| |
Effects of atazanavir/ritonavir and lopinavir/ritonavir on glucose uptake and insulin sensitivity: demonstrable differences in vitro and clinically
|
| |
| |
AIDS: Volume 20(14) 11 September 2006 p 1813-1821
Noor, Mustafa Aa,c; Flint, Oliver Pb; Maa, Jen-Fuec; Parker, Rex Aa
From the aDiscovery and Exploratory Clinical Research, USA
bDiscovery Toxicology, USA
cVirology Medical Affairs Departments, Pharmaceutical Research Institute, Bristol-Myers Squibb Company, Princeton, New Jersey, USA.
Abstract
Background: The HIV protease inhibitor (PI) atazanavir does not impair insulin sensitivity acutely but ritonavir and lopinavir induce insulin resistance at therapeutic concentrations.
Objective: To test the hypothesis that atazanavir combined with a lower dose of ritonavir would have significantly less effect on glucose metabolism than lopinavir/ritonavir in vitro and clinically.
Methods: Glucose uptake was measured following insulin stimulation in differentiated human adipocytes in the presence of ritonavir (2 μmol/l) combined with either atazanavir or lopinavir (3-30 μmol/l). These data were examined clinically using the hyperinsulinemic euglycemic clamp and oral glucose tolerance testing (OGTT) in 26 healthy HIV-negative men treated with atazanavir/ritonavir (300/100 mg once daily) and lopinavir/ritonavir (400/100 mg twice daily) for 10 days in a randomized cross-over study.
Results: Atazanavir inhibited glucose uptake in vitro significantly less than lopinavir and ritonavir at all concentrations. Ritonavir (2 μmol/l) combined with either atazanavir or lopinavir (3-30 μmol/l) did not further inhibit glucose uptake. During euglycemic clamp, there was no significant change from baseline insulin sensitivity with atazanavir/ritonavir (P = 0.132), while insulin sensitivity significantly decreased with lopinavir/ritonavir from the baseline (-25%; P < 0.001) and from that seen with atazanavir/ritonavir (-18%; P = 0.023). During OGTT, the HOMA insulin resistance index significantly increased from baseline at 120 min with atazanavir/ritonavir and at 150 min with lopinavir/ritonavir. The area under the curve of glucose increased significantly with lopinavir/ritonavir but not with atazanavir/ritonavir.
Conclusions: Both glucose uptake in vitro and clinical insulin sensitivity in healthy volunteers demonstrate differential effects on glucose metabolism by the combination PI atazanavir/ritonavir and lopinavir/ritonavir.
Note: Presented in part at the XVth International Conference on AIDS Bangkok, July 2004 and the 7th International Workshop on Adverse Drug Reactions and Lipodystrophy, Dublin, November 2005.
Author Discussion
We examined the effect of low concentrations of RTV combined with ATV or LPV on glucose uptake in vitro and correlated with a clinical study comparing insulin sensitivity in healthy adults treated with the widely used RTV-boosted PI. We found that ATV at a concentration of 3-10 μmol/l did not significantly affect insulin-stimulated glucose uptake whereas LPV as single drug at ≥ 3 μmol/l strongly inhibited glucose uptake in vitro. RTV at 2-3 μmol/l inhibited glucose uptake moderately; significant (i.e., > 50%) inhibition was seen at ≥ 10 μmol/l. At all concentrations tested, ATV combined with RTV (2 μmol/l) had significantly less inhibitory effect on insulin-stimulated glucose uptake than LPV combined with RTV (2 μmol/l).
Clinically, we found that treatment for 10 days with ATV/RTV in healthy HIV-negative subjects did not significantly affect insulin sensitivity, while treatment of the same individuals with LPV/RTV resulted in insulin resistance. The clinical findings are consistent with in-vitro observations comparing LPV and ATV here and reported by other investigators [13]. Additionally, these findings are consistent with euglycemic clamp data in rodents exposed to therapeutic (5-25 μmol/l) concentrations of LPV, RTV, or ATV [21] and confirmatory of a previous observation [16]. Therefore, the findings of insulin-stimulated glucose uptake in vitro and insulin-mediated glucose disposal rates clinically are consistent with a differential effect of ATV/RTV and LPV/RTV on glucose metabolism.
