| |
BMI, Waist Size, and Fitness & Central Adiposity Associated with NAFLD
(nonalcoholic fatty liver disease)
|
| |
| |
"Association of Cardiorespiratory Fitness, Body Mass Index, and Waist Circumference to Nonalcoholic Fatty Liver Disease"
Gastroenterology June 2006
Volume 130, Issue 7, Pages 2023-2030
Timothy S. Church_, Jennifer L. Kuk , Robert Ross , Elisa L. Priest_, Emily Biltoff , Steven N. Blair_
_ The Cooper Institute, Dallas, Texas
School of Physical and Health Education, Queen's University, Kingston, Ontario, Canada
Texas Tech University School of Medicine, Lubbock, Texas
note from Jules Levin: as you can see in table 1 below blood pressure, troglycerides level, HDL cholesterol level, triglyceride to HDL ratio, and the presence of metabolic syndrome, the presence of visceral adiposity, and fitness were all associated with NAFLD; the better these markers were the less likely to have NAFLD.
Nonalcoholic fatty liver disease (NAFLD) represents a spectrum of conditions that span from asymptomatic steatosis to potentially deadly nonalcoholic steatohepatitis (NASH).1, 2, 3, 4, 5, 6 Although simple steatosis is generally a benign condition, NASH can progress to cirrhosis and liver failure, and the 5-year survival rate for individuals diagnosed with NASH is estimated to be 67%.7 NASH is distinguished from simple steatosis by the presence of hepatocyte injury, inflammation, and fibrosis.5, 6, 8 The prevalence of NAFLD and NASH in the general population is estimated to be 16%-30% and 2.1%-6.3%, respectively.5, 7, 8 A liver biopsy examination is required for the diagnosis of NASH, which makes large-scale epidemiologic trials focused on NASH in asymptomatic populations problematic, if not impossible, to conduct ethically.7
The prevalence of both NAFLD and NASH have been shown to be associated strongly with excess body weight, in particular central adiposity.1, 7, 9, 10, 11, 12, 13, 14 However, this association is complicated by body weight and central adiposity both being associated strongly (inversely) with regular physical activity, and physical activity is associated strongly (inversely) with insulin resistance, which has been hypothesized to be a causal mechanism in NAFLD.6, 7, 8 Previous reports that have found overweight/obesity to be associated with a higher prevalence of NAFLD either did not account for physical activity patterns or used self-report measures of physical activity, which leads to substantial misclassification on this exposure.1, 12, 13, 14 Cardiorespiratory fitness is an objective laboratory measurement that reduces the misclassification bias that often results from self-reports of physical activity.15
The purpose of this study was to examine the associations among cardiorespiratory fitness, body mass index (BMI), and waist circumference with a marker of NAFLD in asymptomatic men. In combining computed tomography-measured liver fat with increases in liver enzyme levels we sought to create a surrogate measure of NAFLD that is closer to NASH in the NAFLD continuum than simple steatosis. With the increasing clinical presence of NAFLD and NASH, there is a need for more work examining the potential of physical activity and/or weight control as a preventive and/or therapeutic option in the treatment of fatty liver diseases.
AUTHOR DISCUSSION
The primary finding of this cross-sectional study was a lower prevalence of NAFLD across higher levels of cardiorespiratory fitness and a higher prevalence of NAFLD across higher levels of BMI and waist circumference. When examined within the same model we found fitness level (inversely) and BMI category (directly) both to be associated significantly with the prevalence of NAFLD. However, both the fitness-NAFLD and BMI-NAFLD relationships were attenuated when central adiposity, in particular visceral fat, was taken into account.
The observation that abdominal visceral adiposity, but not abdominal subcutaneous adiposity or BMI, attenuates the fitness-NAFLD relationship is of importance because we have shown previously that at a given weight, individuals who exercise more have less visceral fat than those who are sedentary.20 Further, exercise intervention trials have shown that even without large weight loss, increases in physical activity in white men and women can result in substantial reduction in visceral fat.33, 34 Taken collectively, an important mechanism whereby regular physical activity may reduce the risk of NAFLD is through reduced visceral fat, even without substantial weight loss. However, one cannot assume causality when examining cross-sectional data, and at this point the importance of visceral fat in the development of NAFLD disease is only hypothetical and needs to be examined in longitudinal studies. In contrast to our previous reports examining mortality and markers of metabolic dysfunction across levels of fitness and fatness, we did not find that adjustment for fitness completely eliminated the association of BMI to the outcome of interest.35, 36, 37, 38 In fact, we found the interaction term for fitness and BMI to be significant, suggesting that the combination of low fitness and high BMI levels are associated with a particularly high risk for the prevalence of NAFLD. Thus, the best approach to minimizing the risk for developing NAFLD may be through a combination of weight control and a physically active lifestyle.
