 |
 |
 |
| |
Antiviral Activity, Pharmacokinetics, Safety, and Tolerability of R7128, a Novel Nucleoside HCV RNA Polymerase Inhibitor, Following Multiple, Ascending, Oral Doses in Patients with HCV Genotype 1 Infection Who have Failed Prior Interferon Therapy
|
| |
| |
Reported by Jules Levin,
AASLD, Nov 2-6, 2007, Boston, MA
R. Reddy1, M. Rodriguez-Torres2, E. Gane3, R. Robson4, J. Lalezari5, G.T. Everson6, E. DeJesus7, J.G. McHutchison8, H.E. Vargas9, A. Beard10, C.A. Rodriguez11, G.Z. Hill11, W.T. Symonds10, M.M. Berrey10
1University of Pennsylvania, Philadelphia, PA, USA; 2Fundacion de Investigacion d Diego, Santurce, PR, USA; 3Auckland Clinical Studies Limited, Auckland, New Zealand; 4CCST, Christchurch, New Zealand; 5Quest Clinical Research, San Francisco, CA, USA; 6University of Colorado, Aurora, CA, USA; 7Orlando Immunology Center, Orlando, FL, USA; 8Duke Clinical Research Institute, Durham, NC, USA; 9Mayo Clinic, Phoenix, AZ, USA; 10Pharmasset, Inc., Durham, NC, USA; 11Roche, Palo Alto, CA, USA
AUTHOR SUMMARY
A mean 2.7 log10 decline and maximum 4.2 log10 decline was demonstrated following 14 days of monotherapy
R7128 was generally well-tolerated and demonstrated no evidence of acute target organ toxicity
78% of subjects receiving R7128 with abnormal ALT at baseline normalized
R7128 was effective as a pro-drug delivery of PSI-6130
Terminal plasma half-life of the parent compound, PSI-6130, was ~5 hrs; terminal plasma half-life of the metabolite, PSI- 6206, was ~20 hrs
A maximum tolerated dose of R7128 has not been identified
AUTHOR CONCLUSIONS
R7128 has provided positive proof-of-concept that a direct acting antiviral can deliver sufficient antiviral potency via monotherapy to suppress below the level of detection (<15 IU/mL) in a prior IFN non-responder population
BID dosing of R7128 was superior to QD dosing in this monotherapy trial
Lack of clinical rebound provides early evidence of high genetic barrier for nucleoside inhibitors of NS5b polymerase
The safety and efficacy of this monotherapy study support further development of R7128 in combination with the standard of care (pegylated interferon and ribavirin)
ABSTRACT
Background: R7128 is a prodrug of PSI-6130, an oral cytidine nucleoside analog polymerase inhibitor, currently in development for the treatment of HCV. This multiple ascending dose study assessed safety, tolerability, pharmacokinetics, and preliminary antiviral activity of R7128 in subjects with HCV genotype 1 infection.
Methods: Multiple oral doses of R7128 monotherapy were administered for
14 days to 40 HCV-infected patients (n=10 per cohort with 8 active + 2 placebo) of 750mg QD, 1500mg QD, 750mg BID & 1500mg BID. PK, safety and virology assessments were conducted throughout the study.
Results: All subjects had HCV genotype 1 (30-1a; 10-1b), had previously failed alpha-interferon and were non-cirrhotic. There were no SAEs reported and no AEs required dose modification; no clinically significant changes in vital signs, ECGs, hematology, renal or other laboratory parameters occurred.
18 AEs were reported in 7 subjects receiving 750mg QD, 6 AEs in 3 subjects receiving 1500mg QD, 13 AEs in 4 subjects receiving 750mg BID and 14 AEs in 4 subjects receiving 1500mg BID. The most frequently reported AEs for patients receiving R7128 were headache (13), dry mouth (3), nausea (2), fatigue (2), tiredness (2), and upper respiratory infection (2); in subjects on placebo, headache (4) and diarrhea (4) were most commonly reported.