Although induction of insulin resistance has been demonstrated after a single dose of IDV/RTV (800/100 mg), we administered ATV/RTV or LPV/RTV for 10 days prior to performing the clamp. ATV, LPV, and RTV (unlike IDV) are all highly bound to plasma protein, which significantly decreases the percentage of circulating free drug and increases the time required to steady-state plasma concentrations [22]. We administered the combination of drugs for 10 days to ensure achievement of steady-state plasma ATV and RTV concentrations, approximating chronic exposure. We administered the study drugs prior to the clamp in the fasted state, which resulted in 20-25% lower maximum concentration as expected, but the overall plasma levels were comparable to those reported in HIV-positive patients [15]. The crossover study design allowed for direct comparison between treatments within the same subject and minimized intersubject variability in insulin sensitivity.
The magnitude of decrease in insulin sensitivity with LPV/RTV was consistent with our previous study [5]. The average decrease in the MI index (insulin-stimulated glucose uptake per unit insulin) on LPV/RTV was approximately 2.8 (25%) relative to baseline and 1.9 (18%) relative to ATV/RTV. The magnitude of such a difference is clinically significant as it approximates total hepatic glucose production in the fasted state in non-diabetic individuals (approximately 2 mg/kg per min) [23]. Further, the impairment in whole-body glucose disposal of 20 to 25% observed here with LPV/RTV is comparable to the difference found between healthy non-diabetic individuals and patients with type 2 diabetes [24]. Finally, a decrease of similar magnitude in glucose disposal (-30%) was found in HIV-positive patients with clinical lipodystrophy compared with those without lipodystrophy [25]. This observation is consistent with an emerging understanding of adipocyte biology and the relationship between changes in insulin-mediated glucose disposal (via GLUT4) and subsequent release of the fat-derived chemocytokines known to alter fat mass and distribution [26]. We observed no significant decrement in glucose disposal rate from baseline with ATV/RTV, which identifies an important difference between the glucose metabolic effects of ATV/RTV and LPV/RTV.
In contrast to clamp results, we found that both ATV/RTV and LPV/RTV increased the 2-h time point for glucose and insulin during OGTT. The AUC for insulin increased on both regimens, although the AUC for glucose increased only with LPV/RTV. However, using the two more comprehensive indices of insulin sensitivity known to correlate well with euglycemic clamp data [18,19], we found no significant changes from baseline. This is not surprising given that insulin-stimulated glucose disposal measured by the hyperinsulinemic euglycemic clamp is a more appropriate clinical corollary of insulin-stimulated glucose uptake in vitro and available data suggest acute and reversible changes by PI drugs with both techniques.
The apparent discrepancy between the clamp findings and some OGTT parameters may, therefore, be explained by differences between peripheral (i.e., muscle and adipose tissues) and hepatic insulin sensitivity and reflect non-parallel deterioration of the variable measured by the two techniques. Such discrepancies in induction of insulin resistance by a PI have been reported previously [27] and may be a result of an increase in hepatic glucose production stimulated by longer treatment with PI drugs [28]. In this study, we measured whole-body glucose uptake at hyperinsulinemic levels (-400 pmol/l) that are known to suppress hepatic glucose production completely [29]. While this allowed accurate measurement of peripheral insulin sensitivity, it may have also masked a small but physiologically relevant increase in hepatic glucose production. Therefore, we cannot exclude a lack of effect on hepatic glucose production. An increase in hepatic glucose production measured during clamp has been reported at lower levels of hyperinsulinemia (-200 pmol/l) in HIV-positive patients with clinical evidence of lipodystrophy on PI-based HAART [30].
With the standard 75 g glucose dose, the suppression of hepatic glucose production by insulin is expected to be much less complete than during the hyperinsulinemic euglycemic clamp [31]. Therefore, OGTT reflects both contributions from hepatic glucose production and glucose disposal by all tissues in the body to an approximately equal extent. For this reason, a number of insulin sensitivity indices have been developed and have been shown to correlate better with the glucose clamp studies as they encompass the contributions of both the hepatic and peripheral tissues to overall glucose homeostasis [18]. Since both peripheral insulin resistance and hepatic glucose production are affected by PI drugs [7], such an index during OGTT may better reflect the metabolic abnormalities observed in patients with insulin resistance caused by HIV lipodystrophy. Using two such indices, we did not find a statistically significant decrease in insulin sensitivity from baseline with either treatment.
