 |
 |
 |
| |
PHARMACOKINETIC BOOSTING OF VX-950 AND SCH 503034, INHIBITORS OF HCV PROTEASE, BY CO-DOSING WITH RITONAVIR
|
| |
| |
Author: D.J. Kempf et al, Abbott.
Reported by Jules Levin
41ST EASL
Vienna, April 2006
At this liver meeting Dale Kempf reported that in rat microsomes ritonavir increased VX-950 AUC by >8-fold and C8th by >50-fold, that is the level of Vertex HCV protease inhibitor VX-950 drug levels 8 hours after dosing. In rat microsomes, ritonavir increased Schering HCV protease inhibitor SCH 503034 AUC by 20-fold and C8 by >100-fold. Kempf said you cannot equate these increases to what you would see in humans, the humans studies would have to be conducted to see how much ritonavir would increase the levels of these two HCV protease inhibitors but he suggested ritonavir boosting would (1) increase drug levels of the two HCV PIs significantly as they do with HIV PIs, (2) higher levels may improve the dosing of the HCV PIs; currently these two HCV PIs are administered 3 times per day, (4) ritonavir might allow for a lower dose of the HCV PIs; current dosing for VX-950 is 750 mg 3 times daily, and for SCH 503034 studies are exploring 400 and 800 mg three times daily, (4) higher levels of might improve the risk for drug resistance developing as boosting does with HIV PIs. Note from jules Levin: yesterday Vertex presented an oral discussion on drug resistance as it relates to VX-950. They reported that drug resistance emerged duringthe 14-day monotherapy study for VX-950 but 3-7 moths after stopping VX-950 wild-type virus re-emerged. At the EASL meeting they are presenting data from the combination study of Pegasys + VX-950. Vertex suggested the addition of eginterferon may help suppress drug resistance but they have not yet reported drug resistance data from the study of this combination. There is a chance that another oral agent might be required to avoid resistance, a 3-drug combination. Drug resistance data related to SCH 503034 has not yet been well characterized, not as well characterized as has been by Vertex. Schering says they don't have resistance data yet.
BACKGROUND
VX-950 and SCH 503034 are peptidomimetic HCV protease inhibitors:
- Promising new small-molecule HCV therapeutics in development
- Monotherapy produces substantial declines in HCV RNA in short-term studies
- Both compounds dosed three times daily (VX-950: 750 mg q8h; SCH 503034: 400-800 mg TID)
There is a high correlation between plasma trough levels and virologic response:
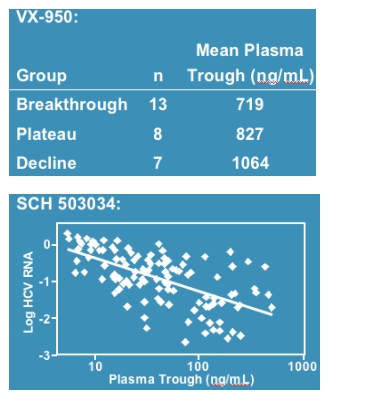
