 |
 |
 |
| |
Pegylated interferon alfa-2a (40KD) plus ribavirin (RBV) in prior non-responders to pegylated interferon alfa-2b (12KD)/RBV: final efficacy and safety outcomes of the REPEAT study
|
| |
| |
Reported by Jules Levin
AASLD, Nov 2-6, 2007, Boston, MA
Donald M. Jensen*, Bradley Freilich, Pietro Andreone
Adrian DiBisceglie, Carlos E. Brandao-Mello, K. Rajender Reddy, Antonio Craxi, Antonio Olveira Martin, Gerlinde Teuber,
Diethelm Messinger, Greg Hooper, Matei Popescu and
Patrick Marcellin
*Center for Liver Diseases, University of Chicago Hospitals, Chicago, IL, USA
AUTHOR SUMMARY
SAFETY SUMMARY
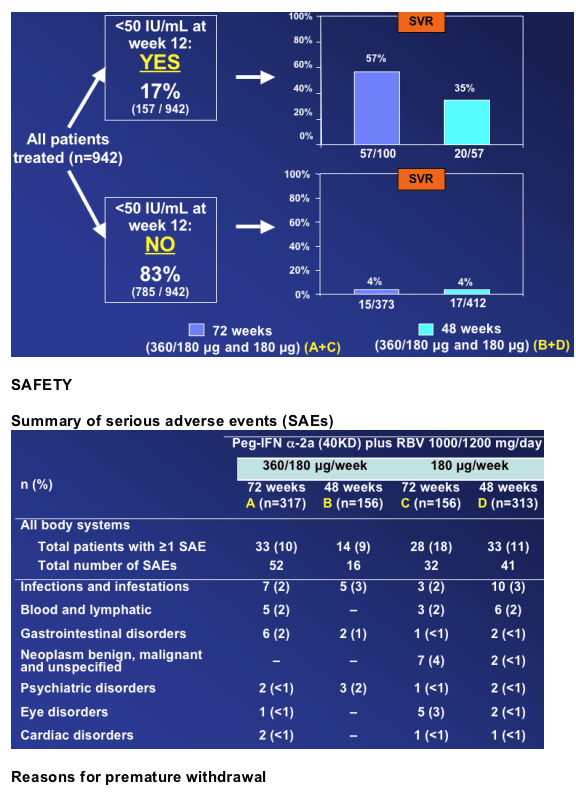
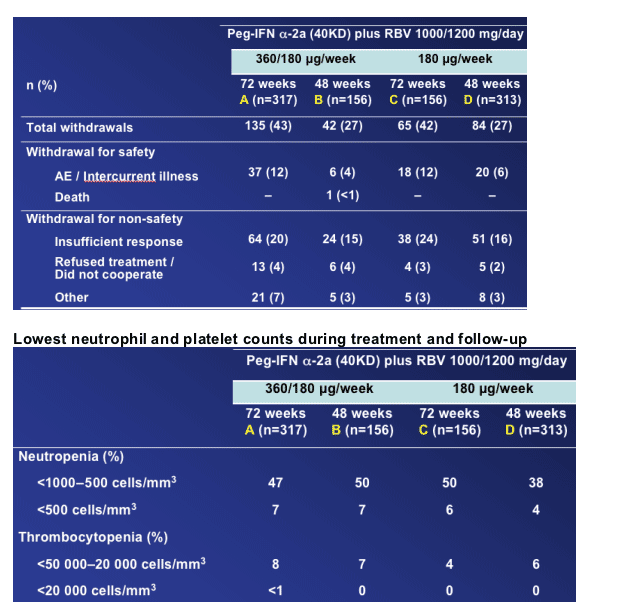
Incidence and types of serious adverse events were generally consistent across all four arms
Premature withdrawals for adverse events or laboratory abnormalities were higher for the longer treatment durations
EFFICACY SUMMARY
In the difficult-to-cure population of prior non-responders to Peg-IFN alfa-2b (12KD) plus RBV:
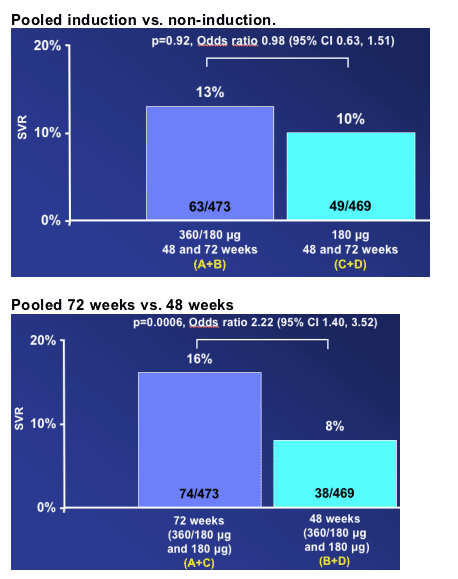
-- 72 weeks of retreatment with Peg-IFN alfa-2a (40KD) plus RBV provided the highest SVR rates
-- A 12-week induction with Peg-IFN alfa-2a (40KD) 360 μg/week did not appear to add value to the longer treatment duration
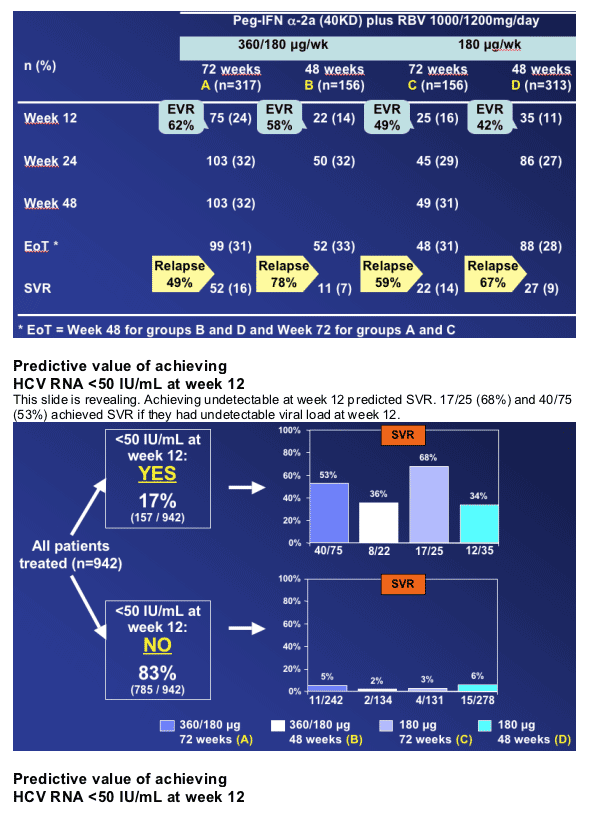
-- If HCV RNA is <50 IU/mL at week 12 the probability of SVR is high
BACKGROUND
Combination therapy with pegylated interferon (IFN) plus ribavirin (RBV) has led to a substantial increase in SVR rates when compared to conventional IFN/RBV combination therapy1
