 |
 |
 |
| |
Pioglitazone Improves Limb Fat Loss
|
| |
| |
Reported by Jules Levin
8th Intl Workshop on Adverse Drug reactions and Lipodystrophy in HIV
San Francisco, Sept 26, 2006
This morning was the final session at the Lipodystrophy Workshop and the best. There were presentations of 4 potentially beneficial therapies. There was an update on the pioglitazone for lipoatrophy study previously reported at CROI in Feb 2006. There was a presentation showing leptin improved visceral fat accumulation in the belly, insulin resistance in the liver, and dyslipidemia. Acipimax improved triglycerides and glucose homeostatis. I will report results from these studies. There was an important discussion today following up on the discussion here yesterday regarding insulin resistance and diabetes. The results of the DREAM Study were recently published in the Lancet journal. This study found that rosiglitazone significantly reduced the risk of progressing to diabetes in individuals with impaired glucose tolerance. The discussion was about whether intervention with rosiglitazone in individuals with impaired glucose tolerance is advisable. This appears controversial, but I think it was generally agreed that a study is interesting. As you know, insulin resistance is associated with elevated triglycerides, with visceral fat accumulation, and with heart disease.
Effect of pioglitazone on limb fat and circulating adipokines in HIV-related lipodystrophy
(ANRS 113: Lipiot)
Parts of these results were previously presented at the CROI 2006.
Jacqueline Capeau (INSERM U680, Universite Pierre et Marie Curie, Paris; Tenon University, Paris) reported the study results today at the Workshop.
Study Aim
To assess the safety of pioglitazone 30 mg qd in well-controlled lipoatrophic HIV-1 infected adults receiving antiretroviral therapy.
To measure the effects of this treatment on limb fat mass and adipokine level, over 48 weeks.
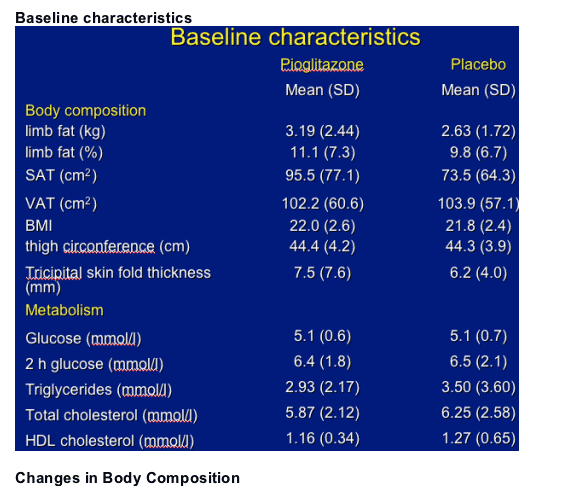
130 patients randomized: 64 in the pioglitazone and 66 in the placebo group
-- 28% on d4T/stavudine (25% in the pioglitazone et 25%in the placebo group)
Study End points
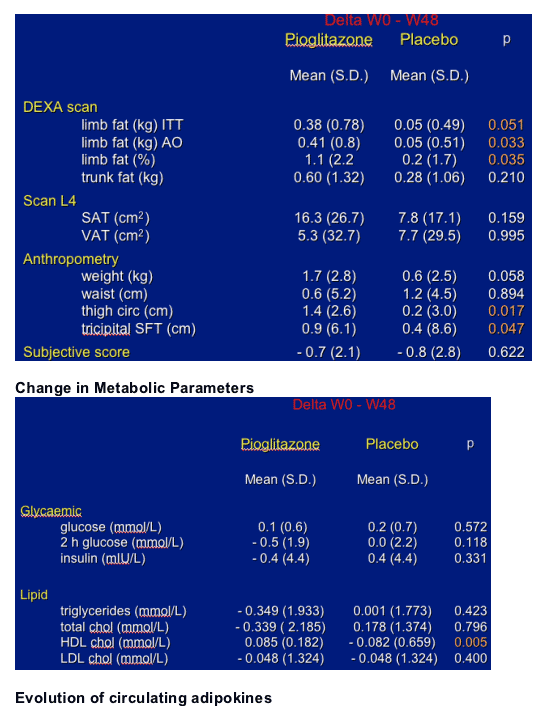
Primary: change in limb fat between week 0 and 48 (DEXA).
