 |
 |
 |
| |
High rates of sustained virologic response (SVR) in patients >50 years infected with HCV genotype 1 with positive prognostic factors treated with peginterferon alfa-2a (40KD) (PEGASYS®) and ribavirin (COPEGUS®)
|
| |
| |
Reported by Jules Levin
AASLD, Nov 2-6, 2007, Boston, MA
K.R. Reddy,1 D. Messinger,2 M. Popescu,3 S.J. Hadziyannis4
1Division of Gastroenterology, University of Pennsylvania, Philadelphia, PA, USA; 2IST GmbH, Mannheim, Germany; 3Roche, Basel, Switzerland; 4Henry Dunant Hospital, Athens, Greece.
This research was funded by Roche, Basel, Switzerland.
Conclusions
Subgroups of HCV genotype 1 patients >50 years can achieve high SVR rates (particularly those with a low baseline viral load or without cirrhosis).
Older patients (>50 years) had more ribavirin dose reductions due to laboratory abnormalities
Maintaining exposure to peginterferon alfa-2a (40KD) and/or ribavirin during treatment is essential to achieving high SVR rates, particularly in older patients.
More attention is required when modifying dose of ribavirin in older patients; small decrements are advisable in order to maintain the highest tolerable ribavirin exposure.
The assessment of virologic response at weeks 4 and 12 can be used to predict which patients are most likely to achieve an SVR, regardless of age as high SVR rates were achieved by all patients with an RVR or cEVR.
In summary, older HCV patients are an important population and a full treatment course with pegylated interferon plus ribavirin should be considered based on their on-treatment week 4 and week 12 response.
Introduction
The prognosis of patients with chronic hepatitis C virus (HCV) is influenced by viral factors such as HCV genotype and viral load as well as by patient factors that include age, gender, race, advanced fibrosis/ cirrhosis and drug adherence.[1_4] Disease management is complicated by the fact that the HCV patient pool is ageing.[5] Further, older patients tend to have an accumulation of negative prognosis factors such as advanced fibrosis/cirrhosis and thus do not respond to treatment with pegylated interferon therapy as well as younger patients.[1_4,6] Tolerance to therapy is also lower in older patients, resulting in reduced drug exposure and potentially impaired rates of sustained virologic response (SVR, defined as undetectable HCV RNA 24 weeks post-treatment).[3.4] Whilst early treatment prevents the accumulation of negative prognostic factors, the growing number of older patients still require treatment.
Using data from HCV genotype 1 patients enrolled in two large phase III studies of peginterferon alfa-2a (40KD) (PEGASYS®) plus ribavirin (COPEGUS®),[1,2] we analyzed the impact of baseline and on-treatment factors on SVR in older versus younger patients.
Methods
Patients eligible for this study were treatment-naïve with quantifiable HCV RNA (COBAS AMPLICORTM HCV MONITOR TEST), elevated ALT and compensated liver disease at baseline and a liver biopsy consistent with chronic HCV within 6_15 months of study start.
Patients included in the analysis were randomized to receive peginterferon alfa-2a (40KD) (PEGASYS®) 180 _g/wk plus ribavirin (COPEGUS®) 1000/1200 mg/day for 48 weeks.
The primary efficacy end-point (SVR) and on-treatment responses (RVR,
cEVR and pEVR) were defined as:
_ SVR: undetectable (<50 IU/mL) HCV RNA 24 weeks post-treatment
_ Rapid virologic response (RVR): undetectable HCV RNA at week 4
_ Complete early virologic response (cEVR): no RVR, but undetectable HCV RNA at week 12
_ Partial early virologic response (pEVR): no RVR and no cEVR, but at least a 2-log10 drop in HCV RNA from baseline to week 12.
Cumulative drug exposure, expressed as the percentage of target dose for both peginterferon alfa-2a and ribavirin accounts for dosage reductions due to patient non-adherence, or physician-initiated modifications due to adverse events or laboratory abnormalities.
Chi-square test was used to analyze the association between age (≦50, >50 years) and various baseline factors as well as viral response variables.
Univariate and stepwise multivariate logistical regression (MLR) analyses were used to identify baseline and on-treatment factors (virologic response and cumulative dose of study drugs) predictive of SVR for patients of >50 years of age.
Results
Baseline characteristics of the 569 patients from two phase III studies[1,2] included in this analysis are shown in Table 1.
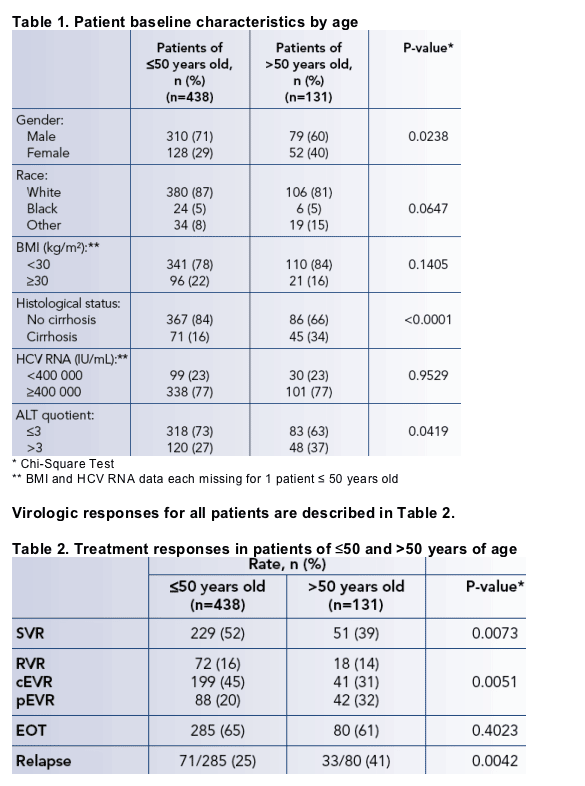
SVR: sustained virologic response; RVR: rapid virologic response; cEVR; complete early virologic response; pEVR: partial early virologic response; EOT: end of treatment response; Relapse: an EOT response but no SVR.