Insulin resistance has been proposed to be essential to the development of NAFLD. We did not find our crude index of insulin resistance to be a mediator of the benefits of physical activity or lower weight. There are other potential mechanisms that we did not test such as markers of systemic inflammation and oxidative stress, which have been described as core elements of NAFLD. We previously reported that higher levels of fitness are associated with reduced levels of the systemic inflammation marker C-reactive protein, and others have reported physical activity to have antioxidant effects.37, 39 In summary, although visceral adiposity attenuates the association between physical activity and the prevalence of NAFLD, there likely are other potential mechanisms whereby regular physical activity may reduce the risk for the development or progression of NAFLD that deserve exploration.
There currently exists no definitive strategy for the prevention or treatment of NAFLD. Although weight maintenance or weight loss often is mentioned as a therapeutic option for NAFLD, there are no large randomized clinical trials that have examined the value of weight loss in preventing or treating NAFLD. Given the lack of therapeutic options related to NAFLD, there is a need for clinical trials to evaluate lifestyle interventions for the treatment of NAFLD. Our data suggest that regular physical activity also should be considered in future trials both in the context of promoting and maintaining weight loss, in particular reducing central adiposity, and for its weight-independent health benefits.
Treating liver fat as a continuous variable and using regression analysis, we previously reported that liver fat is associated inversely with fitness within this same population.19 The current analysis is significantly different from this previous report in that liver fat was examined as a dichotomous variable in combination with markers of liver injury to create a marker of NAFLD. The significance of the addition of liver enzyme levels to the creation of the NAFLD index is evidenced by 43 participants having an L/S of 1.0 or less, but only 24 participants had an L/S of 1.0 or less with an increased ALT level combined with an AST/ALT level of less than 1.0.
It is noteworthy that of the 24 individuals with NAFLD, 21 had a waist circumference greater than 100 cm. This suggests that the measurement of waist circumference in combination with the measurement of liver enzyme levels may have the potential to serve as an easy clinical screening tool for identifying individuals who are at high risk for having or developing NAFLD. This is an area deserving of future work.
Diet composition influences liver fat content and regular exercisers may have healthier diets than nonexercisers, although data are inconsistent on this issue.40, 41, 42 We do not have sufficient dietary data on this group of participants to evaluate the influence of diet. Cardiorespiratory fitness is determined primarily by exercise habits, but there is also a genetic component, which accounts for 25%-40% of the variation in fitness.43, 44 We have no data on how the genetic influence on fitness might be related to the findings of the present study. The predominantly white, middle- to upper-class study population limits the generalizability of the results, but should not affect the internal validity. In fact, the homogeneity of our study group regarding socioeconomic factors is a benefit because it reduces the likelihood of confounding by these factors. It is unclear whether the observations in this study can be extended to women. As with any cross-sectional study, causality between the independent variables of fitness and BMI or other measures of body fat and fat distribution and the dependent variable (NAFLD) cannot be established. Lack of serum hepatitis C status is also an important limitation of this study.
This study has a number of strengths including an objective measure of cardiorespiratory fitness and detailed information on medical history and smoking. Perhaps the most important strength is examining the fitness-NAFLD relationship within levels of body composition and fat distribution as assessed by BMI value, waist circumference value, abdominal visceral adiposity, and abdominal subcutaneous adiposity.
Conclusion
Fitness (inversely) and BMI (directly) were associated with the prevalence of NAFLD. However, these associations were attenuated when abdominal obesity was included in the statistical model. There is a need for future work examining the role of weight loss and physical activity in the prevention and treatment of NAFLD.
