 |
 |
 |
| |
In vitro activity and preclinical pharmacokinetics
of the HCV protease inhibitor TMC435350
|
| |
| |
Reported by Jules Levin
AASLD, Nov 2-6, 2007, Boston, MA
Kenneth Simmen1, Oliver Lenz1, Tse-I Lin1, Greg Fanning1, Pierre Raboisson1, Herman de Kock1, Gerben van 't Klooster1, Asa Rosenquist2, Michael Edlund2, Magnus Nilsson2, Lotta Vrang2, Bertil Samuelsson2.
1Tibotec Pharmaceuticals Ltd, Eastgate Village, Little Island, Cork, Ireland; 2Medivir AB, Box 1086, SE-141 22 Huddinge, Sweden
AUTHOR CONCLUSIONS
TMC435350 is a novel small molecule HCV NS3/4A protease inhibitor with:
· potent anti-HCV activity in enzymatic and cellular assays.
· good selectivity to other viruses and human proteases.
· synergy in combination with IFN-_ or an NS5B polymerase thumb domain inhibitor, and additivity with ribavirin.
· drug-like properties.
· favorable liver exposure as shown in pharmacokinetic and preclinical safety studies.
TMC435350 is currently being evaluated in phase I clinical trials.
Abstract
Background: As a class, HCV NS3/4A protease inhibitors have shown promise in clinical trials for the treatment of chronic hepatitis C virus infection. TMC435350 is a novel and potent macrocyclic NS3/4A protease inhibitor. To further assess the potential of TMC435350, we characterized the in vitro activity of TMC435350 alone or in combination with different classes of HCV inhibitors. The preclinical plasma pharmacokinetics and tissue distribution were also studied in vivo.
Methods: The effect on HCV RNA level and the emergence of drug-resistant colonies was analyzed in the replicon model with TMC435350 alone, or in combination with interferon alpha, ribavirin, or an HCV polymerase thumb domain inhibitor. Pharmacokinetic profiles were evaluated following single or repeated dosing in rats. The tissue distribution of TMC435350 was studied in male rats at time points from 0.5 up to 31 hours after a single oral dose of 40 mg/kg.
Results: In biochemical HCV NS3/4A protease assays, TMC435350 exhibited Ki
values of 0.5 and 0.4 nM for subtypes 1a (H77) and 1b (con1) enzymes respectively.
TMC435350 was found in the subgenomic genotype1b replicon model to have an EC50 of 8 nM and a selectivity index (SI) of > 2000.
The combination of TMC435350 with different classes of HCV inhibitors
further increased its activity in reducing HCV RNA in an additive to synergistic
manner, and further reduced the emergence of resistant replicon colonies.
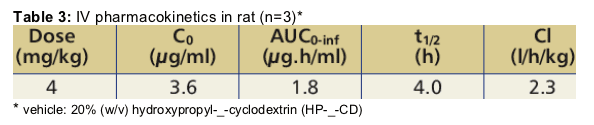
After single oral administration of a PEG400-based solution of TMC435350 at
40 mg/kg in rats, the mean peak plasma concentration (Cmax) was 1430 ng/ml and was observed at two hours post-dose (tmax).
The absolute bioavailability of TMC435350 was calculated at 44% after single oral administration of a 40 mg/kg dose.
TMC435350 was found to be extensively distributed to the liver, small- and large
intestines (tissue/plasma ratios >35).
Concentrations in other organs were similar to plasma.
Notably, TMC435350 was still quantifiable in the liver tissue up to 31 hours post-dosing.
Conclusions: TMC435350 is a novel potent and specific HCV protease inhibitor, with good oral bioavailabilty and a favorable liver distribution. In addition, in vitro studies support the potential use of TMC435350 in combination with other HCV inhibitors.
