 |
 |
 |
| |
TDM for ART Hepatoxicity
|
| |
| |
Reported by Jules Levin
Hello from the 2nd International Workshop on HIV and Hepatitis Co-infection in Amsterdam (Jan 12-14, 2006). The meeting started today with several well done introductory talks reviewing Immunopathogenesis of viral hepatitis in the coinfected patient (Andy Talal, Cornell Hosp in NYC) and Natural History of Hepatitis C/B in HIV Coinfection (M Puoto, Brescia, Italy). Puoto and other presenters today emphasized that asides from AIDS, liver disease in HIV is the most serious condition in leading to complications and death, making this Workshop very important since it's the only venue devoted to coinfection. This meeting can be an important venue for discussions regarding treatment for coinfected patients and a source for treatment and policy recommendations. There was a very good talk by Doug Dieterich (NYC) on fatty liver in HIV, and how important its presence is in accelerated HCV disease progression and impairing response rates to peginterferon and ribavirin; fatty liver is completely over looked among treaters. Dieterich discussed the importance of assessing lipid abnormalities and insulin resistance in coinfected patients, these 2 conditions are risk factors, and treatment for fatty liver: glitazones, diet, and exercise. In a panel discussion we talked about the importance of evaluating the stage of liver disease coinfected patients before starting HAART, so you can be aware if a patient has cirrhosis or not before starting HAART-there are important implications to stage of liver disease.
An important research paper was presented by Stephanie Dominguez from a French research group. A discussion of TDM to evaluate ART drug levels in patients with cirrhosis emerged following this presentation. I think it was roundly agreed that TDM can be useful and should be utilized for this situation:
"Nevirapine Plasma Concentration in HIV/HCV and HIV Infected Patients, a Case Control Study: NEVADOSE"
Dominguez S, Peytavin G, Falconer K, Cassard B, Boubezari I, Valantin MA, Ghosn J, Tubiana R, Poynard T, Katlama C, Benhamou Y (Paris, France)
AUTHOR CONCLUSIONS
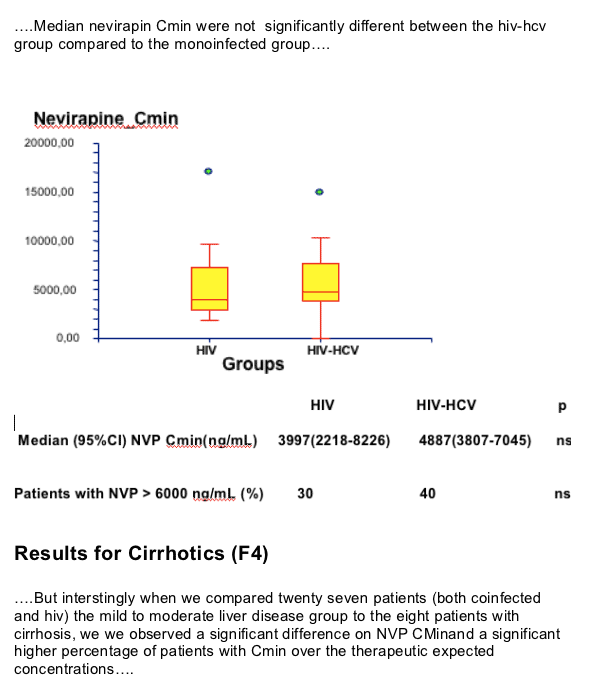
--median NVP Cmin were similar in HCV/HIV and HIV patients
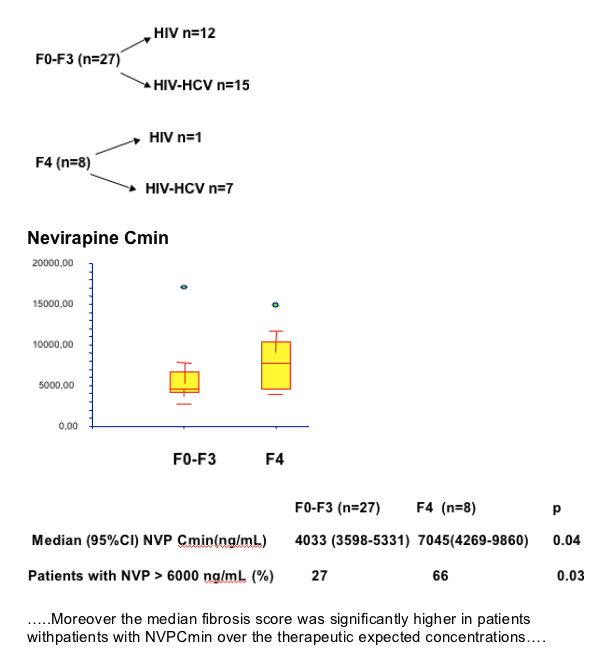
--median NVP Cmin were significantly higher in patients with cirrhosis (F4)
--significant overdosage was observed in patients with cirrhosis
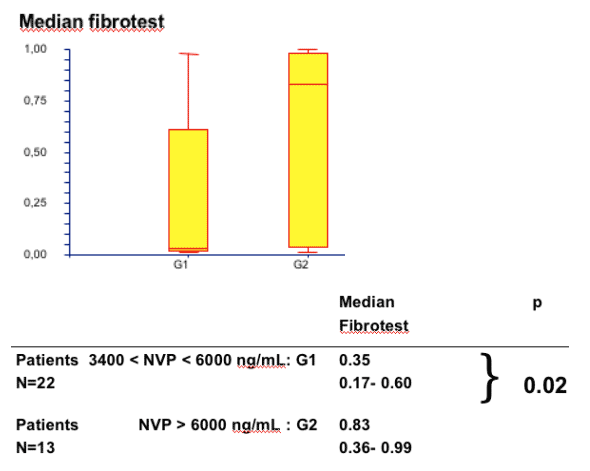
--Fibrotest was significantly different between patients with adequate concentrations vs patients over the expected therapeutic concentrations (note from Jules Levin: in other words, using Fibrotest the study found more advanced fibrosis in coinfected patients with high NVP levels).
