 |
 |
 |
| |
Response-guided therapy in a prospective trial of peginterferon alfa-2a (40KD)/ribavirin treatment in patients with HCV genotype 1 or 4
|
| |
| |
Reported by Jules Levin
AASLD, Nov 2-6, 2007, Boston, MA
P. Ferenci,1 H. Laferl,2 T.-M. Scherzer,1 H. Brunner,3 A. Maieron,4 M.
Gschwantler,5 R.E. Stauber,6 R. Hubmann,7 K. Staufer,1 C. Datz,8 M. Bischof,9 H. Hofer,1 K. Löschenberger,10 P.E. Steindl-Munda,1
On behalf of the Austrian Hepatitis Study Group
1Department of Internal Medicine III, Medical University of Vienna, Vienna, Austria; 2Kaiser Franz Josef Hospital, Vienna, Austria; 3Krankenhaus Hietzing, Vienna, Austria; 4Krankenhaus der Elisabethinen, Linz, Austria;
5Wilhelminenhospital, Vienna, Austria; 6Medical University Graz, Graz, Austria; 7General Hospital, Linz, Austria; 8Krankenhaus Oberndorf, Salzburg, Austria; 9Krankenhaus Rudolfstiftung, Vienna, Austria; 10Roche Austria, Vienna, Austria
This research was funded by Roche, Vienna, Austria.
CONCLUSIONS
These data confirm that a substantial proportion of patients with HCV genotype 1 or 4 infection benefit from an abbreviated 24-week regimen of peginterferon alfa-2a (40KD) plus ribavirin.
RVR predicted SVR in patients infected with HCV genotype 4 and more patients infected with HCV genotype 4 than genotype 1 achieved an RVR at week 4 of treatment with peginterferon alfa-2a (40KD) plus ribavirin.
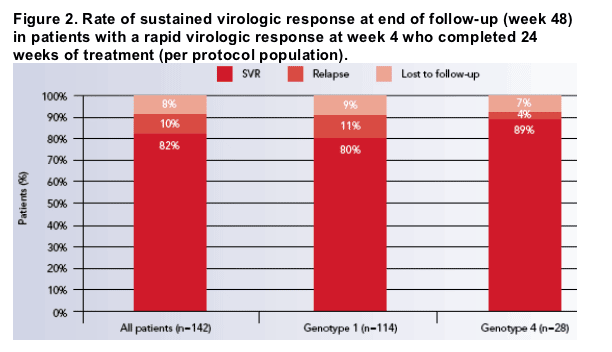
SVR rates among the 142 patients who completed 24 weeks of combination therapy were high in patients infected with HCV genotype 1 (80%) or 4 (89%) who achieved an RVR and were similar to those reported for genotype 1 patients with RVR treated for 24 or 48 weeks with the same combination (89%).[2]
Relapse during follow-up was uncommon in patients infected with HCV genotype 4, which suggests that abbreviated regimens are particularly well suited for this subgroup.
INTRODUCTION
The combination of pegylated interferon plus ribavirin is the treatment of choice for patients with chronic hepatitis C.[1]
Optimization of interferon-based treatment regimens remains an important goal as pegylated interferons are likely to remain the backbone of treatment in the
foreseeable future.
The rate and extent of virologic response to combination therapy in patients infected with hepatitis C virus (HCV) genotype 1 or 4 is highly variable. Indeed, it has recently been recognized that some patients with genotype 1 infection are "easier to cure" than others, and that the key to guiding treatment in these individuals is the presence of a rapid virologic response (RVR) after 4 weeks of combination therapy.[2,3] The prospect of shorter treatment for HCV genotypes 1 and 4 is appealing because an abbreviated treatment regimen provides a substantial benefit in terms of tolerability, quality of life, and cost.
HCV genotype 4 infection is less common than HCV genotype 1 infection, and available data suggest that their natural history and treatment outcomes are
broadly similar. Thus, treatment recommendations are identical for patients infected with either of these two genotypes.[1]
Abbreviated treatment for HCV genotypes 1 and 4 based on RVR status has not been evaluated in prospective studies. For this reason, we incorporated this strategy into the design of a large, randomized study that has as its overall objective the optimization of combination therapy.
OBJECTIVE
The objective of this analysis is to report sustained virologic response (SVR) rates in patients infected with HCV genotype 1 or 4 who achieved an RVR at week 4 and were assigned to complete 24 weeks of treatment with peginterferon alfa-2a (40KD) plus ribavirin in a large prospective study of response-guided therapy.
METHODS
Patients
Treatment-naive adults with chronic hepatitis C virus genotype 1 or 4 infection were eligible for the trial.
Patients were required to have a positive anti-HCV antibody test, quantifiable HCV RNA by PCR (COBAS AMPLICOR™ HCV MONITOR Test, v2.0), elevated alanine aminotransferase (ALT) levels in serum at baseline, and liver biopsy findings consistent with the diagnosis of chronic hepatitis C.
Treatment
All patients received peginterferon alfa-2a (40KD) (PEGASYS) 180 _g/week plus ribavirin (COPEGUS) 1000 or 1200 mg/day.
The duration of treatment was determined on the basis of a qualitative HCV RNA test after 4 weeks of treatment and/or a quantitative HCV RNA test after 12 weeks of treatment.
