 |
 |
 |
| |
Pegasys/RBV In Post Liver Transplant for Recurrent HCV
|
| |
| |
Reported by Jules Levin
EASL, April 26-30, Vienna, Austria
Multicentre randomised trial of HCV treatment with peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS) in liver transplant patients with established recurrent hepatitis C: interim analysis'
Christophe Duvoux
Hopital Henri Mondor-Paris XII University, Creteil
France
For a French Multicentre Study Group
BACKGROUND
- After liver transplantation (LT), HCV recurrence is almost universal with 10 to 30% of patients developing cirrhosis within 5 years.
- Treatment of HCV recurrence after LT is difficult and somewhat disappointing:
-Sustained virological response with combination therapy had been achieved in 21* to 45%** of the cases.
- The risk of treatment-induced rejection ranges from 2 to 25%**
- Tolerance is poor: 20 to 50% of treatment discontinuation.
- The role of a maintenance ribavirin treatment has not been studied.
*Samuel et al. Gastroenterology 2003;
**Dumortier et al. J Hepatol 2004
AUTHOR CONCLUSIONS
Encouraging antiviral effects were observed following 1 year of combination therapy with peginterferon alfa-2a (40KD) PEGASYS and ribavirin (COPEGUS)
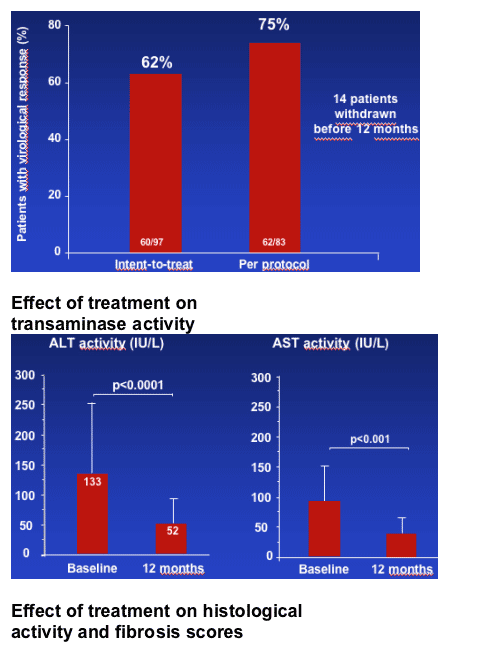
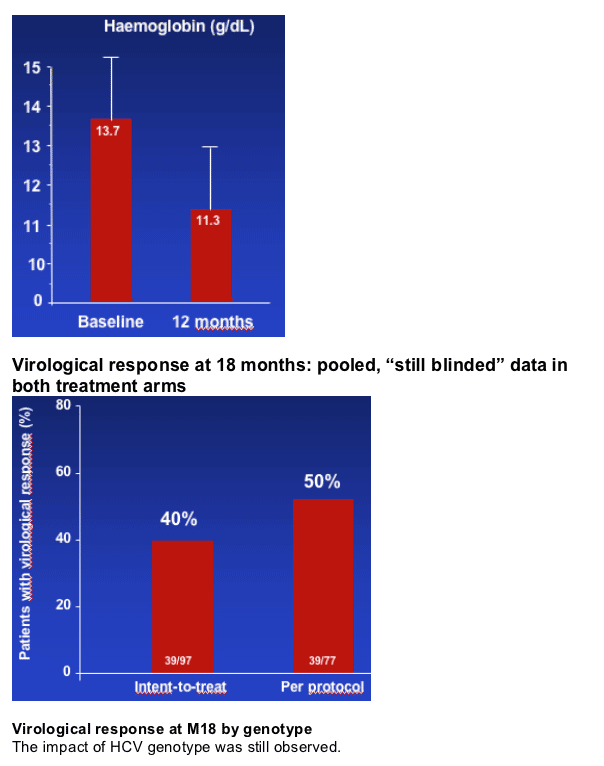
A virological response (HCV RNA <50 IU/L) was achieved in 62% of patients overall (ITT analysis) and in 75% of pts in per protocol analysis at one year.
Virological response 6 months after cessation of combination therapy was achieved in 50% of patients (per protocol) in per protocol analysis.
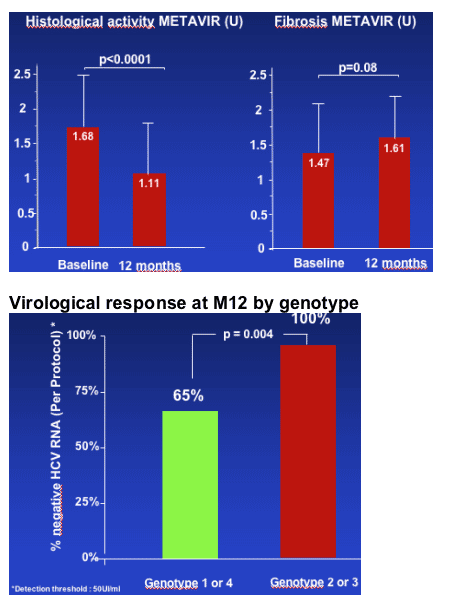
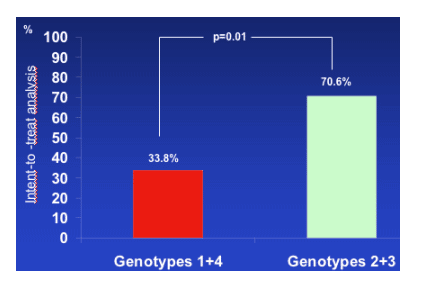
Virological response was associated with genotype.