It is worth noting that RTV maximum plasma concentration was over twofold higher with ATV than with LPV and the overall average daily exposure to RTV with ATV was higher relative to that with LPV. In spite of a higher level of exposure to RTV, the combination of ATV/RTV had less effect on overall insulin sensitivity and glucose metabolism. This suggests that both LPV and RTV have independent inhibitory effects on glucose disposal. Regardless of the underlying mechanisms, our findings in vitro and their clinical confirmation in healthy volunteers indicate that the adverse effect on intermediary metabolism by ATV combined with the lower 'boosting' dose of RTV is significantly less than that of LPV/RTV.
This study has potential limitations. We assessed glucose uptake in human adipocytes and observed greater baseline variation between experiments than in previous studies with the mouse 3T3-L1 adipocyte cell line. However, similar effects have been reported in fibroblasts, myocytes, and adipocytes [9,12], and the effects by individual drugs between each replicate experiments were consistent. We measured peripheral insulin sensitivity after only short-term treatment with ATV/RTV and LPV/RTV and did not quantify hepatic glucose production. While this is an acute effect on glucose metabolism in healthy volunteers, ATV-based HAART has not been associated with insulin resistance in clinical trials of HIV-infected patients after 48 weeks of treatment [14], and a RTV-boosted ATV regimen was associated with a lower incidence of dyslipidemia than LPV/RTV [32]. Given that the euglycemic clamp is a research tool, comparisons between HIV-infected patients with and without clinical lipodystrophy found a similar decrease in insulin sensitivity [25]. Whether these acute changes ultimately contribute to the long-term cardiovascular morbidity is not known. However, epidemiological data suggest that morbidity is higher in patients who have developed metabolic complications [33]. Finally, we studied men only; the observed metabolic effects may differ in pre- and postmenopausal women.
In summary, we show that ATV inhibits insulin-stimulated glucose uptake significantly less than LPV at similar concentrations in vitro, and that RTV (2 μmol/l) combined with ATV or LPV does not further inhibit glucose uptake beyond the level seen with individual drugs. We also demonstrate that a lower, 'boosting' concentration of RTV 100 mg given together with ATV does not significantly affect whole-body insulin sensitivity estimated by euglycemic clamp. Although the AUC for insulin and the 2-h time point increased on both treatments, and we did not quantify hepatic glucose production, the more comprehensive indices of insulin resistance during OGTT did not change on either treatment. Future studies are needed to assess how these observations translate to differences in patients receiving HIV PI drugs.
Results
In-vitro studies
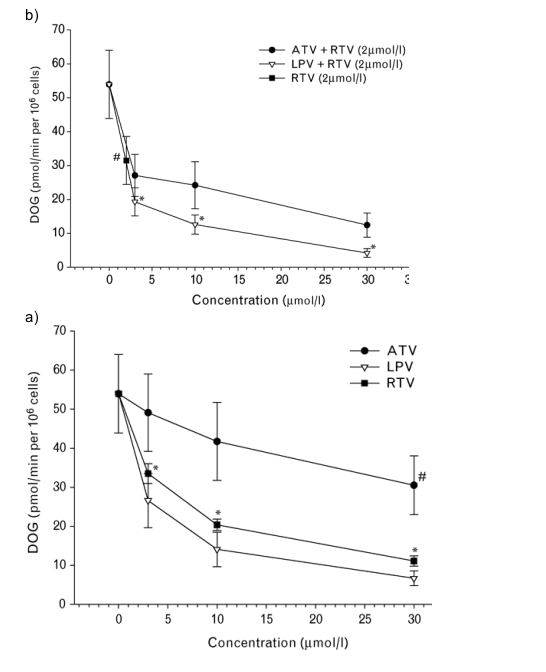
All three PI drugs tested inhibited insulin-stimulated glucose uptake in human adipocytes in a concentration dependent manner at therapeutic concentrations (≥10 μmol/l). As single drugs at a concentration of 3 μmol/l, RTV and LPV but not ATV rapidly inhibited insulin-stimulated glucose uptake, and ATV had significantly less inhibitory effect than the other two PI at the same concentrations (Fig. 1a). Ritonavir was tested at the concentration of 2 μmol/l in combination studies to model its maximum concentration under the respective 'boosting' conditions, with either ATV or LPV at therapeutic concentrations (≥ 3 μmol/l). At this concentration (2 μmol/l), RTV inhibited insulin-stimulated glucose uptake to the same extent as 3 μmol/l. However, the combination of LPV/RTV was significantly more potent at inhibiting glucose uptake than ATV/RTV at all concentrations (Fig. 1b).