Background: Pharmacokinetic Boosting of HIV Protease Inhibitors with Ritonavir
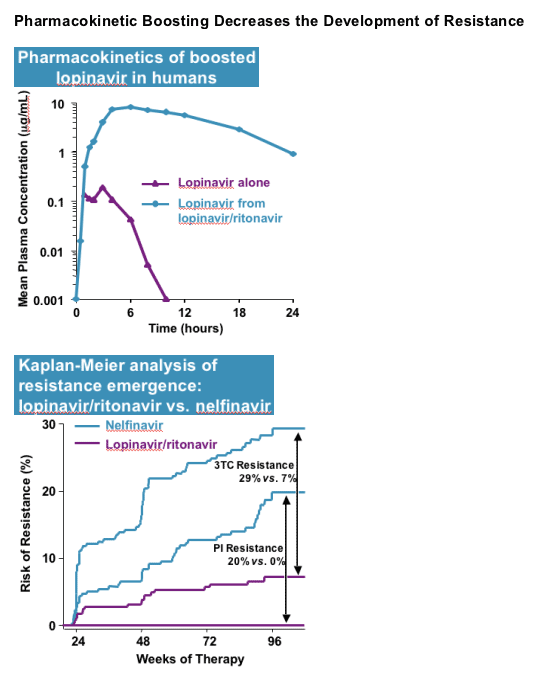
Ritonavir (Norvir ) is a drug originally licensed for HIV therapy, but now used in low doses to boost the plasma levels of HIV protease inhibitors (HIV PIs)
- The efficacy of peptidomimetic HIV PIs is also correlated to plasma trough levels
- The plasma trough levels of HIV PIs are greatly enhanced by co-dosing with ritonavir by virtue of potent inhibition of cytochrome P450 3A (CYP 3A)
- The majority (75-80%) of HIV PI use is in ritonavir-boosted regimens
Pharmacokinetic boosting with ritonavir:
- Increases efficacy
- Allows for more convenient twice daily or once daily dosing
- Delays the development of drug resistance
Pharmacokinetic Boosting Decreases the Development of Resistance
BACKGROUND
Structurally, some HCV PIs are similar to HIV PIs
- Peptidomimetic, equivalent in size to penta- or hexapeptides
- Highly hydrophobic
HIV PIs are largely or exclusively metabolized by CYP 3A
- Metabolic pathways for HCV PIs have not been described

METHODS
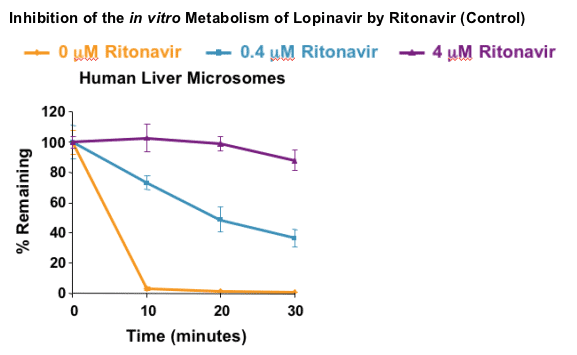
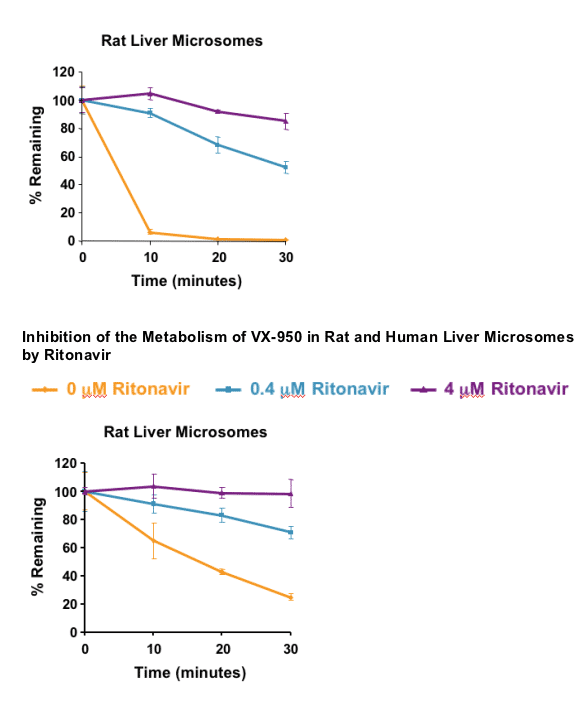
In vitro screening assay:
- Rat liver microsomes harvested from male Sprague-Dawley rats
- Pooled human liver microsomes (n = 10 individuals)
- Disappearance of parent HCV PI at 1 mM initial concentration
- Inhibition by 0.4 and 4.0 mM ritonavir assessed
In vivo screening assay:
- HCV PIs and ritonavir prepared as solutions in PEG-400
- Male Sprague-Dawley rats dosed orally with HCV PIs by gavage at 5 mg/kg, alone or with 5 mg/kg of ritonavir
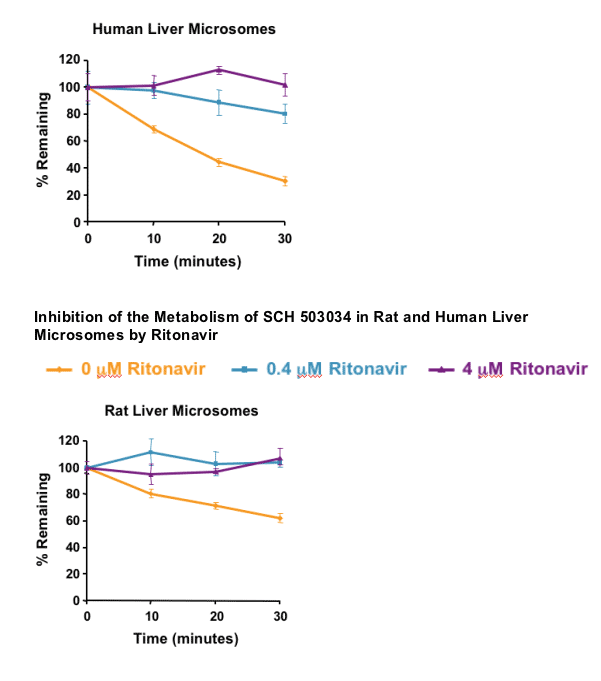

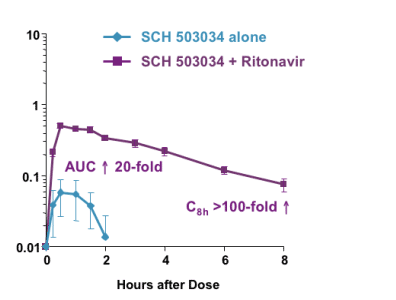
DISCUSSION
VX-950 and SCH 503034 metabolism is strongly inhibited by ritonavir in vitro and in vivo:
- Metabolism likely primarily due to CYP 3A
- Enhancement of the plasma levels of these HCV PIs by co-dosing with ritonavir in humans is likely
Co-dosing of VX-950 and SCH 503034 with ritonavir in humans should be investigated:
- Conversion from TID to BID or QD dosing likely, lower doses likely
- Possible improved efficacy and/or delayed resistance from higher trough levels
Low doses of ritonavir required for pharmacokinetic boosting (100-200 mg/day) are well-tolerated in HCV-HIV-coinfected individuals
- Ritonavir exposure at boosting doses is only 5-8% that of the original full-dose (600 mg BID) ritonavir
- No evidence of incremental hepatotoxicity in several studies comparing boosted to non-boosted HIV PI-based regimens
|
| |
|
 |
 |
|
|