Despite this, up to half of all patients treated with pegylated interferon alfa-2b (12KD) plus RBV fail to achieve an SVR2,3
Initial retreatment studies have suggested a benefit of induction doses and a prolonged duration of treatment in previous non-responders to IFN/RBV4,5
1Strader DB, et al. Hepatology 2004;39:1147-71
2Manns MP, et al. Lancet 2001;358:958-65
3Jacobson IM, et al. Hepatology 2007;46:971-81
4Diago M, et al. Aliment Pharmacol Ther 2007;26:1131-38
5Tong, M, et al. J Viral Hepat 1998;5:323-31
AIM
The aim of this international, multicenter, randomized clinical trial was to examine the effect of both induction dosing of peginterferon alfa-2a (40KD) plus longer treatment duration, in patients who had failed to respond to a previous course of pegylated interferon alfa-2b (12KD) plus RBV.
Definition of non-responders
Pegylated interferon alfa-2b (12KD, Peg-IFN alfa-2b) plus RBV combination therapy given for ≥12 weeks
At least 1.0 μg/kg/week Peg-IFN alfa-2b (12KD) and at least 800 mg/day RBV as starting dose
At least one HCV RNA test performed on treatment after a minimum of 12 weeks of therapy
Positive HCV RNA test results at every assessment after initiation of Peg-IFN alfa-2b (12KD)
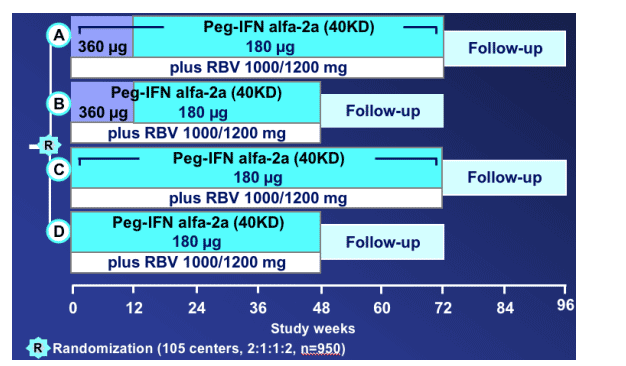
REPEAT study design
There are 4 study arms.
Arm A: 360ug/wk Pegays for 12 weeks, total of 72 weeks therapy.
All 4 groups receive RBV 1000/1200mg.
Arm B: 12 weeks high dose, total of 48 weeks therapy.
Arm C: 72 weeks standard therapy dosing.