Secondary:
- change in SAT and VAT (CT Scan)
- change in glucose and lipid parameters
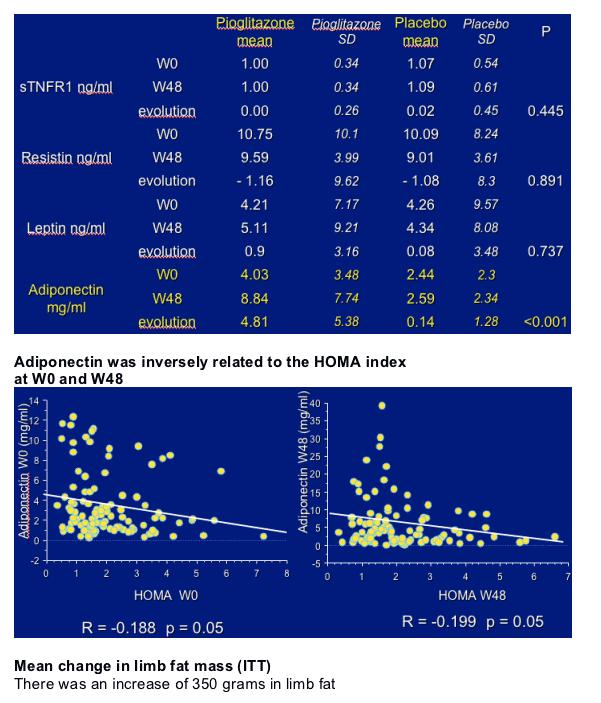
- change in adipokines
- change in subjective discomfort
The authors concluded:
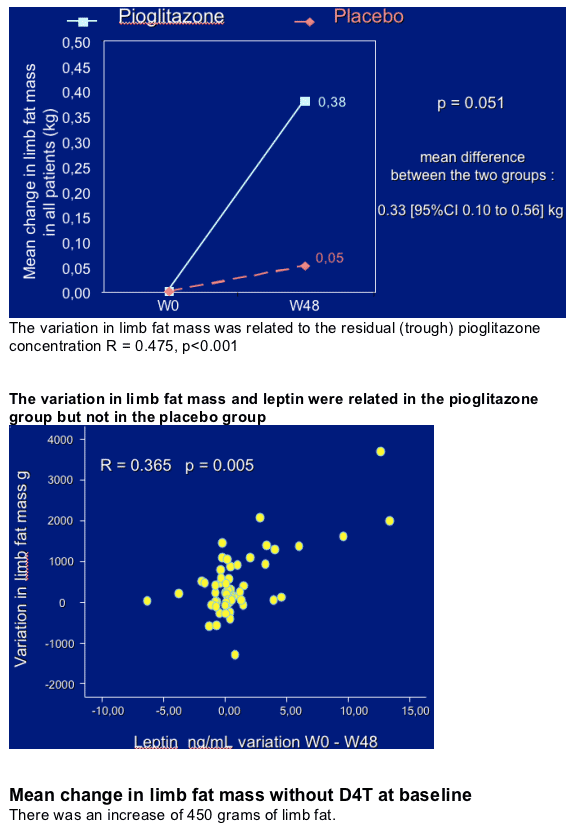
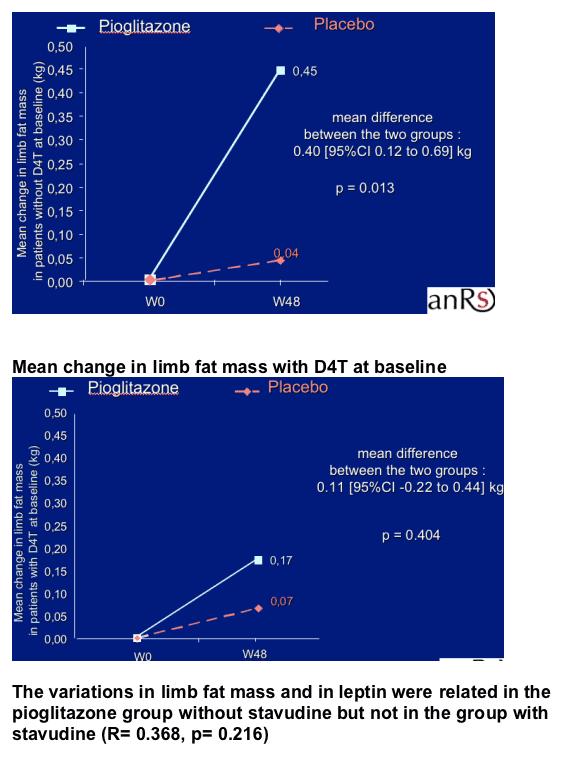
Pioglitazone 30 mg daily for 48 weeks had a beneficial effect on limb fat mass with a mean increase of 380 g while no significant change was observed in patients in the placebo group.
