 |
 |
 |
| |
Robust Synergistic Antiviral Effect of R1626 in Combination with Peginterferon alfa-2a (40KD), with or without Ribavirin, Interim Analysis Results of Phase 2a Study
|
| |
| |
Reported by Jules Levin
AASLD, Nov 2-6, 2007, Boston, MA
Paul J. Pockros, David Nelson, Eliot Godofsky, Maribel Rodriguez-Torres, Greg Everson, Michael W. Fried, Reem H. Ghalib, Stephen A. Harrison, Lisa M. Nyberg, Mitchell L. Shiffman, George Z. Hill, Anna Chan
AUTHOR CONCLUSIONS
Robust synergistic antiviral effect of R1626 in combination with PEGASYS and RBV
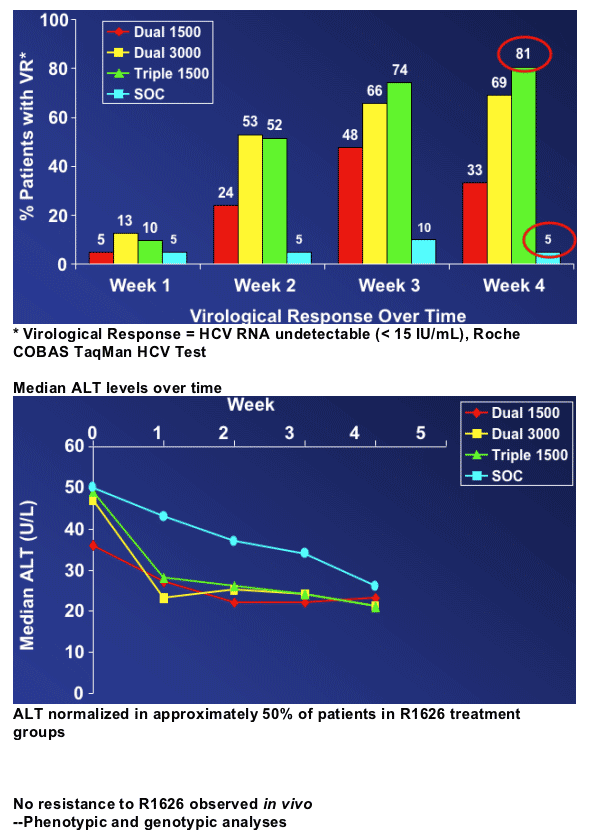
--81% patients with undetectable HCV RNA by week 4
(R1626 1500 mg bid + PEG-IFN_-2a + RBV)
Lack of viral resistance
--Observed viral load rebound was not due to viral resistance selection
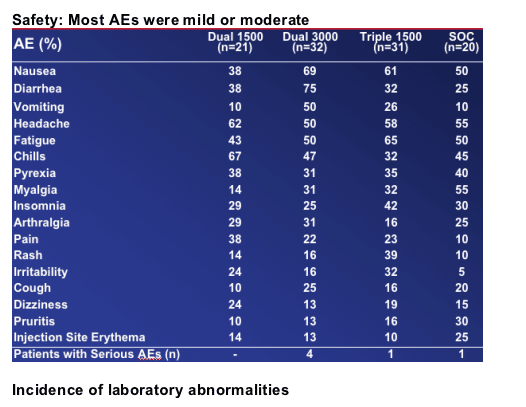
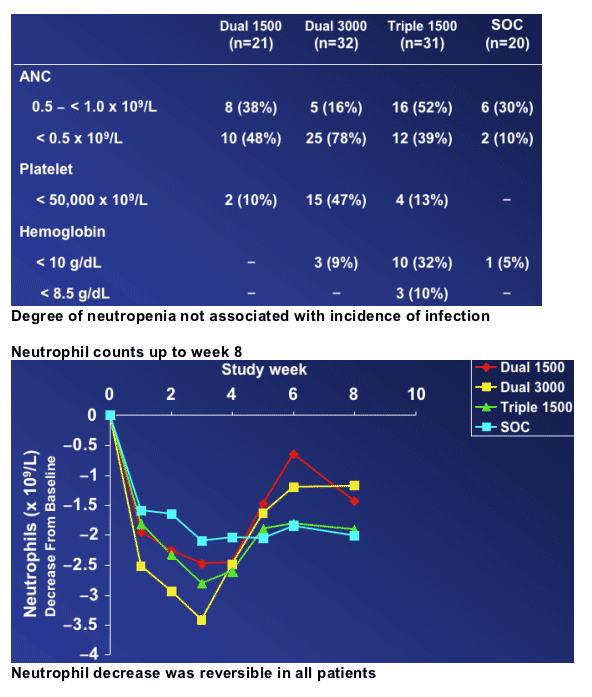
Dosing of R1626 was limited by neutropenia
--Degree of neutropenia not associated with incidence of infectionRBV
NEXT STEP