Fig. 1. The effect of HIV protease inhibitors on glucose uptake in differentiated human adipocytes. (a) Atazanavir (ATV), ritonavir (RTV) and lopinavir (LPV) were added to a final concentration of 3, 10, and 30 μmol/l as indicated 10 min prior to the glucose uptake assay. [14C]-Deoxy-D-glucose (DOG) uptake was measured for 10 min at 37 C under insulin-stimulated conditions (0.4-0.6 μmol/l insulin for 10 min). The data (mean ±SEM; n = 9), representative of five independent experiments, were normalized to the value obtained from insulin-stimulated control cells. # P < 0.01 versus no drug control; * P < 0.01 versus ATV. (b) Differentiated human adipocytes were treated with RTV at 2 μmol/l combined with either ATV or LPV at 3, 10, and 30 μmol/l and DOG uptake was measured as described above. The data (mean ±SEM; n = 6) were normalized to the value obtained from insulin-stimulated control cells. # P ≦ 0.01 versus no drug control; * P ≦ 0.01 versus ATV+RTV. All statistical comparisons were by Dunnet's test.
Clinical studies
Twenty six subjects were enrolled and randomized. One subject was excluded because of an upper respiratory infection unrelated to treatment. Twenty-five subjects had available baseline and on-treatment data. The clamp data from one subject, and from another during the ATV/RTV treatment only, were considered invalid owing to technical complications with the procedures and excluded from the analysis. The OGTT data from one subject during the LPV/RTV treatment were considered invalid owing to protocol violation and were excluded from the OGTT analysis. The subjects were all male and ranged in age from 24 to 47 years (mean, 34.2; SEM 7.3); 13 were Caucasian non-Hispanic, six African-American, and six Hispanic/Latino. There were no serious adverse clinical events during the course of the study. Baseline body weight, fasting serum insulin, plasma glucose, lipids, and lipoprotein levels were within normal range (Table 1).

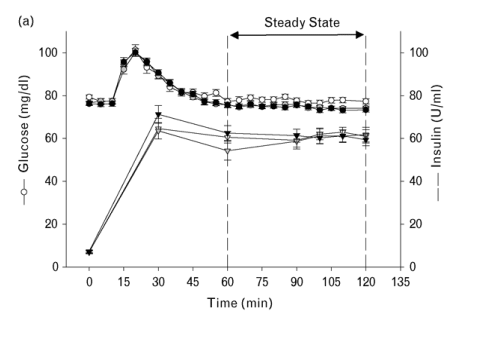
During the last hour of the clamp, the average plasma concentrations for the administered drug were near peak levels (Fig. 2a,b). The geometric means for maximum plasma concentrations were 4.6/1.2 μmol/l for ATV/RTV and11.3/0.5 μmol/l for LPV/RTV. The geometric means for average daily concentration (reflecting chronic exposure) were 2.0/0.35 μmol/l per 24 h for ATV/RTV and 7.7/0.25 μmol/l per 24 h for LPV/RTV. A primed-continuous infusion of insulin achieved steady-state serum insulin levels of approximately 60 μU/ml (430 pmol/l) after 30 min. Steady-state whole blood glucose levels of approximately 750 mg/l (4.3 mmol/l) were reached at 60 min with a variable infusion of 20% glucose and maintained for the duration of the study under all conditions (Fig. 3a). The adjusted means of insulin-stimulated glucose disposal per unit of insulin (M/I; mg/kg per min per μU/ml insulin) at baseline and after treatment with ATV/RTV and LPV/RTV were 11.5 (SEM, 1.0), 10.4 (SEM, 1.1), and 8.6 (SEM, 0.8), respectively (Fig. 3b). There was no significant change from baseline M/I after treatment with ATV/RTV (-1.1; 95% CI, -2.72 to 0.41; P = 0.132). The M/I index decreased significantly (25%) from baseline after treatment with LPV/RTV (-2.8; 95% CI, -4.15 to -1.68; P < 0.001) and was significantly lower than that on ATV/RTV [-1.8 (-18%); P = 0.023).
Fig. 2. Plasma pharmacokinetic profiles ( n = 25) of atazanavir (ATV), lopinavir (LPV) and ritonavir (RTV) during the hyperinsulinemic euglycemic clamp, dosing with ATV/RTV (a) or LPV/RTV (b). The glucose disposal rate was measured 2-4 h after dosing. All drugs were administered in the fasted state. (1 μmol/l drug is 705 ng/ml ATV, 629 ng/ml LPV and 721 ng/ml RTV).
