 |
 |
 |
| |
Differentiation of early virologic response (EVR) into RVR, complete EVR (cEVR) and partial EVR (pEVR) allows for a more precise
prediction of SVR in HCV genotype 1 patients treated with peginterferon alfa-2a (40KD) (PEGASYS) and ribavirin (COPEGUS)
|
| |
| |
Reported by Jules Levin
AASLD, Nov 2-6, 2007, Boston, MA
P. Marcellin,1 D.M. Jensen,2 S.J. Hadziyannis,3 P. Ferenci4
1Universite Paris 7, Clichy, France; 2University of Chicago, Chicago, IL, USA; 3Henry Dunant Hospital, Athens, Greece; 4Department of Internal Medicine IV, Medical University of Vienna, Vienna, Austria
This research was funded by Roche, Basel, Switzerland.
CONCLUSIONS
This analysis demonstrates that the label "difficult-to-cure" is a broad generalization that does not apply to the majority of patients with HCV genotype 1 infection.
Indeed, more than half of genotype 1 patients clear HCV RNA from serum by week 12 of treatment with peginterferon alfa-2a (40KD) plus ribavirin and have an excellent chance of achieving an SVR.
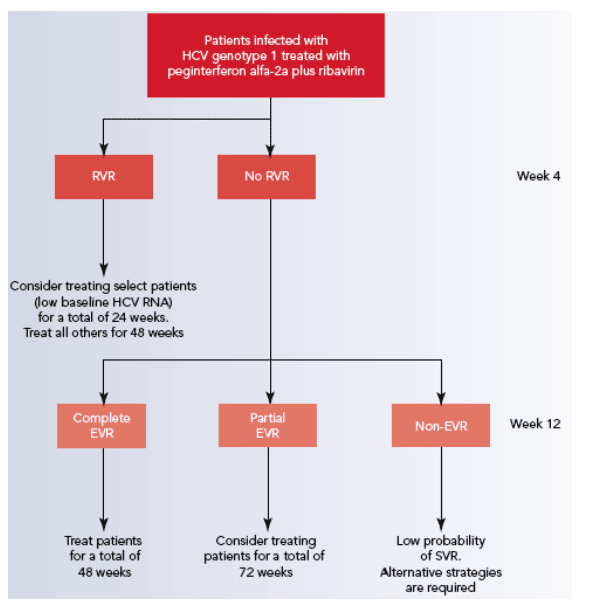
The on-treatment virologic response may be used to customize the treatment duration as follows (Figure 3).
--Select patients achieving an RVR have high rates of SVR and may benefit from an abbreviated 24-week treatment regimen (among patients with low baseline
HCV RNA levels who achieve an RVR, the SVR rate is 93%[7]).[1]
--Patients with a complete EVR also have high rates of SVR, but should be encouraged to remain on therapy for the standard duration of therapy for HCV genotype 1 (48 weeks).
--Patients with a partial EVR have lower rates of SVR with the standard 48-week regimen and may benefit from an intensified 72-week treatment regimen.[8]
--Non-EVR patients have a low chance of achieving an SVR and consideration should be given to changing the treatment strategy.
Response-guided therapy has the potential to optimize treatment outcomes in patients with HCV genotype 1 infection.
INTRODUCTION
In patients with chronic hepatitis C, the on-treatment response at weeks 4 and 12 of pegylated interferon plus ribavirin combination therapy may be used to predict the probability of a sustained virologic response (SVR).
Patients achieving a rapid virologic response (RVR; HCV RNA <50 IU/mL at week 4) have a high rate of SVR, irrespective of HCV genotype.[1,2]
The standard definition of an early virologic response (EVR) is undetectable HCV RNA (<50 IU/mL) by qualitative PCR or a ≥2 log drop in HCV RNA at week 12
by quantitative PCR.[3] However, the positive predictive value of an EVR is not high enough to be a useful clinical predictor of SVR.[4]
It may be possible to improve the prediction of SVR by subdividing patients with an EVR (and no RVR) into those with undetectable HCV RNA (referred to as a complete EVR), those with a ≥2 log drop in HCV RNA but not cEVR (referred to as a partial EVR) and those with <2 log drop in HCV RNA at week 12 (non-EVR). This strategy may allow for tailoring of the treatment duration to the individual response during treatment.
OBJECTIVE
We performed a retrospective analysis of data from two large, multinational phase III studies of patients treated with peginterferon alfa-2a (40KD) plus ribavirin to determine the probability of SVR in patients with an RVR, complete EVR, partial EVR and non-EVR.[5,6]
METHODS
Patients
Treatment-naive adults eligible for these trials had chronic hepatitis C, quantifiable HCV RNA, elevated alanine aminotransferase levels in serum and compensated liver disease.