A global phase 2b study (490 patients) is ongoing to evaluate R1626 in triple combination for up to 48 weeks
R1626 at doses of 500, 1000 and 1500 mg bid
PEG-IFN_-2a at 90 µg and 180 µg
Standard dose of RBV
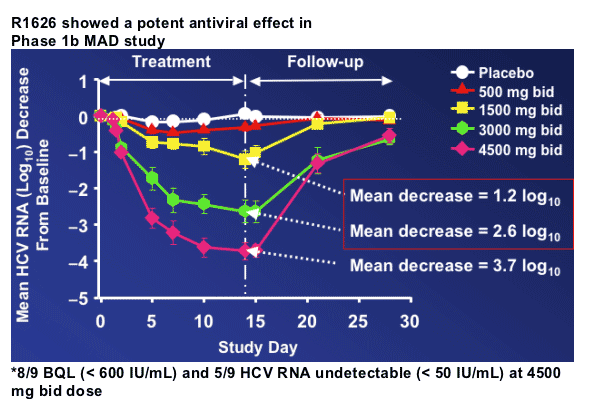
R1626: Characteristics
R1626 (F. Hoffmann-La Roche Ltd) is a pro-drug of the nucleoside analog, R1479-TP, a potent and selective inhibitor of the HCV polymerase enzyme
--Rapid conversion in intestinal mucosa to R1479
The mode of action of R1626 as a chain terminator may offer an advantage vs. non-nucleoside analog inhibitors (NNIs) of the polymerase and protease inhibitors (PIs)
--Similar potency against all genotypes and subtypes
--Potential high barrier to resistance selection
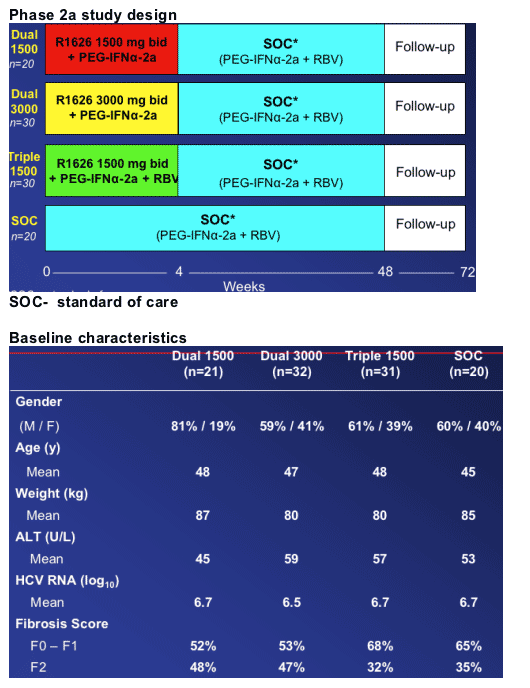
Phase 2a study objectives and design
Objective
--Evaluate antiviral activity and drug resistance, and define safety/tolerability (Maximum Tolerable Dose)
Design
--Multi-center, randomized, double-blind (R1626 and RBV), active-controlled