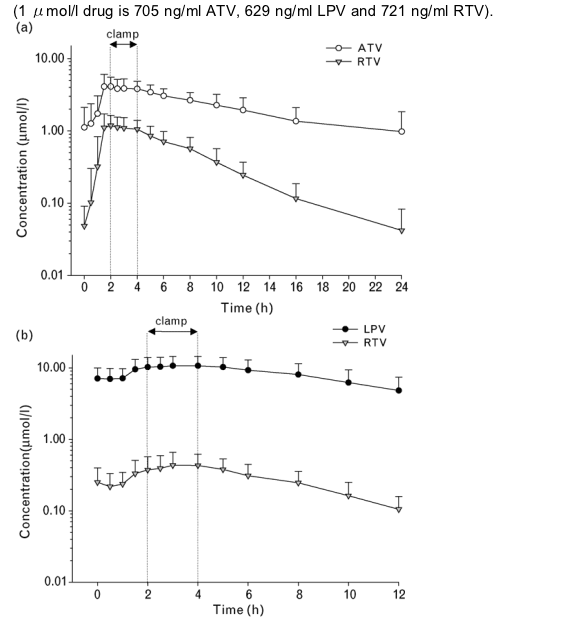
Fig. 3. Clinical assesement of insulin sensitivity. (a) Mean steady-state plasma insulin and glucose concentrations during the hyperinsulinemic euglycemic clamp in subjects studied at baseline (open symbols; n = 24) and after 10 days of dosing with atazanavir/ritonavir (ATV/RTV; shaded symbols; n = 23) or lopinavir/ritonavir (LPV/RTV; closed symbols; n = 24). Steady state was defined as the period between 60 and 120 min. (b) Mean insulin-mediated glucose disposal rate (MI: mg/min per kg body weight per μU/ml insulin) during steady state (60-120 min) at baseline (n = 24) and after 10 days treatment with ATV/RTV (n = 23) or LPV/RTV (n = 24). (c) Mean HOMA insulin resistance index during oral glucose tolerance testing at baseline and after 9 days of treatment with ATV/RTV or LPV/RTV. (d) Mean changes in the area under the curve from 0 to 180 min (AUC0-180) in insulin (μU min/dl) and glucose (mg min/dl) versus baseline after treatment with ATV/RTV or LPV/RTV. (To convert insulin from μU/dl to pmol/l, multiply by 0.7175. To convert glucose from mg/dl to mmol/l, multiply by 0.05551.) Bars indicate SEM.
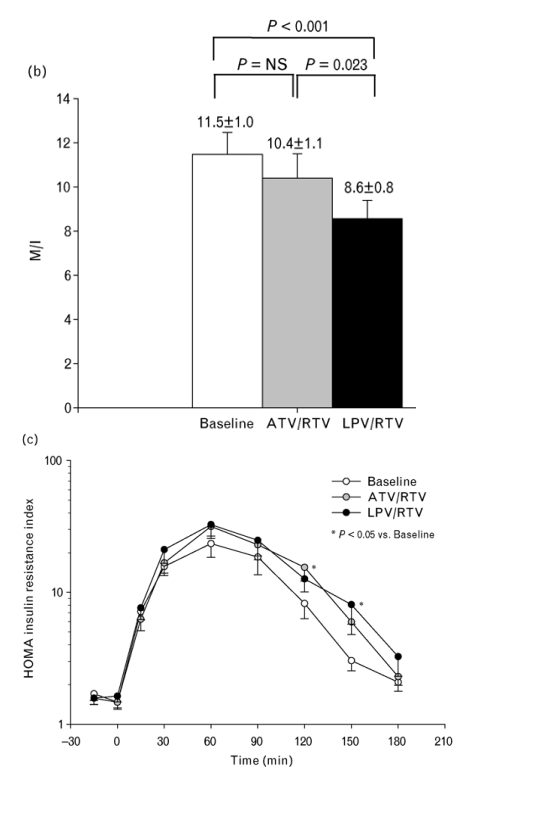
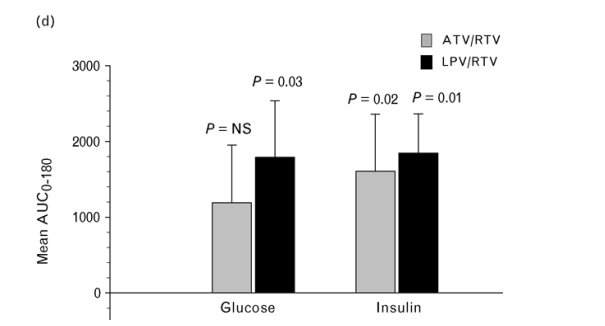
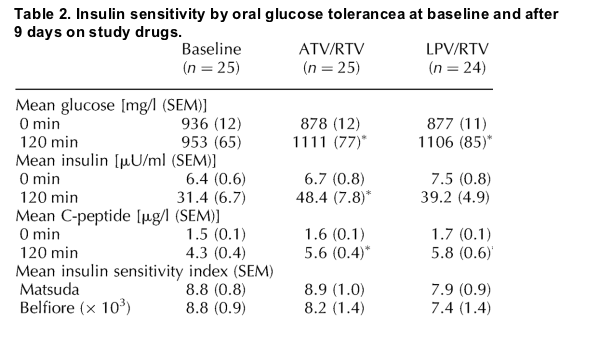
During OGTT, the adjusted means of glucose for the 2-h time point at baseline and with ATV/RTV or LPV/RTV were 953 (SEM, 6.5), 1111 (SEM, 7.7) and 1106 mg/l (SEM, 8.5), respectively (P < 0.02 versus baseline for both treatments; Table 2). The HOMA-IR index was increased at 120 min for ATV/RTV and at 150 min for LPV/RTV (P < 0.05 for both; Fig. 3c) The insulin AUC increased significantly from baseline for both regimens (Fig. 3d). However, the increase in glucose AUC from baseline was significant only with LPV/RTV. The insulin sensitivity index was not changed significantly from baseline for either drug pair, although there was a trend (P = 0.2) towards decreased sensitivity on LPV/RTV (Table 2).

Methods
In-vitro glucose uptake
