 3 log drop after a continual decline in viral load.
Review of the demographic and pharmacokinetic factors for this patient do not demonstrate any clear reason for the slower response;
Regardless, this patient started SOC after the completion of study dosing, and subsequently became HCV RNA undetectable, and remained undetectable at the most recent follow-up point.
Undetectable* HCV RNA Achieved at Day 14
Of the 8 patients receiving VX-950+pegasys, 6 had <30 IU/mL and 4 had <10 Iu/mL.
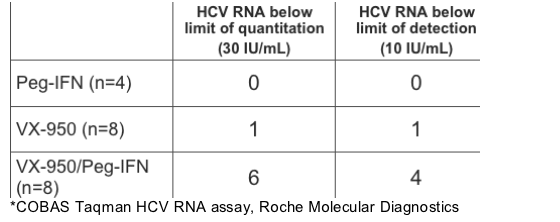
*COBAS Taqman HCV RNA assay, Roche Molecular Diagnostics
VX05-950-103 Viral resistance
- Virus samples were collected for sequencing at baseline, Day 4, Day 8, Day 12, Day 15 and at 7-day follow-up.
- In the VX-950 group: 4 subjects had viral plateau or viral breakthrough
--Viral sequencing work is in progress
- In the VX-950/Peg-IFN group: there was no viral breakthrough seen
--- Most samples collected after Day 4 were below the limit of the sequencing assay, as were many of the samples in the VX-950 group.
Sequencing work is in progress for the samples which can be sequenced
|
| |
|
 |
 |
|
|