* Chi-square test
Overall SVR rates were significantly lower in patients >50 years (39%) compared to patients ≦50 years (53%), which seems to be mainly a consequence of a higher relapse rate (Table 2).
SVR rates in patients >50 years who achieved either an RVR (83%) or cEVR (61%) were similar to those seen in younger patients (Figure 1).
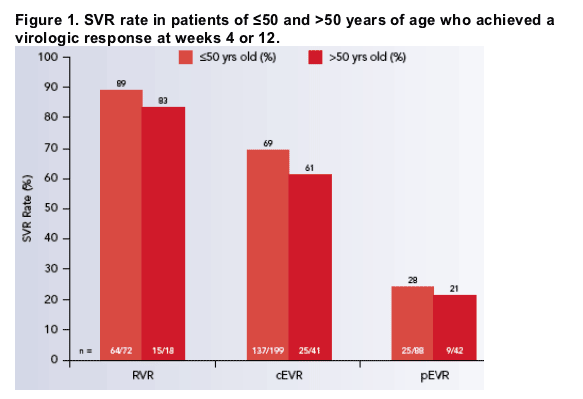
Patients >50 years with low baseline viral loads (<400 000 IU/mL) or without cirrhosis achieved higher SVR rates (57% and 44%, respectively) than those with high viremia or cirrhosis (34% and 29%, respectively).
Mean cumulative peginterferon alfa-2a exposure tended to be lower in patients of older age (6868 _g in patients >50 years versus 7230 _g in patients ≦50 years [p=0.0986]), whilst mean cumulative ribavirin exposure was significantly lower in older patients (252g versus 304g for younger patients [p<0.0001]).
The effect of dose modifications on SVR rates of all patients is shown
in Figure 2.
_ A higher proportion of older patients had <60% target ribavirin exposure than patients <50 years (38% versus 22%, respectively) and the rate of SVR in these patients is dramatically reduced compared to the rate of SVR among patients who achieved higher ribavirin exposure. Nonetheless, older patients achieve slightly higher SVR rates than younger patients at the lowest ribavirin exposure level. Importantly, we observed that 60-<80% target ribavirin exposure also
correlated with lower SVR rates in older patients.
_ Peginterferon alfa-2a dose reductions to <80% target exposure is also associated with lower SVR rates, particularly in older patients: SVR rates in older patients decreased to 17_18% when peginterferon alfa-2a exposure was <80%, compared to 52% when exposure levels were ≥80%. However, as with ribavirin, older patients achieved slightly better responses than younger patients at the lowest peginterferon alfa-2a exposure level.
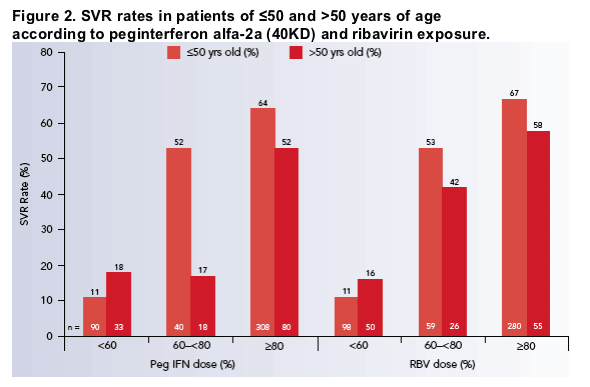
Univariate and MLR analysis were used to predict SVR rates in patients
>50 years
_ Low baseline viral load (p=0.007), high ALT ratio (p=0.011) and cirrhosis (p=0.048) were significantly associated with increased SVR rates (MLR with baseline factors).
_ Achievement of RVR or cEVR was significantly predictive of SVR, as was cumulative exposure to both peginterferon alfa-2a and ribavirin (Table 3).
A similar percentage of patients of ≦50 and >50 years completed therapy (76% and 73%, respectively), and patient withdrawal from treatment due to any adverse event was also independent of age (11% and 12%, respectively).
Conversely, more patients >50 years had ribavirin dose reductions due to laboratory abnormalities (mainly anemia) than those of younger age (43% vs. 17%).
References
1. Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med 2002; 347: 975-982.
2. Hadziyannis SJ, Sette H, Jr., Morgan TR, et al. Peginterferon alpha-2a and ribavirin combination therapy in chronic hepatitis C: a randomized study of treatment duration and ribavirin dose. Ann Intern Med 2004; 140: 346-355.
3. Reddy KR, Shiffman ML, Morgan TR, et al. Impact of ribavirin dose reductions in hepatitis C genotype 1 patients completing peginterferon alfa-2a/ribavirin treatment. Clin Gasteroenterol Hepatol. 2007; 5: 124_129.
4. Foster GR, Fried MW, Hadziyannis SJ, et al. Prediction of sustained virological response in chronic hepatitis C patients treated with peginterferon alfa-2a (40KD) and ribavirin. Scand J Gasteroenterol. 2007; 42: 247_255.
5. McHutchison JG and Bacon BR. Chronic hepatitis C: an age wave of disease burden. Am J Manag. Care 2005; 11: S286_295.
6. Manns MP, McHutchison JG, Gordon SC. Peginterferon alfa-2b plus ribavirin compared with interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: a randomised trial. Lancet 2001; 358: 958-965.
|
| |
|
 |
 |
|
|