Human subcutaneous pre-adipocyte cells were obtained from Zen-Bio (Research Triangle Park, North Carolina, USA). The primary pre-adipocytes in tissue-culture 48-well plate format were induced to differentiate into adipocytes using medium containing IBMX (1-methyl-3-(2-methylpropyl)-7H-purine-2,6-dione; Sigma-Aldrich, St Louis, Missouri, USA), dexamethasone, insulin, and rosiglitazone for 3 days, followed by 3 days in medium containing insulin and rosiglitazone alone. On day 11-13 of culture, 18 to 24 h prior to assay, cells were incubated in low-glucose DMEM; for 1 h prior to assay, they were incubated in serum-free DMEM without glucose. For the glucose uptake assay, the medium was first supplemented with insulin (400-600 nmol/l) followed 10 min later by individual PI (ATV, LPV, or RTV). Uptake of [14C]-deoxy-D-glucose (- 20 μmol/l; Perkin-Elmer, Wellesley, Massachusetts, USA) was measured over an additional 10 min. Total exposure time to drug was 20 min. Results (in picomole/liter) were standardized against time (minutes) and cell number (106 cells). The background glucose uptake was subtracted using measurement of glucose uptake in the presence of 100 μmol/l cytochalasin B, as described by Murata et al. [6]. The same human donor was used to provide cells for these studies; however, each batch had to be separately differentiated for 6 to 8 days longer than was required for 3T3-L1 differentiation we reported earlier [10].
Clinical studies of insulin sensitivity
Healthy HIV-seronegative adults gave informed consent and were enrolled in the study provided that a screening physical examination was normal and their weight was reported to be stable over the preceding 6 months. All subjects had a negative HIV-1 antibody test prior to the study. Exclusion criteria included: body mass index > 30 kg/m2, serum total cholesterol > 6.2 mmol/l (2.4 g/l), triglycerides > 3.84 mmol/l (2.0 g/l), fasting glucose > 7 mmol/l (1.26 g/l), serum aspartate or alanine aminotransferases > 50 U/l and creatinine > 107 μmol/l (14 mg/l).
