 |
 |
 |
| |
Clinical Relevance of Rapid Virological Response (RVR) in Decompensated HCV-Related Cirrhosis Treated With Peginterferon and Ribavirin
|
| |
| |
Reported by Jules Levin
AASLD, Nov 2-6, 2007, Boston, MA
Angelo Iacobellis,1 Massimo Siciliano,2 Brigida E. Annicchiarico,2 Maria Rosa Valvano,1 Grazia Anna Niro,1 Laura Accadia,1 Nazario Caruso,1 Giuseppe Bombardieri,2 Angelo Andriulli1
1Division of Gastroenterology, "Casa Sollievo della Sofferenza" Hospital, IRCCS, San Giovanni Rotondo, Italy; 2Institute of Pathology and Semeiotica Medica, Catholic University, Rome, Italy
Summary
RVR, baseline viral load, and HCV G2/3 infection are positive predictors of SVR in patients with chronic hepatitis C and decompensated cirrhosis
The unfavorable rate of viral clearance in HCV G1-infected patients who did not attain RVR (SVR <20%) suggests not prolonging treatment in these patients
There is a clear indication to treat patients with chronic hepatitis C infected with HCV G2 and decompensated cirrhosis (SVR >55%) in Child-Turcotte-Pugh A and B classes
Conclusion
An individualized approach to treatment of patients with chronic hepatitis C and decompensated cirrhosis that accounts for viral genotype and early viral kinetics may help identify those patients most likely to attain SVR
Abstract
Aim: Rapid virological response (RVR), defined as undetectable HCV viral load with qualitative PCR after 4 weeks of treatment, has gained increasing acceptance as a predictor of sustained virological response (SVR) to treatment with pegylated interferon plus ribavirin. We determined whether RVR to combination antiviral therapy could predict sustained response and help optimize HCV treatment in patients with decompensated cirrhosis.
Methods: A total of 94 patients underwent treatment with peginterferon alfa-2b (1.5 _g/kg weekly) and ribavirin (800 or 1000 mg daily) for 48 and 24 weeks for geno 1/4 and geno 2/3, respectively. All patients were monitored on a 24 week follow up off therapy.
Patients: Mean age was 62.4 ± 7 yrs; 50 patients were infected by geno 1/4; 96% of them were staged in Child-Pugh class B and the remaining patients in class A. All patients were naïve to combined antiviral therapy.
Results: Overall, SVR was achieved in 34 patients (36.1%); 8 (16%) with geno 1 or 4, and 26 (59%) with geno 2 and 3 (P < 0.01). At treatment week 4, 34 patients cleared HCV (RVR), 10 patients (25%) with geno 1/4 and 24 (54.5%) with geno 2/3. Four patients with RVR had to stop therapy precociously
for leukopiastrinopenia, 1 patient for severe anemia, and 1 more patient for development of spontaneous bacterial peritonitis. Of 34 RVR patients, 24 achieved SVR (70.6%) at a significant difference between genotypes: 6 of 10 geno 1 and 4 patients (60%), and 18 of 24 geno 2 and 3 (75%; P < 0.01). Twenty-eight (82.3%) of 34 SVR patients had pre-treatment viral load ≦600,000 UI/mL.
Conclusion: The present study further supports the feasibility of antiviral treatment with peginterferon alfa-2b and ribavirin in Child-Pugh class B cirrhotics. Rapid virological response, besides low pre-treatment HCV viral load and genotype 2 and 3, are positive predictive factors of SVR and patients with none of those should be discontinued from treatment. Failure to lose detectable HCV-RNA by qualitative PCR testing after 4 weeks of treatment and pre-treatment viral load >600,000 IU/mL were strongly associated with failure to respond to a full 48-week course of therapy in geno 1/4 (NPV 96.5%).