Results
Table 1 presents the characteristics of the participants by the absence (n = 194) and presence of NAFLD (n = 24, 11.0%). Similar in age, participants with NAFLD were heavier, had more central adiposity, were less fit, and had worse cardiovascular risk factor profiles compared with participants without NAFLD. For example, almost 92% of individuals with NAFLD had metabolic syndrome compared with 30.9% of individuals without NAFLD (P < .001). There was no difference in alcoholic drinks consumed per day in the 2 groups.

Table 2 presents participant characteristics, including liver measures, across fitness tertiles. Age was not associated with fitness level (P = .08). There was a direct relation between alcoholic drinks consumed per day and fitness group and an inverse association between all measures of adiposity and fitness. Higher fitness was associated with better measures of liver injury and liver fat. The unadjusted prevalence of NAFLD was related inversely to fitness tertiles. Although the fasting glucose level was related inversely to fitness level, the prevalence of insulin resistance was not (P = .08).
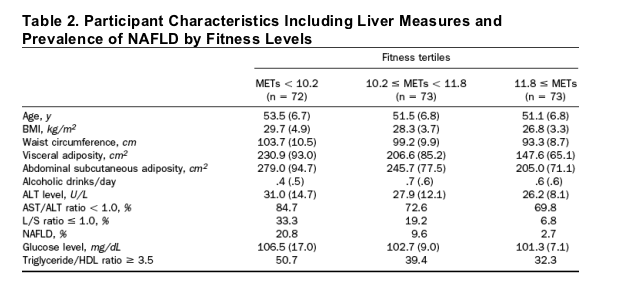
Table 3 presents participant characteristics including liver measures, BMI categories, and waist tertiles. BMI and waist circumference categories were associated positively with age and negatively with fitness. Neither BMI nor waist circumference categories were associated with alcohol use. Lower BMI and waist circumference values were associated with better measures of liver injury and liver fat. The unadjusted prevalence of NAFLD showed a positive association across BMI and waist circumference categories.
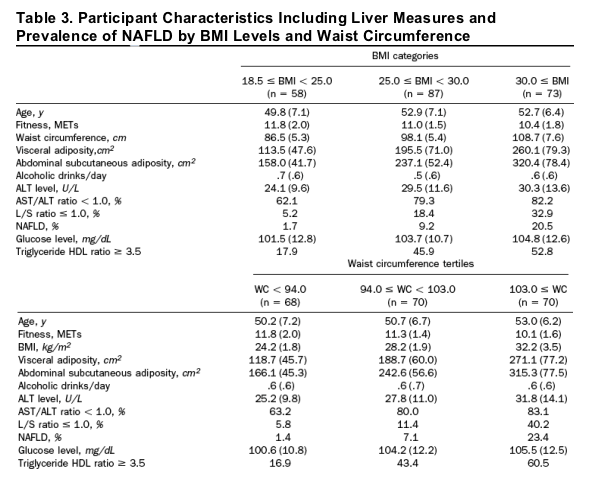
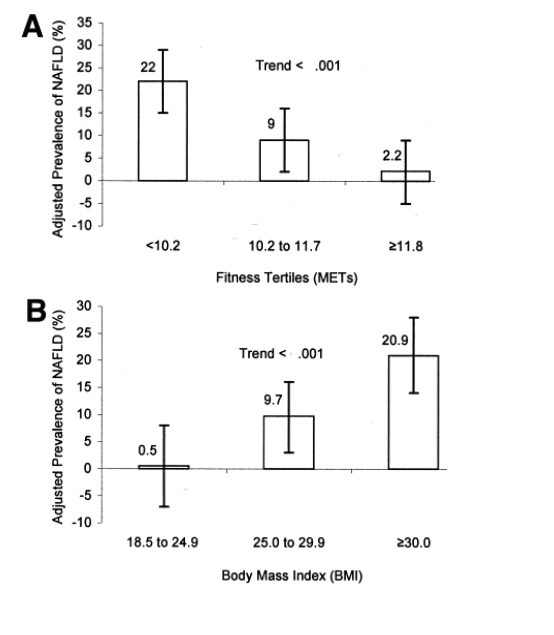
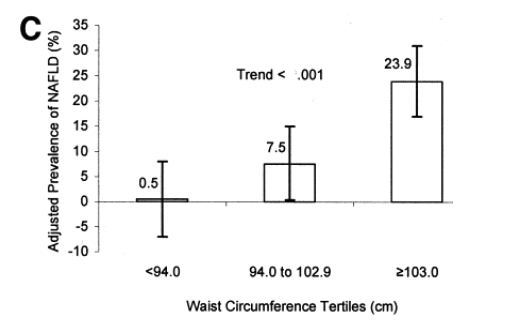
As depicted in Figure 1, the adjusted (age and alcohol use) prevalence of NAFLD was associated inversely across fitness categories (P for trend <.001), and associated positively across BMI and waist circumference categories (P for trend <.001 for each).