Plasma exposure to the prodrug, R7128, was negligible. PSI-6130 concentrations reached Cmax approximately 2-3 hours after dosing. Cmax and AUC exhibited less than proportional increases with increasing daily doses. Terminal half-life was approximately 5h for PSI-6130 and 20h for PSI-6206, the uridine metabolite.
HCV RNA in R7128 treatment groups declined in a dose dependent manner and reached nadir values at Day 15. The range at Day 15 in HCV RNA decrease from baseline in the 1500mg BID cohort was -1.2 to -4.2 (below the limit of detection).
Conclusion: R7128 monotherapy was generally well-tolerated and resulted in significant, dose-dependent suppression of HCV replication following 14 days of monotherapy. These results support continued development of R7128 for the treatment of HCV infection in combination with pegylated interferon and ribavirin.
METHODS
Study Design
Multiple oral doses of R7128 were administered as monotherapy to 40 HCV-infected patients
Subjects previously failed an interferon-containing regimen with or without ribavirin due to nonresponse or relapse.
10 subjects (8 active and 2 placebo) per cohort
Dosing regimens included: 750mg QD, 1500mg QD, 750mg BID, and 1500mg BID administered orally for 14 days.
Safety Assessments
Physical exam, vital signs, clinical laboratory tests, electrocardiograms (ECGs), and adverse events (AEs) assessments were performed throughout the study.
Pharmacokinetic (PK) Assessments
Full PK profiles for R7128 (pro-drug), PSI-6130 (parent molecule), and PSI-6206 (uridine metabolite) and the intermediate form were analyzed on Day 1 and Day 14.
Samples for plasma HCV RNA analysis using the Roche Cobas TaqMan (limit of detection <15 IU/mL) and resistance monitoring samples were collected throughout the study.
RESULTS
Demographics and Clinical Safety
Baseline demographics were similar across groups (Table 1)
No serious adverse events were reported. No subjects discontinued R7128 due to an AE.