--4-week combination with peginterferon alfa-2a (40KD] (PEGASYS) ± RBV (COPEGUS) with follow-up safety data on SOC to week 48
--Patient population
--Treatment-naïve, genotype 1
--Non cirrhotic (Metavir ≦ 2, Knodell ≦ 1, Ishak ≦ 4)
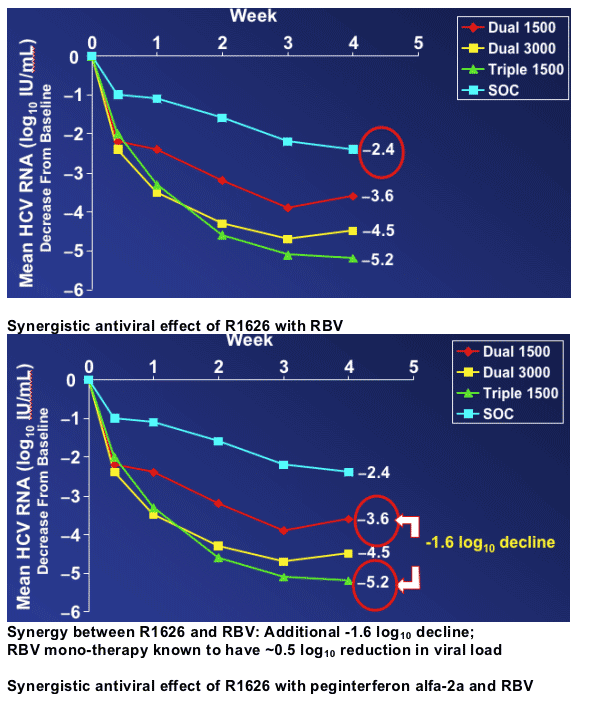
Reduction in HCV RNA over time
- SOC had expected -2.4 log10 decline by week 4
- Triple 1500 achieved -5.2 logs viral load reduction by week 4
- Dual 3000 achieved -4.5 log reduction at week 4
- Dual 1500 achieved -3.6 log viral load reduction by week 4
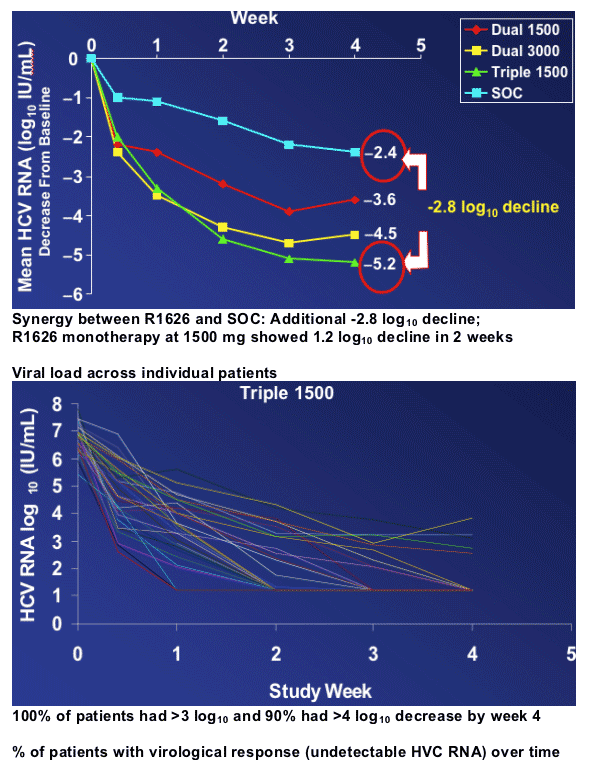
8 patients with viral rebound defined as sustained
≥ 0.5 log10 increase above nadir
--All 8 patients had stopped R1626 prior to viral rebound
--All samples in phenotypic assay were as sensitive as the baseline
--Sequence analysis of the NS5B revealed no known resistance mutations (S96T or S96T/N142T) or any other common amino acid substitutions
In addition, sequence analysis showed no known resistance mutations at baseline for all patients (n=150)
|
| |
|
 |
 |
|
|