Figure 1. Data from 218 men in the Aerobics Center Longitudinal Study. (A) Prevalence of NAFLD across tertiles of fitness; (B) prevalence of NAFLD across BMI categories of normal weight (18.5 kg¥m-2 ≦ BMI < 24.9 kg¥m-2), overweight (25.0 kg¥m-2 ≦ BMI < 29.9 kg¥m-2), and obese (BMI ≥ 30.0 kg¥m-2); and (C) NAFLD across tertiles of waist circumference. Numbers above the bars are the prevalence, and the error bars represent 95% confidence intervals. All data are adjusted for age and alcohol use.
To identify possible biological mechanisms, the association between fitness and odds of prevalence of NAFLD was examined using multivariable logistic regression models, adjusted for age and alcohol, with adjustment for potential mediators (Table 4). This was repeated for BMI and waist circumference. BMI values, abdominal subcutaneous fat values, fasting glucose level, and insulin resistance did not significantly modify the association between fitness and prevalence of NAFLD when each was put into the regression model individually. Both waist circumference and visceral adiposity eliminated the statistical significance of the fitness-NAFLD relationship, suggesting central adiposity is a mediator of the benefit associated with higher levels of fitness. Fitness, abdominal subcutaneous fat value, fasting glucose level, and insulin resistance did not modify significantly the association between BMI and risk of prevalence of NAFLD. However, similar to the fitness-NAFLD relationship, both waist circumference and visceral adiposity eliminated the statistical significance of the BMI-NAFLD relationship. The fitness X BMI interaction term was found to be marginally significant (P = .048) when examined in the regression model including fitness and BMI as predictors of the prevalence of NASH. In regard to the waist circumference values and NAFLD, abdominal subcutaneous fat values did not modify this association; but visceral adiposity eliminated the statistical significance of the waist circumference-NAFLD relationship.
ABSTRACT
Background & Aims: There is a need for more work examining the potential of physical activity and/or weight control as a preventive and/or therapeutic option in the treatment of fatty liver diseases. The purpose of this study was to examine the association between cardiorespiratory fitness, body mass index (BMI), and waist circumference with markers of nonalcoholic fatty liver disease (NAFLD).
Methods: Participants consisted of 218 apparently healthy nonsmoking, nonalcoholic men aged 33-73 years. Cardiorespiratory fitness was assessed by a maximal treadmill test. Liver and spleen density were measured using a computed tomography scan. We defined the presence of NAFLD as the following 3 conditions being met: (1) liver to spleen density of 1.0 or less, (2) serum alanine transaminase level greater than 30 U/L, and (3) serum aspartate transaminase/alanine transaminase level less than 1.0.
Results:
Twenty-four (11%) of the participants met the NAFLD definition.
There was an inverse association between fitness categories, and a positive association for BMI categories (and waist circumference categories) with the prevalence of NAFLD (P for trend <.001 for all).
Fitness and BMI were independent of each other in their associations with the prevalence of NAFLD.
The addition of waist circumference to the regression model attenuated the association with prevalence of NAFLD for both fitness (P value changed from <.0001 to .06) and BMI (P value changed from <.001 to .22).
Conclusions: Fitness (inversely) and BMI (directly) were associated with the prevalence of NAFLD. However, these associations were attenuated when abdominal obesity was included in the statistical model.