Study design
This was a randomized, two-treatment, two-period crossover study. All enrolled subjects were admitted to the Bristol-Myers Squibb Clinical Pharmacology Unit (CPU) as inpatients. Subjects were assessed at baseline by an OGTT and a euglycemic, hyperinsulinemic clamp on 2 consecutive days. The baseline evaluations were followed immediately by 10 days of treatment as inpatient on two occasions separated by a wash out phase of at least 7 days. Treatments were randomly assigned as a combination of commercially available HIV protease inhibitors: ATV 300 mg (Reyataz Bristol-Myers Squibb, Princeton, New Jersey, USA) and RTV 100 mg (Norvir, Abbott Laboratories, Abbott Park, Illinois, USA) once daily, or LPV/RTV 400/100 mg (Kaletra capsules, Abbot Laboratories) twice daily. All study drugs were administered under direct supervision with a light meal except on days when an OGTT or clamp was scheduled. On these days (days 9 and 10 of each period) the morning dose of the study drug was administered in the fasting state (> 10 h) with 240 ml water 1 h prior to the procedure. On day 11, the subjects were discharged from study drugs and instructed to return for repeat studies on the alternate treatment. The subjects were placed on a diet containing at least 150 g carbohydrates daily and abstained from vigorous exercise throughout the study.
Measurements
Blood samples were collected in the fasted state just prior to the clamp procedure and measured by an enzymatic colorimetric method (Roche Diagnostics, Indianapolis, Indiana, USA). Whole blood and plasma glucose were measured using a glucose analyzer (YSI 2300 STAT-Plus, YSI Inc., Yellow Springs, Ohio, USA). Serum insulin levels and C-peptide were determined by radioimmunoassay (Diagnostics Product Corp., Los Angeles, California, USA) with an upper limit of interassay coefficient of variation of 5.2% and an intra-assay coefficient of variation of 5.1% for insulin. ATV, LPV, and RTV concentrations in plasma were measured by liquid chromatography coupled with tandem mass spectrometry with a lower limit of quantification of 1.00 ng/ml for ATV and 5.00 ng/ml for LPV and RTV.
Oral glucose tolerance test
At 8:00 a.m., after a 10 h overnight fast, the subjects received 75 g glucose orally. Blood samples were collected at -15, 0, 15, 30, 60, 90, 120, 150, and 180 min for the measurement of plasma glucose (sodium fluoride-containing tubes) and serum insulin. The areas under the curve (AUC) for glucose and insulin were calculated by trapezoidal estimation integrating from 0 to 180 min. Fasting and post-glucose plasma glucose and serum insulin levels were used to calculate the insulin resistance value for each time point by the homeostasis model assessment (HOMA): [serum insulin (μU/ml) X plasma glucose (mmol/l)]/22.5 [17]. The Matsuda index [18] was calculated as [10 000/square root of (fasting glucose X fasting insulin) X (mean glucose X mean insulin) during OGTT] and the Belfiore index was calculated as [2/(mean OGTT glucose concentration X mean OGTT insulin concentration)+1] [19].
Hyperinsulinemic euglycemic clamp
The clamp was performed as described by DeFronzo et al. [20] and as reported earlier by us [5]. At time 0 min, insulin (Humulin R, Eli Lilly, Indianapolis, Indiana, USA) was administered as a primed continuous intravenous infusion, followed by a constant infusion at 40 μU/m2 per min for 120 min. Whole blood glucose concentration was measured every 5 min after the start of the insulin infusion. A variable infusion of 20% dextrose was used to maintain plasma glucose concentration at euglycemia (approximately 750 mg/l) with a coefficient of variation < 5% based on the negative feedback principle. Blood samples were collected for post-hoc determination of serum insulin concentrations. Insulin data from visibly hemolyzed specimens (approximately 2.5% of 514 data points) were excluded from final calculations.
Statistical analyses
Analysis of variance, followed by Dunnet's post-hoc test for the difference between means, was used to analyze the in-vitro data. In the clinical study, the primary endpoint was the difference from baseline in the mean glucose disposal rate per unit of insulin (MI) for ATV/RTV and LPV/RTV. Twenty four subjects were required to achieve 80% power for detecting a 20% difference (approximately 2.3 mg/kg per min per μl/ml insulin) between ATV/RTV and LPV/RTV based on change from the baseline glucose disposal rates. Analysis of variance (ANOVA) for the two-period, two-treatment crossover design was used to compare the effects of treatment. The factors in ANOVA model were sequence, subject within sequence, period, and treatment. For each variable, adjusted mean and 95% confidence intervals (CI) were calculated. P values were evaluated at the two-tailed 0.05 level. Data are presented as mean ±SEM unless otherwise specified.
|
|
| |
| |
|
|
|