There was no trend of drug or exposure-related AEs.
The placebo group reported the highest number of AEs with 34 events in 7 of 8 subjects.
The most commonly reported AEs are summarized in Table 2: a majority were mild in grade.
There were no clinically significant changes noted in the vital sign parameters across the treatment groups.
No trends in laboratory abnormalities were noted, and no treatment emergent grade 2 or higher labs were reported.
Of those receiving R7128 with abnormal ALT at baseline, 78% normalized by Day 14.
No clinically significant changes were reported for serial ECGs, including no QTc prolongation beyond 500ms.
Antiviral Activity
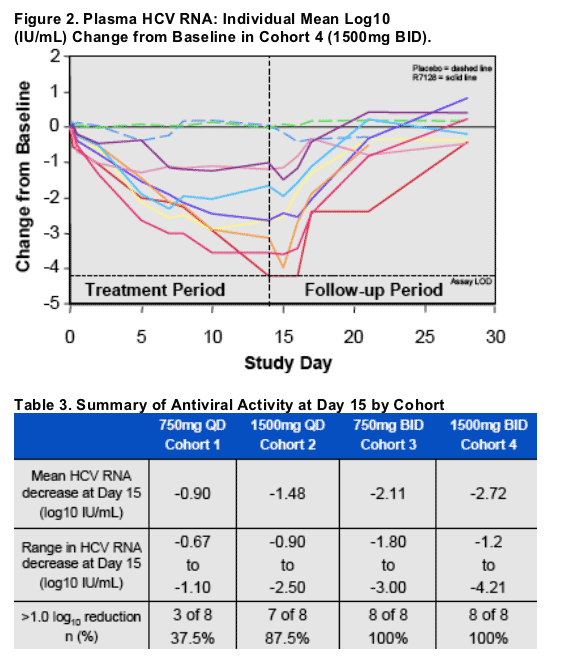
R7128 demonstrated dose-dependent HCV RNA decreases through 14 days of monotherapy in all dose groups (Table 3)
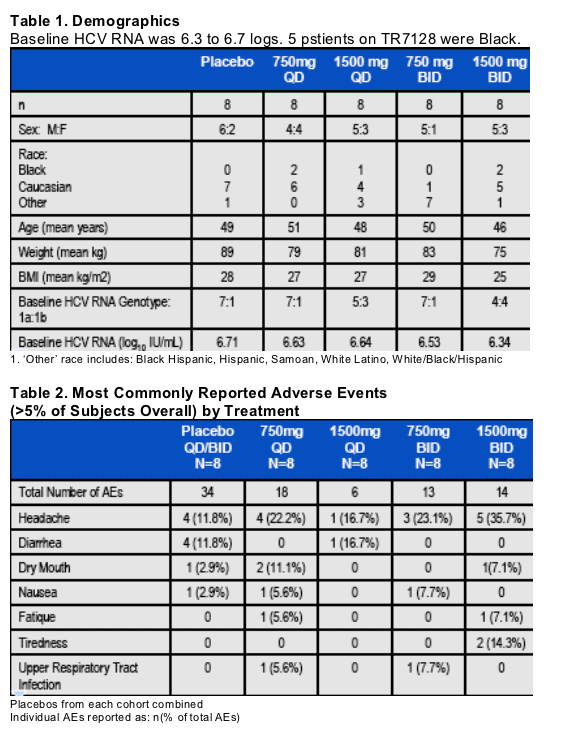
Viral nadir occurred at Day 15 with no evidence of clinically relevant rebound during therapy (Fig. 1).
HCV RNA values in the placebo group remained at baseline.
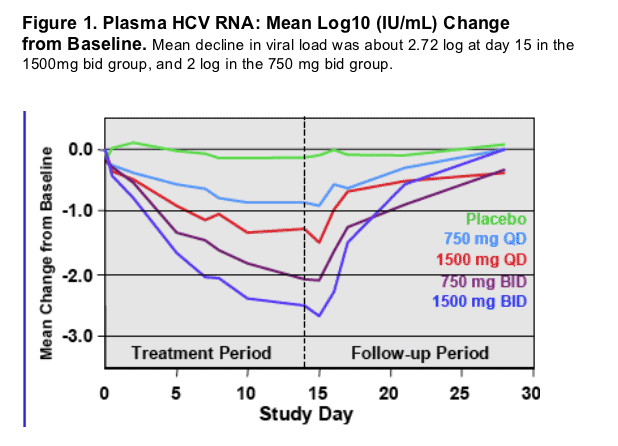
The range at Day 15 in HCV RNA decrease from baseline in the 1500mg BID cohort was -1.2 to -4.2 log10 IU/mL with this subject having HCV RNA below the limit of detection (Fig. 2).
Two subjects with ~1.2 log10 IU/mL decline included a prior null responder and a subject with an inadequate IFN response at 12 weeks.
Pharmacokinetics
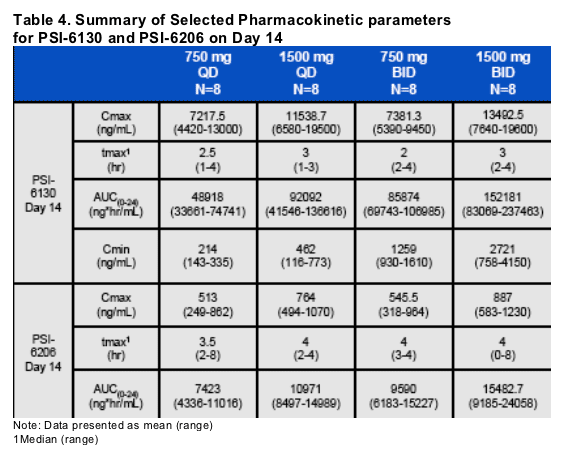
Oral dosing with R7128 resulted in dose-dependent but not dose proportional exposure to PSI-6130, the active moiety.
Plasma concentrations of the pro-drug, R7128, were below the limit of detection for all subjects
The monoester intermediate form was detected only at higher doses at tmax in a subset of subjects indicating efficient conversion of pro-drug to parent.
The terminal plasma elimination half-life (t1/2) of PSI-6130 was ~5 hrs, while the t1/2 of the active metabolite, PSI-6206, was ~20 hrs.
|
| |
|
 |
 |
|
|