INTRODUCTION
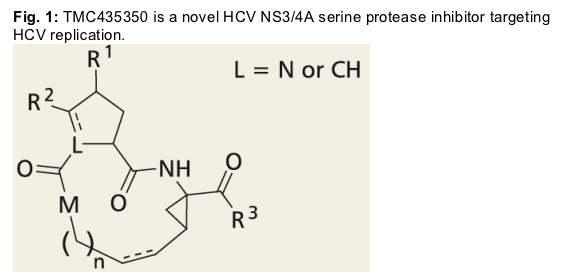
TMC435350 was identified as a potent inhibitor from a series of macrocyclic analogs in enzymatic assays using HCV genotype 1 NS3 proteases and cellular replicon models. Selectivity and cytoxicity were further characterized, and studies with IFN-_, ribavirin and an NS5B polymerase thumb domain inhibitor1 in the replicon demonstrated the potential of these combinations.
RESULTS
HCV enzymatic activity
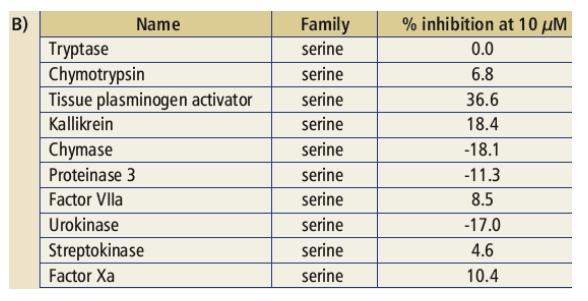
Table 1: Inhibitory activity of TMC435350 in genotype 1a and 1b HCV NS3/4A protease assays (A), and against a panel of human proteases (% inhibition at 10 _M (B) or as IC50 (C)). All data was generated using standard FRET based assays.

* Protease catalytic domains aa 1-181 cloned with a C-terminal His6 tag, was
expressed in E. coli and purified. NS4A peptide was added for the biochemical assay.
· Potent inhibitor of both genotype 1a and 1b HCV NS3/4A proteases.
· Inhibition of HCV NS3/4A protease was > 1,000 times stronger than inhibition of tested host proteases.
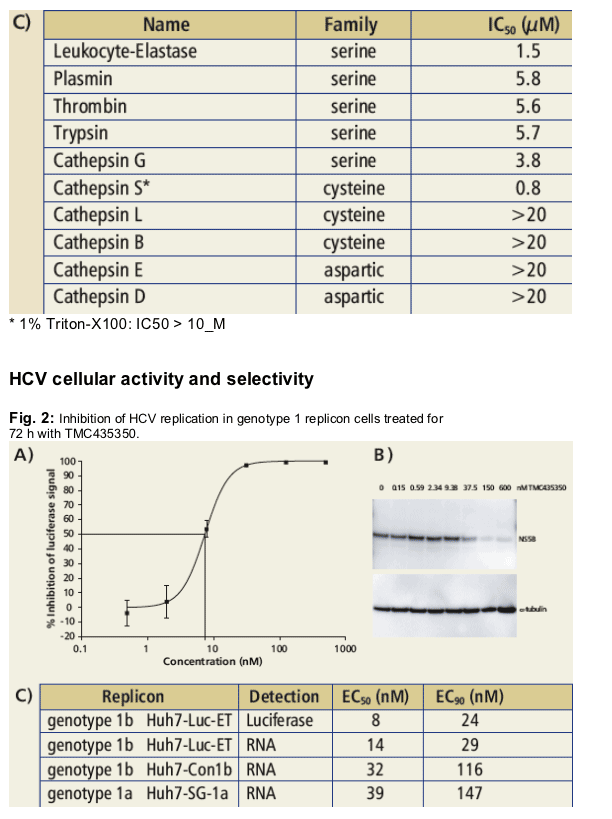
Dose-dependent inhibition of HCV replication was monitored with genotype 1b Huh7-Luc-ET (kindly provided by R. Bartenschlager as HCV con1b bicistronic replicon clone ET), Huh7-con1b and Huh7-SG1a replicon cells (kindly provided by Apath L.L.C. as con1b and H77 replicons) using luciferase reporter read-out, immunoblotting and quantitative RNA detection by real-time PCR.