Keeping in mind the frequent use of growth factors, tolerability was good
-- 7% discontinuation rate for haematological reasons
-- 2% incidence of reversible rejection.
Taken together, these results indicate a favourable risk/benefit ratio of this therapy.
Study Aims
- To test for the efficacy of a one-year combination therapy with peginterferon alfa-2a (40KD) plus ribavirin in liver transplant patients with established recurrent HCV infection.
- To investigate whether a 1-year maintenance therapy with ribavirin following a 1-year combination therapy had a positive impact on post LT HCV recurrence.
Interim results after 1 year of combination therapy will be presented
In addition, preliminary long-term results will also be shown.
Patients
15 LT centres between 10/2002 and 08/2004
Inclusion criteria:
-- Primary liver transplantation for HCV cirrhosis
-- Post-transplant follow-up: 1 to 5 years
-- Previously untreated recurrent HCV infection with serum positive HCV RNA
-- Fibrosis stage >/= 1 (METAVIR scoring system), on a liver biopsy obtained 1 to 5 years after transplantation
Exclusion criteria:
-- Contra-indication to IFN or RBV
-- Active acute or chronic rejection
-- Living donor liver transplantation
-- Mycophenolate mofetil-based immunosuppression
-- Severe concomitant disease (renal failure, cardiac failure, severe sepsis)
HIV and HBV coinfection
Methods: Study design
And pts were followed for an additional period of 6 months after completion of this second phase of the study.
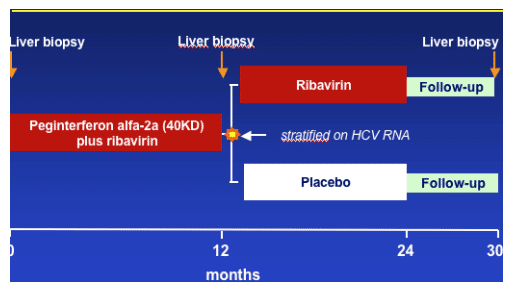
Modalities of treatment
- Peginterferon alfa-2a (40KD) was initiated at 90 ug/week for 2 weeks, then increased to 180 ug/week or adjusted as a function of haematological tolerance.
- Ribavirin was initiated at 600 mg/day, then increased to
800 mg/day or adjusted as a function of haematological tolerance.
- Treatment was stopped for safety reasons or laboratory abnormalities (Hb < 8g/dL, PMN <750/uL, platelets <30,000/uL).
- Use of growth factors was allowed at investigator discretion.
Endpoints
Primary
Virological response:
-- Non-detectable serum HCV RNA* after 1 year of combination therapy (12 months)
-- Non-detectable serum HCV RNA* at the end of follow-up (30 months)
*COBAS AMPLICOR HCV Test v2.0; lower limit of detection 50 IU/mL
Secondary
-- Liver function tests
-- Histological changes from baseline biopsy and biopsies at Months 12 and 30 (reviewed by a single pathologist in a blinded fashion)
-- Safety parameters
Baseline characteristics of the patients

The presenter said: 69.3% of the patients who continued on peg-interferon-ribavirin combination achieved a sustained virological response at the end of follow-up, whereas only 53.4% of those who discontinued therapy at week 24 did. The difference was significant.
Factors associated
with virological response at M12
No correlation between virological response and:
- Time since LT
- Histological score at baseline
- Liver function tests at baseline
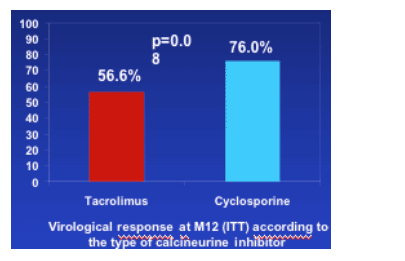
A trend toward a better response on CsA
Serious adverse events
22 serious adverse events related to antiviral therapy occurred in 18 patients
2 rejections episodes (2%)
-- Moderate: steroid pulses; treatment discontinuation
-- Mild: increase in basal CNI doses
-- Reversible in both cases
Treatment was discontinued in 12 patients, due to SAE:
Haematological reasons (n=7) :
-- anaemia 6, leucopenia 1
-- Psychiatric reasons (n=2)
-- Impairment in renal function (n=2)
-- Rejection (n=1)
Other significant SAEs: Ischemic cardiopathy (1), Diabetes (1), Infection (4)
No serious adverse event-related death
Effect of treatment on haemoglobin
37% patients required erythropoietin and 12% G-CSF
|
| |
|
 |
 |
|
|