Arm D: 48 weeks standard dosing.
Study endpoints
Primary endpoint:
--Compare the efficacy of 72 weeks of Peg-IFN alfa-2a (40KD) plus RBV with high-dose induction versus the standard doses of the same combination given for 48 weeks
Secondary endpoints:
--Compare the efficacy of high-dose induction with standard dose
--Compare the efficacy of 72 weeks of treatment with 48 weeks of treatment
Evaluate the safety of Peg-IFN alfa-2a (40KD) plus RBV combination therapy given for 48 and 72 weeks, including high-dose induction regimens
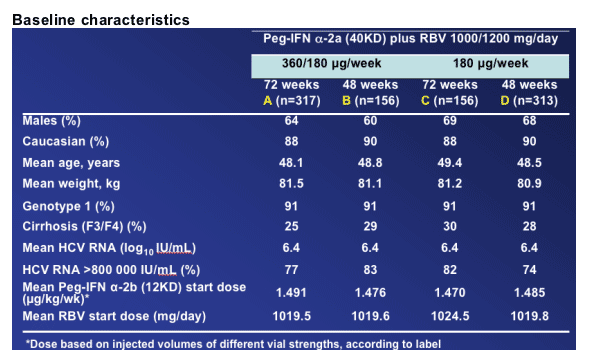
RESULTS
All results are for
ITT population (n=942
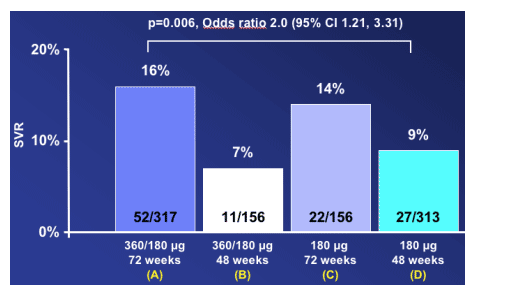
Primary comparison:
72 week induction vs. 48 week non-induction
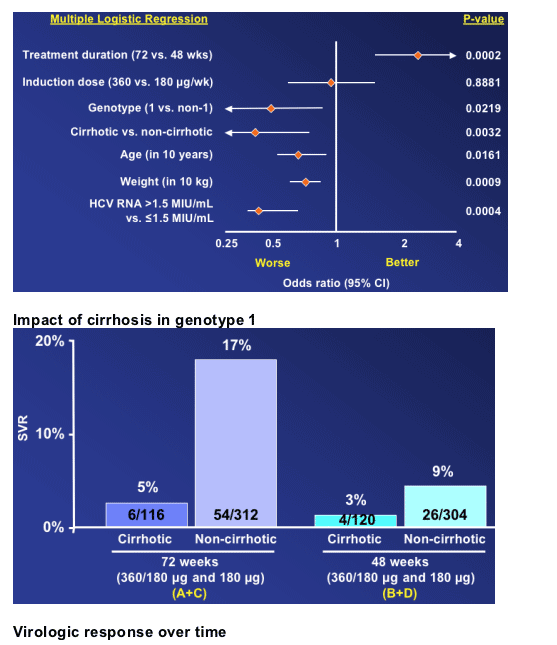
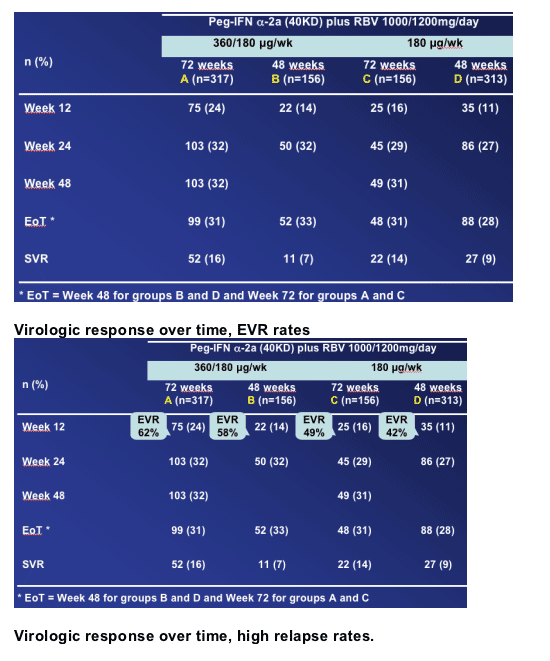
Arm A, the induction dose and 72 weeks therapy, achieved 16% SVR. Arm C, the 72 week and standard 180ug dose group, achieved 14% SVR. Arm B, the induction dose group with only 48 weeks therapy, had a 7% SVR rate. And arm D, standard 180ug dose and 48 weeks, achieved 9% SVR rate.
|
| |
|
 |
 |
|
|