--NVP could be safely used in HIV and HIV/HCV patients with mild to moderate liver disease
--however, TDM is useful in both HIV and HCV/HIV patients
--more data are warranted to confirm our results in larger studies
(NOTE from Jules Levin: in talking with Dominguez after her talk she mentioned that at last year's Workshop she presented study data finding similar results when examining nelfinavir and efavirenz levels in cirrhotic coinfected patients, but that levels of lopinavir (RTV boosted) were not increased in coinfected patients.
Background provided by Dominguez
Nevirapine (NVP)
--Has been associated with ALT elevationv
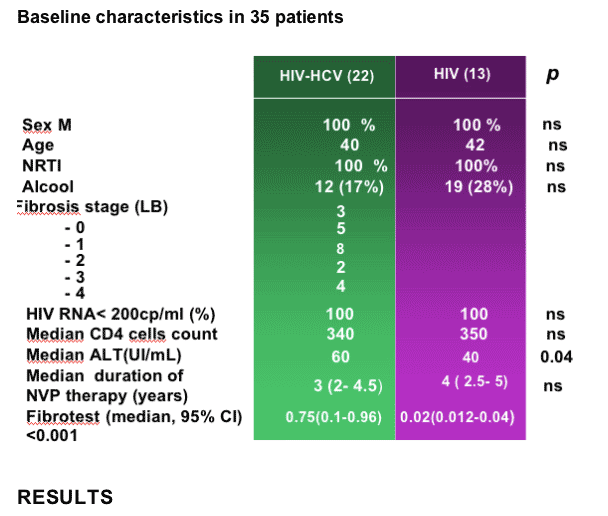
--Prevalence of ALT elevation was found higher in HIV/HCV coinfected vs HIV monoinfected patients (Sulkowski MS, Hepatology 2002;35: 182-9)
Limited datas exist on the impact of liver disease on the pharmacokinetics of NNRTIs
NNRTIs are lipophilic drugs that are metabolized by Cytochrome P450 (CYP)
ARV clearance may be decreased according to hepatic impairment3
Interactions between HCV-related liver lesions and NVP plasma concentration may explain the severity of the underlying liver disease
Liver fibrosis may progress faster in HIV/HCV coinfected patients receiving NVP4
STUDY OBJECTIVE
To analyze the potent relationship between NVP plasma concentration and liver fibrosis related to chronic HCV infection.
METHODS
Cross sectional and prospective study conducted in 2004-2005
Patients
--Receiving NVP (200 mg bid)
--HIV/HCV coinfected and recent interpretable liver biopsy (<2 years)
--HIV monoinfected
--HIV and HIV/HCV patients were matched according to sex and antiretroviral regimen
Measurements
--NVP trough plasma concentrations at steady state (Cmin) were measured using HPLC method.
--Normal range for NVP Cmin :3400-6000 ng/ml
--Liver lesions were assessed by the METAVIR scoring system
--We used the Fibrotest ‚ to evaluate the fibrosis stage at time of NVP Cmin measurement
--CD4 cell count was measured by flow cytometry
--HIV RNA was quantified by Amplicor Roche
Comparisons of NVP Cmin between HIV and HIV/HCV and between patients with and without cirrhosis were performed by the Wilcoxon Rank Test
Variables are expressed as medians and 95% CI
No drug adjustments were performed
|
|
| |
| |
|
 |
 |
|
|