Only patients infected with HCV genotype 1 were included in this analysis.
Treatment
Patients included in this analysis were randomly assigned to 48 weeks of treatment with peginterferon alfa-2a (40KD) 180 _g/week plus a standard dose of ribavirin (1000 mg/day for patients with a body weight ≦75 kg; 1200 mg/day for patients with a body weight >75 kg).
Outcomes
Virologic response at weeks 4 and 12 was divided into four mutually exclusive categories (Table 1).

RESULTS
A total of 569 patients with HCV genotype 1 infection were included in the present analysis (intention-to-treat population).
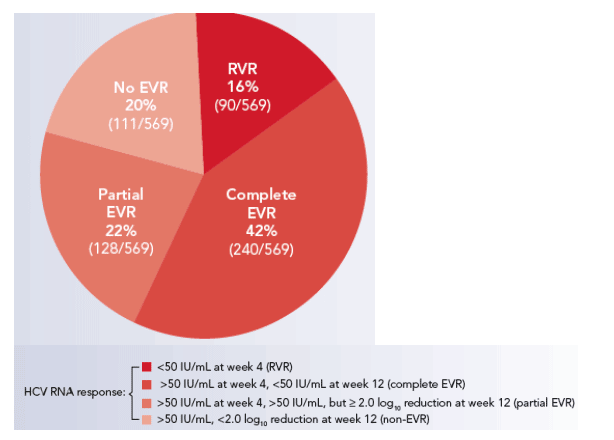
The proportion of patients with an RVR (16%), complete EVR (42%), partial EVR (22%) and non-EVR (20%) are illustrated in Figure 1.
Figure 1. Virologic responses at weeks 4 and 12 in patients treated for 48 weeks with peginterferon alfa-2a 180 _g/week plus ribavirin 1000/1200 mg/day in two large randomized international phase III studies.[5,6]
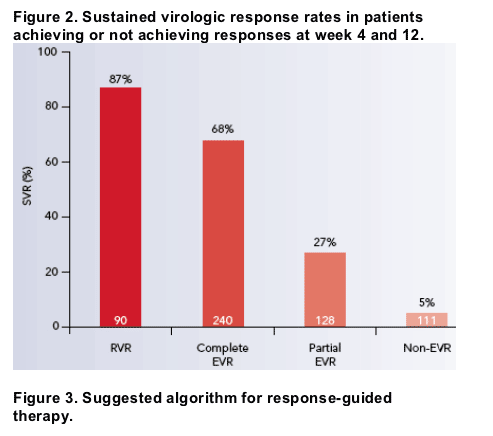
The rate of SVR was highest in patients with an RVR (87%) and decreased progressively in patients with less rapid and complete virologic response: 68%
among those with a complete EVR, 27% among those with a partial EVR and 5% among those without an EVR at week 12 (Figure 2).
REFERENCES
1. Jensen DM, Morgan TR, Marcellin P, et al. Early identification of HCV genotype 1 patients responding to 24 weeks peginterferon alpha-2a (40 kd)/ribavirin therapy. Hepatology 2006; 43(5): 954-960.
2. Shiffman ML, Suter F, Bacon BR, et al. Peginterferon alfa-2a and ribavirin for 16 or 24 weeks in HCV genotype 2 or 3. N Engl J Med 2007; 357(2): 124-134.
3. Dienstag JL, McHutchison JG. American Gastroenterological Association medical position statement on the management of hepatitis C. Gastroenterology 2006; 130(1): 225-230.
4. Ferenci P, Fried MW, Shiffman ML, et al. Predicting sustained virologic responses in chronic hepatitis C patients treated with peginterferon alfa-2a (40 KD)/ribavirin. J Hepatol 2005; 43(3): 425-433.
5. Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med 2002; 347(13): 975-982.
6. Hadziyannis SJ, Sette H, Jr., Morgan TR, et al. Peginterferon-alpha2a and ribavirin combination therapy in chronic hepatitis C: a randomized study of treatment duration and ribavirin dose. Ann Intern Med 2004; 140(5): 346-355.
7. Pegasys. Summary of Product Characteristics. Revised June 2007.
8. Sanchez-Tapias JM, Diago M, Escartin P, et al. Peginterferon-alfa2a plus ribavirin for 48 versus 72 weeks in patients with detectable hepatitis C virus RNA at week 4 of treatment. Gastroenterology 2006; 131(2): 451-460.
|
| |
|
 |
 |
|
|