 |
 |
 |
| |
Tenofovir and Lopinavir Levels in Pregnant Women and Infants
|
| |
| |
46th ICAAC, September 27-30, 2006, San Francisco
Mark Mascolini
Two ICAAC studies yielded new insights on tenofovir and lopinavir levels during pregnancy and in children under 2 years old.
High tenofovir levels in cord blood and amniotic fluid
Of six antiretrovirals measured in 13 pregnant women, tenofovir attained the highest concentrations in cord blood and amniotic fluid at delivery [1]. Although the study is small, the authors believe its results suggest that tenofovir accumulates in the fetal compartment and may help prevent mother-to-child transmission, especially when cesarean section is not possible.
Rosa Yeh and coworkers from the University of North Carolina at Chapel Hill studied 13 HIV-infected pregnant women without opportunistic infections, serious bacterial complications, or current or former obstetrical complications. All women were taking AZT and 3TC, 7 were taking lopinavir/ritonavir, 5 nelfinavir, and 4 tenofovir. The researchers collected multiple samples in the first, second, and third trimesters of pregnancy, in cord blood and amniotic fluid at delivery, and 2 to 12 weeks after delivery.
For all drugs studied except lopinavir, genital tract concentrations proved similar during pregnancy to those measured in earlier studies of nonpregnant women. Genital tract levels of lopinavir were significantly lower in the second trimester (P = 0.03) than in nonpregnant women and almost significantly lower in the third trimester (P = 0.08) and after delivery (P = 0.06). Genital tract-to-blood plasma ratios were similar in this study group and nonpregnant women, except for nelfinavir. Nelfinavir concentrations tended to be lower in the second trimester and after delivery than in nonpregnant women (P = 0.07).
Except for nelfinavir and tenofovir, antiretroviral cord blood and amniotic fluid concentrations were similar in this study and earlier studies (Table). Nelfinavir cord-to-blood plasma ratios proved higher in this study (0.7, interquartile range 0.5 to 0.8) than in an earlier report (median 0.27, range 0.07 to 0.43 [2]).
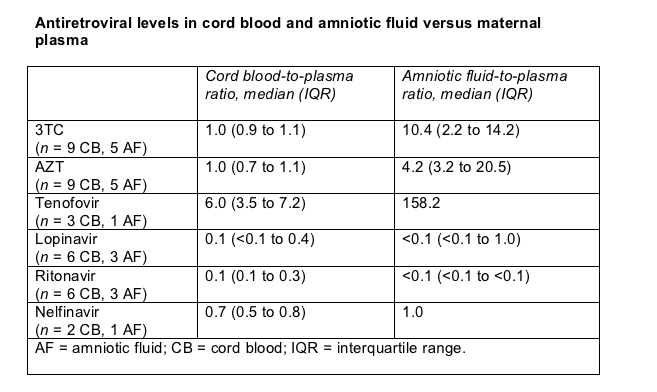
Cord-to-blood plasma ratios of tenofovir were higher in these women taking multiple doses of that drug (Table) than in women taking single-dose tenofovir in an earlier study [3]. Indeed, cord-to-blood plasma ratio and amniotic fluid-to-blood plasma ratio were higher for tenofovir than for any other antiretroviral studied, although this study included only three cord blood samples and one amniotic fluid sample for tenofovir. Yeh and colleagues suggested these findings point to accumulation of tenofovir in cord blood and amniotic fluid during pregnancy.
Lopinavir levels low in children under 2 years
Although the US license for lopinavir/ritonavir sanctions the PIs' use in children older than 6 months, a 23-child study in the Netherlands found low minimum concentrations of lopinavir in most children under 2 years old [4]. Dose adjustments or better food intake corrected low concentrations in all children.
David Burger of Radboud University Nijmegen Medical Center measured lopinavir levels 2 to 4 weeks after 23 children started taking the licensed lopinavir/ritonavir dose of 230/57.5 mg/m2. All children took their PIs with a standardized breakfast, and Burger measured drug levels before dosing and at seven points in the 12 hours after dosing.
The study group included 4 children under 6 months old, 3 who were 6 months to 2 years old, and 17 who were more than 2 years old. (One child was excluded from the analysis.) Eight were girls, 12 were African, 10 had a mixed racial background, and 2 were white. Fifteen took lopinavir oral solution, 6 took soft-gel capsules, and 4 took both.
Average lopinavir levels for the group proved similar to those listed in lopinavir prescribing information. But 5 of 7 children under 2 years old had a subtherapeutic level, defined as a minimum concentration below 1.0 mg/L, compared with 2 of 16 children older than 2, a significant difference (P = 0.011). Improving food intake in 2 children and raising the lopinavir/ritonavir dose approximately 37% in 5 brought minimum concentrations into the therapeutic range in all children.
Burger called for prospective pharmacokinetic studies of higher lopinavir doses in children under 2 years old.
Mark Mascolini writes about HIV infection (markmascolini@earthlink.net).
References
1. Yeh RF, Patterson KB, Dumond JB, et al. Genital tract, cord blood, and amniotic fluid exposures of 6 antiretroviral drugs during pregnancy and postpartum. 46th ICAAC. September 27-30, 2006. San Francisco. Abstract A-378.
2. van Heeswijk RP, Khaliq Y, Gallicano KD, et al. The pharmacokinetics of nelfinavir and M8 during pregnancy and postpartum. Clin Pharmacol Ther 2004;76;588-597.
3. Rodman J, Flynn P, Shapiro D, et al. 13th Conference on Retroviruses and Opportunistic Infections. February 5-8, 2006. Denver. Abstract 708.
4. Burger DM, Verweel G, Sheehan N, et al. Plasma concentrations of the HIV protease inhibitor lopinavir are suboptimal in children aged two years and below. 46th ICAAC. September 27-30, 2006. San Francisco. Abstract A-386.
|
| |
|
 |
 |
|
|