METHODS
Patient Data
The Aerobics Center Longitudinal Study is an epidemiologic investigation of patients who received a thorough medical examination at the Cooper Clinic in Dallas, Texas. Data collection for the Aerobics Center Longitudinal Study began in 1970, and to date there are more than 70,000 study participants in the database.16, 17, 18 Most of the participants are non-Hispanic whites, residents of the United States, and are well educated. Participants for this analysis are derived from a previously generated subgroup of men (n = 585) examined during 1996 to 2001 who were selected to explore the associations between visceral fat, subcutaneous fat, fitness, and cardiovascular disease risk factors.19, 20, 21 Criteria for inclusion in this database were age 30-75, nonsmoking, available fitness and anthropometric data, and usable computed tomography scan for visceral and subcutaneous adiposity measurement. The computed tomography scans were performed as part of a coronary artery calcium measurement protocol. To ensure a broad mix of fitness and BMI levels, potential participants were stratified into 3 fitness and 3 BMI categories resulting in 9 fitness-fatness categories. Approximately equal numbers of participants were selected randomly from each fit-fat category. All Cooper Clinic patients have essentially the same routine laboratory measurements performed, but because of changes in the Cooper Clinic laboratory protocol, only 297 of the 585 had alanine transaminase (ALT) levels measured. Other exclusion criteria specific to this analysis included persons with a self-reported history of myocardial infarction or stroke (n = 8), diabetes (n = 5), consumption of 2 or more alcoholic drinks per day (n = 42), liver disease (hepatitis, jaundice, or cirrhosis, n = 21), or other missing data (n = 3), resulting in a study population of 218 individuals.6, 12 The self-report data came from an extensive medical history questionnaire that includes demographic characteristics, health history, and family medical history. The well-educated men in our study are able to report prior diagnoses with considerable accuracy. We earlier verified self-report of hypertension in the cohort and found a sensitivity of 98% and a specificity of 99%.16 All participants gave their informed consent to participate in the clinical examination and for their data to be used for research purposes. The Cooper Institute Institutional Review Board annually reviewed and approved the study protocol.
Clinical Examination
The clinical examinations were administered in the mornings after an overnight fast. The participants also were instructed to refrain from exercise during the day before their examination. The examination consisted of a physical examination by a clinic physician, blood draw, blood pressure measurement, anthropometry, a health habit inventory, and the completion of an extensive questionnaire described previously. Height and weight were measured on a standard physician's balance beam scale and stadiometer. BMI, used as an index of body fatness, was calculated as weight in kilograms, divided by height in meters squared. Waist circumference was measured at the level of the umbilicus using a plastic tape measure.
Cardiorespiratory fitness was assessed by a maximal treadmill exercise test following a modified Balke protocol. Patients began walking at 88 m/min (3.3 mph) with no elevation. After the first minute the incline was increased to 2% and was increased 1% each minute thereafter until the 25th minute. For the few participants still able to continue the test beyond 25 minutes, the elevation was maintained at 25%, and the speed was increased by 5.4 m/min (.2 mph) each minute until the end of the test. The test was terminated when the participants were exhausted or if the physician stopped the test for medical reasons. Time on the treadmill test with this protocol is correlated highly (R = .92) with measured maximal oxygen uptake.22 Thus, cardiorespiratory fitness in this study is analogous to maximal aerobic power (VOmax) and is used here as an objective laboratory marker for exercise participation in the several weeks before the treadmill test. We expressed cardiorespiratory fitness as maximal metabolic equivalents ([METs] or work metabolic rate/resting metabolic rate = 3.5 mL¥kg-1¥min-1) attained during the treadmill test. METs were calculated from estimated VOmax for the Balke protocol using the formula VOmax = 1.44¥(minutes on treadmill) + 14.99.22
Biochemistry Analyses
We obtained blood samples from an antecubital vein and samples were analyzed by automated methods in a laboratory that participates in and meets quality control standards of the Centers for Disease Control and Prevention Lipid Standardization Program. Serum ALT and aspartate transaminase (AST) levels were measured using a Hitachi clinical chemistry system (Boehringer-Mannheim Diagnostics, Indianapolis, IN). The metabolic syndrome was defined using the ATP III guidelines of having 3 or more of the following 5 risk factors: high blood pressure (≥130 mm Hg systolic or ≥85 mm Hg diastolic), central obesity (waist circumference ≥ 102 cm), high TG (≥150 mg/dL), low HDL-C (<40 mg/dL), and high fasting plasma glucose level (≥110 mg/dL).23 The triglyceride to HDL-C ratio was used as an index of insulin resistance with a ratio 3.5 or greater used to identify individuals with insulin resistance.24
Measurement of Liver Fat and Abdominal Subcutaneous and Visceral
Adiposity
The protocol for the measurement of liver fat, abdominal subcutaneous adiposity, and visceral adiposity has been described previously.19, 20 Briefly, axial images of the abdominal region were obtained using an electron beam computed tomography scan (Imatron; General Electric, Milwaukee, WI) following a standard protocol. Approximately 40 transverse images were acquired from the midregion of the iliac crest to the caudal region of the heart. Images were obtained using 130 kV and 630 mA with a 48-cm field of view and a 512 X 512 matrix.
