 |
 |
 |
| |
Rapid and early virological response rates are increased with 12 week 360 μg/wk peginterferon alfa-2a (40KD) and standard ribavirin in HCV genotype 1 treatment naive patients: efficacy and safety analysis of the induction phase of the CHARIOT study
|
| |
| |
Reported by Jules Levin
AASLD, Nov 2-6, 2007, Boston, MA
S. Roberts1; M. D. Weltman2; D. Crawford3; W. Cheng4; W. Sievert5; G. W. McCaughan6; P. V. Desmond7; M. Yoshihara8; J. E. Miller8; J. Depamphilis9; P. Marks10; G. J. Dore10; C. On Behalf of the1
1. The Alfred Hospital, Melbourne, VIC, Australia.
2. Nepean Hospital, Sydney, NSW, Australia.
3. Greenslopes Hospital, Brisbane, QLD, Australia.
4. Royal Perth Hospital, Perth, WA, Australia.
5. Monash Medical Centre, Melbourne, VIC, Australia.
6. Royal Prince Alfred Hospital, Sydney, NSW, Australia.
7. St. Vincent's Hospital, Melbourne, VIC, Australia.
8. Roche Products, Sydney, NSW, Australia.
9. Roche, Nutley, NJ, USA.
10. National Centre in HIV Epidemiology and Clinical Research, Sydney, NSW, Australia.
ABSTRACT
Background: The CHARIOT study evaluates the efficacy & safety of a 360 μg/wk dose peginterferon alfa-2a (Peg-IFNα-2a) induction regimen compared to standard 180 μg/wk Peg-IFNα-2a, in combination with standard ribavirin (RBV) in treatment-naive patients with chronic hepatitis C genotype 1. We report results of the planned interim analysis of on-treatment virological responses over the first 12 wks of therapy.
Methods: In this international, multi-centre, open label study patients were stratified by baseline HCV RNA and randomised 1:1 to 180 μg or 360 μg Peg-IFNα-2a qw for 12 wks followed by 36 wks of 180 μg Peg-IFNα-2a plus RBV 1000–1200 mg/d for 48 wks. Endpoints include virologic response at wks 4, 8 and 12.
Results: 845 patients were evaluable for efficacy & safety analysis. Age, gender, weight, BMI, ethnicity, viral load and fibrosis scores were similar in the two groups. At wks 4, 8 and 12, virological response rates were greater (p<0.001) in the 360 μg/wk arm (Table). Significantly higher response rates (p<0.001) at wk 12 occurred in patients receiving 360 μg dose in all subgroups including baseline HCV RNA ≥800,000 IU/mL (71% vs 55%) and <800,000 IU/mL (84% vs 71%), weight ≥85 kg (74% vs 56%) and <85kg (74% vs 62%), and age >40yrs (70% vs 54%) and ≦40yrs (84% vs 73%). Induction therapy was associated with higher rates of diarrhoea (17% vs 12%), pyrexia (14% vs 8%), chills (14% vs 7%) and weight loss (10% vs 3%). Depression and fatigue reports were similar. Discontinuations were similar in both groups, however induction therapy led to more dose modifications (Table). Use of haematopoetic growth factors was the same in both groups (4 patients [<1%]).
TABLE: Week 4 RVR was 26% for Peg180ug/wk & 35% for peg360ug/wk. Week 8 response was 49% for low dose Peg vs 61% for higher dose Peg. A week 12 EVR, early viral response, was achieved by 79% in peg 180ug and by 89% using high dose Pegasys 360ug/wk. therapy discontinuations were 4% & 5%, tx d/c due to AEs or lab abnormalities were 2 & 3%, SAEs were 3 & 4%; but dose modifications were 14% for Pegasys 180ug/wk & 22% for Pegasys 360ug/wk; neutropenia was 2% for Peg180ug & 5% for Peg360ug/wk, thrombocytopenia was <1% for Peg180 & 4% for Peg360, and anemia was 7% for Peg180 & 10% for Peg360.
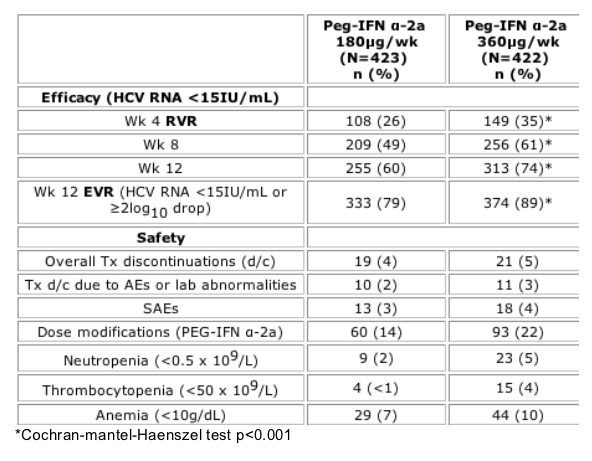
Conclusion: Induction dosing with 360 μg/wk Peg-IFNα-2a is more effective than standard 180 μg/wk dosing in achieving early and rapid virological responses and is safe and well tolerated. The effect was independent of baseline viral load, weight, BMI and age.
|
| |
|
 |
 |
|
|