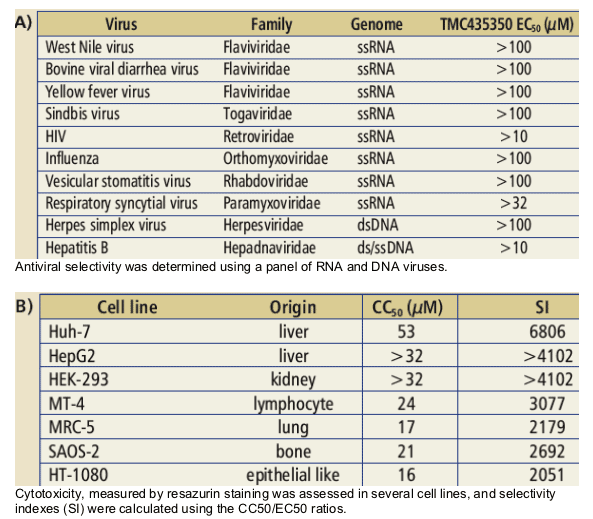
Table 2: Antiviral selectivity (A) and cytotoxicity (B) testing
· Potent and highly specific HCV inhibitor (EC50 = 8 nM).
· Demonstrated minimal cytotoxicity in tested human cell lines (SI>2,000).
Combination studies with IFN-_
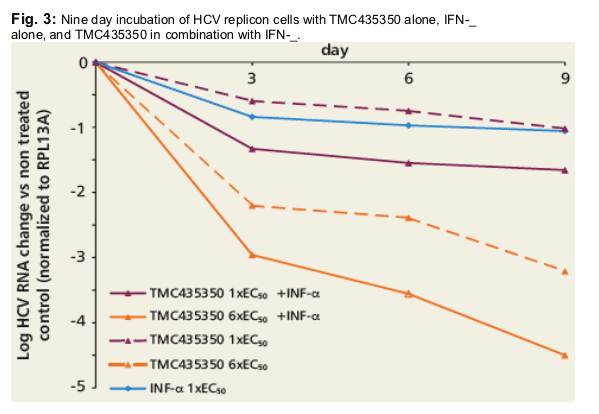
Huh7-Luc-ET cells were treated for 9 days with IFN-_, TMC435350, and TMC435350 in combination with IFN-_ (Biosource recombinant human IFN-_). Medium with compounds was refreshed every 3 days and cells were harvested on days 3, 6 and 9. HCV RNA and cellular RPL13A levels were measured by quantitative RNA detection.2
In the replicon model, combination of TMC435350 with IFN-_ resulted in:
· > 4 log10 reduction of HCV RNA in 9 days.
· increased suppression of drug resistant replicon colonies.
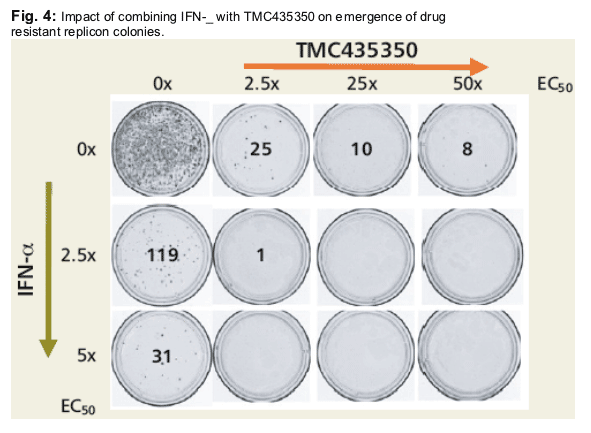
Huh7-Luc-ET cells were treated for 4-6 weeks with G418 and TMC435350 +/- IFN-_. Medium with compounds was refreshed every 3-4 days. Resistant Colonies were stained with neutral red and counted.3
In vitro synergy studies
· In replicon cells, TMC435350 showed additivity when combined with ribavirin, and synergy in combination with IFN-_ or an NS5B polymerase thumb domain inhibitor (thiophene class)1.