From a single computed tomography image that included both the liver and the spleen, the attenuation values for both the liver and the spleen were obtained by identifying 2 regions of interest within each organ and then calculating the corresponding average density value. Because of the normally higher density of the liver compared with the spleen, a lower mean liver attenuation value relative to that of the spleen indicates fatty infiltration of the liver.25 A ratio of mean liver to spleen attenuation (L/S) values was used as an index of liver fat. A liver to spleen ratio of less than 1 has been validated as an index of fatty change of the liver and is used routinely to diagnose fatty liver.25, 26, 27, 28, 29 We previously reported the CV for repeat liver and spleen attenuation measurements by the same observer (intraobserver) to be 5.2% and 2.8%, respectively, and interobserver CV measurements were 5.1% and 4.3%, respectively.19
Abdominal subcutaneous and visceral adiposity were measured at the L3-L4 vertebral disc spaces using specialized image analysis software (Tomovision, Inc, Montreal, Canada). Adipose tissue areas (cm3) were computed using an attenuation range of -190 to -30 Hounsfield units. Visceral adiposity was determined by delineating the intra-abdominal cavity at the innermost aspect of the abdominal and oblique wall musculature and the posterior aspect of the vertebral body. The abdominal subcutaneous adiposity area was defined as the area of adipose tissue between the skin and the outermost aspect of the abdominal muscle wall.
NAFLD Definition
In creating the surrogate measure for NAFLD we sought to combine a measurement of liver fat with a measure of liver injury that is characteristic of the more serious side of the NAFLD spectrum such as NASH. As noted earlier, an L/S of 1.0 or less is accepted widely as an indication of excess fatty infiltration of the liver, and a recent publication identified an ALT level of 30 U/L as the upper limit of normal in men.25, 26, 30 Further, an AST/ALT ratio of less than 1.0 is a common feature of NASH (present 65%-90% of the time) and very rarely is associated with alcohol-induced liver damage.5, 7, 8, 31 We combined these findings to define the presence of NAFLD as the following 3 conditions being met: (1) L/S of 1.0 or less, (2) ALT value of greater than 30 U/L, and (3) AST/ALT value of less than 1.0. Forty-three (19.7%) participants had an L/S of 1.0 or less, 77 (35.3%) had an increased ALT level, and 165 (75.7%) had an AST/ALT level of less than 1.0. Twenty-five (11.5%) participants had an L/S 1.0 or less and increased ALT value, 39 (17.9%) had an L/S of 1.0 or less and an AST/ALT value of less than 1.0, and 75 (34.4%) had an AST/ALT value of less than 1.0 with an increased ALT value.
Statistical Methods
We computed the arithmetic mean and SD (dichotomous variables presented as percentages) of each variable by the presence or absence of NAFLD and across tertiles of METs and waist circumference, and standard BMI classifications of normal weight (18.5 ≦ BMI < 24.9 kg¥m-2), overweight (25.0 ≦ BMI < 29.9 kg¥m-2), and obese (BMI ≥ 30.0 kg¥m-2).32 Continuous variables were compared using the Student t test and categoric variables were compared using the ƒÔ2 or Fisher exact test where appropriate. Trends across categories were tested using regression models. The age- and alcohol-adjusted prevalence of NAFLD was calculated for MET tertiles, BMI categories, and waist circumference tertiles based on the general linear model. By using logistic regression, the age and alcohol use adjusted odds ratio for having NAFLD was calculated for METs, BMI, and waist circumference with and without adjustment for individual variables that may be mediators. For all statistical tests, the ƒ¿ level adopted for significance was a 2-tailed P value of .05 or less. Statistical analysis was performed with SAS version 8.2 (SAS Institute Inc, Cary, NC).
|
|
| |
| |
|
|
|