Pioglitazone did not modify lipid profile except a favourable increase in HDL cholesterol.
Pioglitazone was well tolerated without any adverse effects related to the drug.
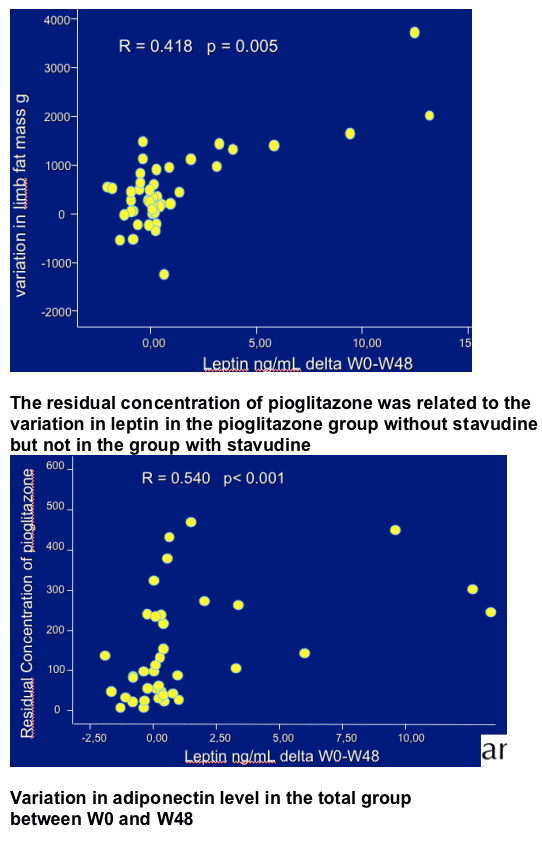
The variations in limb fat mass and leptin were related together and also related to the residual pioglitazone concentration.
In the pioglitazone group without stavudine, the mean limb fat increase was of 450 g and was related to the increase in circulating leptin. The variation in leptin was also related to the residual pioglitazone concentration. These variations were not observed in the group with stavudine.
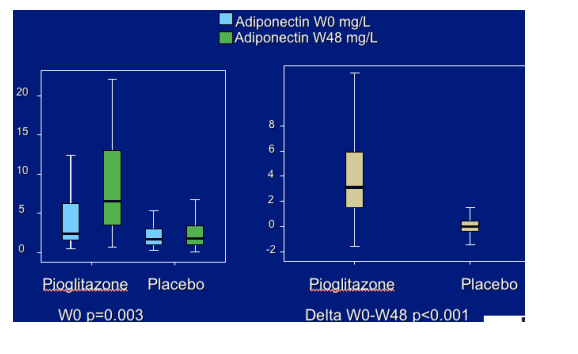
Pioglitazone 24 and 48 weeks significantly increased adiponectin levels: these variations were observed both in the patients without and with stavudine.
The circulating levels of leptin, IL6, resistin and sTNFR1 were not modified.
This study supports the use of pioglitazone in the treatment of peripheral lipoatrophy. Pioglitazone has a positive impact on limb fat mass and adipose tissue endocrine functions by improving adiponectin secretion. Higher dosages of pioglitazone and longer treatments should be considered.
Study Design
Double-blind, placebo-controlled, multicentre, 48 weeks study.
Stratified by sex.
Eligibility
- HIV lipoatrophy self-reported by the patient and confirmed by the investigator
- HIV-1 RNA < 400 cop/ml for last 6 months
- Stable ART for the previous 6 months
- CD4 cells count > 200/mm3
- No anti-diabetic or lipid-lowering drugs
|
| |
|
 |
 |
|
|