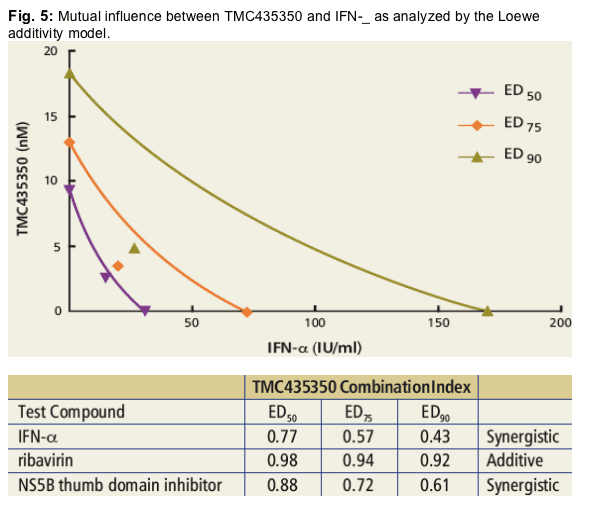
Huh7-Luc-ET cells were treated for 3 days with TMC435350 in combination with IFN-_, ribavirin, or an NS5B thumb domain inhibitor. The relationship between inhibitory activities of TMC435350 and test compound was explored using CalcuSyn (Biosoft, Ferguson, MO). CI values of <1, =1, and >1 indicate synergy, an additive effect, or antagonism, respectively.

· Good systemic oral bioavailability.
· Plasma exposure: C8h-plasma > EC99; liver exposure C31h-liver > EC99.
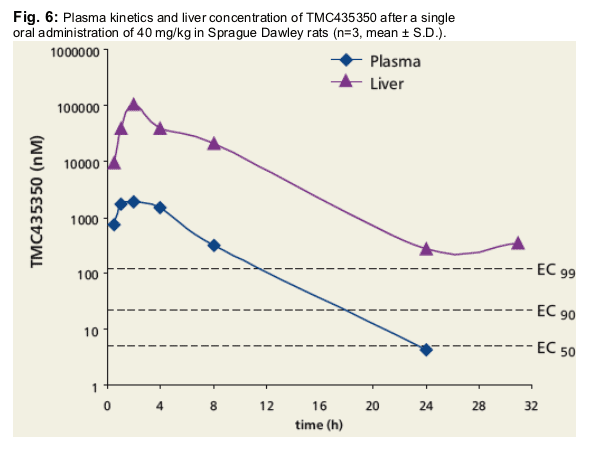
· Extensive distribution to liver and GI tract.
· Concentrations in other organs similar to plasma.
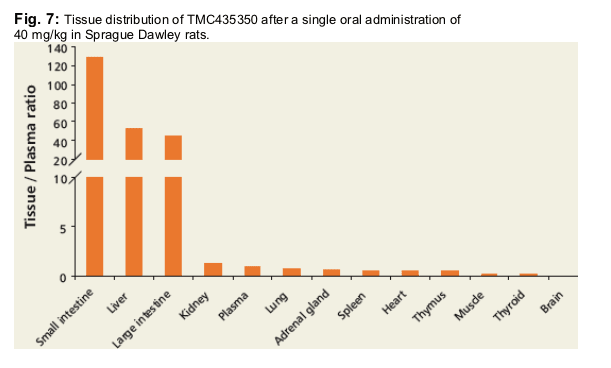
AUC0-31h (except muscle AUC0-8h) tissue to plasma ratios of TMC435350 in tissues of male Sprague-Dawley rats collected after single oral administration of a VitE-TPGS/PEG-400 solution of TMC435350 at 40 mg/kg. N = 3, analysis on pooled tissue.
Preclinical safety
· Negative in a battery of genotoxicity assays.
· Favorable profile in in vitro and in vivo safety pharmacology assays (CNS, cardiovascular, pulmonary functions).
References
(1) Chan, L. et al., 2004. Med. Chem. Lett. 14: 797
(2) Lin, K. et al., 2004. AAC 48: 4784
(3) Mo, H. et al., 2005. AAC 49: 4305
|
| |
|
 |
 |
|
|