Patients with an RVR (HCV RNA <50 IU/mL at week 4) were assigned to complete a total of 24 weeks of combination therapy (Figure 1). The duration of treatment in patients without an RVR was determined on the basis of the virologic response at week 12.
Outcomes
The primary efficacy end point was SVR, defined as undetectable HCV RNA in serum by qualitative PCR at the end of 24 weeks of untreated follow-up (i.e. week 48 in patients with an RVR).
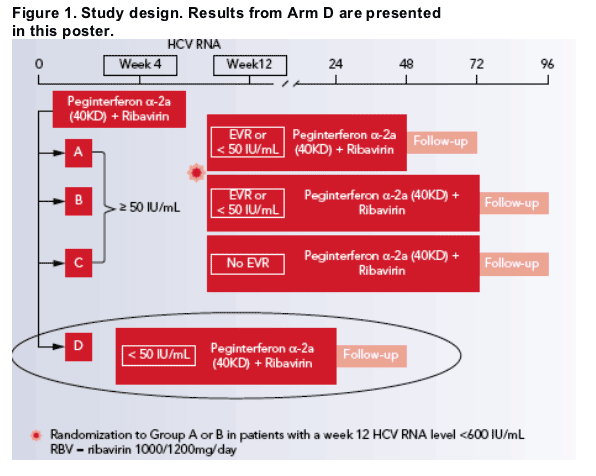
RESULTS
A total of 580 patients were screened for inclusion.
Of the 515 eligible patients treated with peginterferon alfa-2a (40KD) plus ribavirin, a total of 150 individuals (29%) had an RVR at week 4, including 121 of 443 (27%) patients with genotype 1 infection and 29 of 67 (43%) patients with genotype 4 infection.
A total of 142 of 150 patients with an RVR completed 24 weeks of treatment.
Baseline characteristics are presented in Table 1.
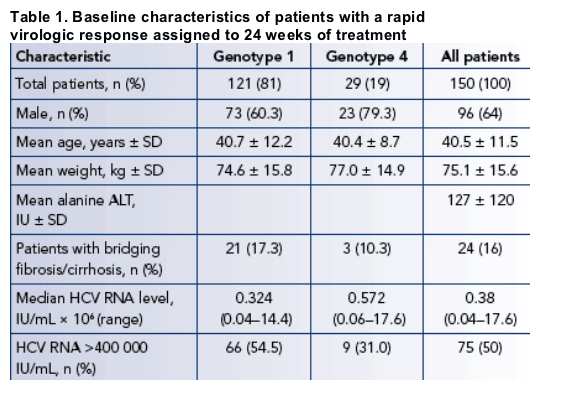
Baseline characteristics predictive of RVR include lower body weight (odds ratio [OR] 0.963, 95% CI: 0.945-0.980; p<0.0001), serum HCV RNA level (>400 000 lU/mL vs. ≦400 000 IU/mL; OR 0.260, 95% CI: 0.164-0.414; p<0.0001), genotype (1 vs. 4; OR 0.295, 95% CI: 0.157-0.555; p=0.0002), and higher serum ALT level (OR 1.007, 95% CI: 1.00 -1.010; p<0.0001).
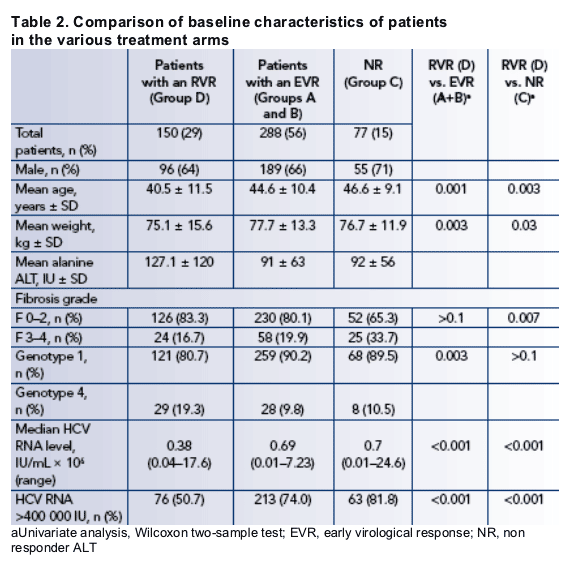
There were significant differences in baseline characteristics between patients with an RVR (group D) and those in other treatment arms in the study (Table 2).
Among patients who completed 24 weeks of treatment, the SVR rate was somewhat higher in those infected with HCV genotype 4 than with genotype 1 (Figure 2).
The difference in SVR rates is attributable to a higher rate of relapse in patients infected with HCV genotype 1 (13/104 [12.5%] patients with end of treatment and end of follow-up results) than genotype 4 (1/26 [3.8%]).
REFERENCES
1. Dienstag JL, McHutchison JG. American Gastroenterological Association medical position statement on the management of hepatitis C. Gastroenterology 2006; 130: 225-230.
2. Jensen DM, Morgan TR, Marcellin P, et al. Early identification of HCV genotype 1 patients responding to 24 weeks peginterferon alpha-2a (40 kd)/ribavirin therapy. Hepatology 2006; 43: 954-960.
3. Zeuzem S, Buti M, Ferenci P, et al. Efficacy of 24 weeks treatment with peginterferon alfa-2b plus ribavirin in patients with chronic hepatitis C infected with genotype 1 and low pretreatment viremia. J Hepatol 2006; 44: 97-103.
|
| |
|
 |
 |
|
|