Background
⋅ The safety and efficacy of anti-hepatitis C virus (HCV) treatment among cirrhotic patients with hepatic decompensation is unclear
- Most concerns focus on the likelihood of attaining acceptable sustained virologic response (SVR) rates while mitigating the increased risk for
treatment-related adverse events1,2
⋅ In a recent study of 66 patients with decompensated cirrhosis who received pegylated interferon (PEG-IFN) alfa-2b (PegIntron®) plus ribavirin, 41% of patients completed 24 weeks of treatment, 39% required dose reductions, and 20% discontinued because of adverse events3
- The risk for further episodes of decompensation was significantly lower for treated patients who attained SVR than for untreated controls
⋅ Rapid virologic response (RVR), defined as undetectable HCV RNA at week 4 of treatment, has gained acceptance as a predictor of SVR; however, its role in the treatment of patients with decompensated cirrhosis has not been explored
Aim
To evaluate the predictive value of early viral kinetics on SVR in patients with chronic hepatitis C and hepatic decompensation who were treated with PEG-IFN alfa-2b plus ribavirin
Methods
Patients
Patients who were naive to combination treatment and who had chronic hepatitis C and a history of hepatic decompensation (Child-Pugh class A or B), including ascites, bleeding, or encephalopathy
- Inclusion and exclusion criteria are listed in Table 1
The study was approved by the ethics committee and conducted according to the guidelines of the International Conference on Harmonization for Good Clinical Practice

Treatment
All patients started receiving PEG-IFN alfa-2b (1.5 μ g/kg/wk) plus oral ribavirin approximately 1 month after hospital discharge
- Patients infected with HCV genotype (G) 1 or G4 received 1000 mg/d (<75 kg body weight) or 1200 mg/d (≥75 kg body weight) of ribavirin
- Patients infected with HCV G2/3 received 800 mg/d (<75 kg body weight) or 1000 mg/d (≥75 kg body weight) ribavirin
- Patients infected with HCV G1/4 were treated for 48 weeks, and patients infected with HCV G2/3 were treated for 24 weeks
All patients were followed up for 24 weeks after completing treatment
Assessments
The primary efficacy end point was SVR, defined as undetectable HCV RNA 24 weeks after completing therapy
- Secondary end point was to determine whether early monitoring of viral kinetics is useful in predicting SVR
⋅ RVR: undetectable HCV RNA at week 4
⋅ Early virologic response (EVR): undetectable HCV RNA at week 12
⋅ HCV RNA levels were assessed at weeks 4, 12, 24, and 48 (G1) during treatment and at follow-up week 24
- Baseline HCV RNA was quantified by polymerase chain reaction (PCR) assay (Cobas Amplicor Monitor 2.0; Roche Diagnostics, F. Hoffmann-La Roche, Basel, Switzerland; lower limit of detection 600 IU/mL)
- On-treatment and off-treatment HCV RNA levels were determined by qualitative PCR assay (Amplicor HCV test, Roche Diagnostics; lower limit of detection 50 IU/mL)
⋅ HCV genotyping was performed by reverse hybridization (Innolipa HCV; Innogenetics, Ghent, Belgium)
Statistical and Logistic Regression Analyses
⋅ All analyses were performed according to the intention-to-treat principle
⋅ Continuous variables were expressed as mean (± SD), and chi-square and Student t tests performed as appropriate; all P values were 2 tailed
⋅ Age, sex, genotype, baseline viral load, RVR, neutrophil count, white blood cell count, platelet count, and Child-Turcotte-Pugh score were included as variables in univariate and multivariate analyses
Results
Baseline Patient Characteristics (Table 2)

Virologic Response Rates
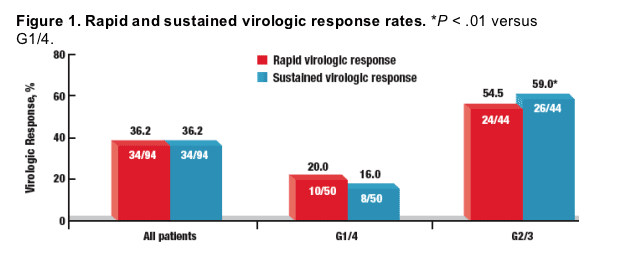
⋅ 36.2% (34/94) of patients attained RVR (G1/4, n = 10; G2/3, n = 24) (Figure 1)
⋅ 36.2% (34/94) of patients attained SVR (G1/4, n = 8; G2/3, n = 26) (Figure 1)
⋅ SVR rates were significantly lower in patients with HCV G1/4 infection than in those with HCV G2/3 infection (16% vs 59%, P < .01)
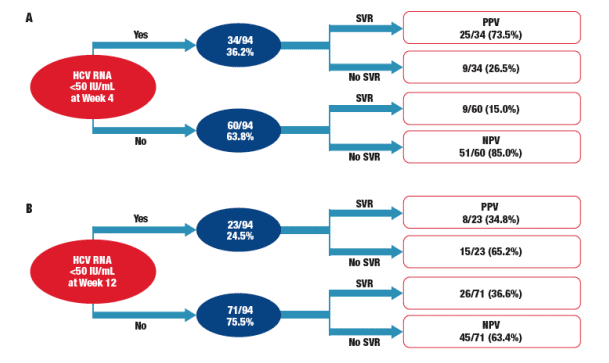
Correlation Among RVR, EVR, and SVR
Almost 75% of patients who attained RVR also attained SVR
- Positive predictive value (PPV) of RVR for SVR: 73.5% (25/34) (Figure 2A)
More patients infected with HCV G2/3 who attained RVR also attained SVR than did patients infected with G1/4 who attained RVR (G1/4, n = 6; G2/3, n = 19)
- Negative predictive value (NPV; no RVR and no SVR): 85.0%
Slightly more than one third of patients who also attained EVR also attained SVR (G1/4, n = 2; G2/3, n = 6)
- PPV of EVR for SVR: 34.8% (Figure 2B)
More patients infected with HCV G2/3 who also attained EVR also attained SVR than patients infected with HCV G1/4 who attained EVR (G1/4, n = 2; G2/3, n = 6)
- NPV (no EVR and no SVR): 63.4%
Figure 2. Predictive value of (A) RVR and (B) EVR for SVR. EVR = early virologic response; NPV = negative predictive value; PPV = positive predictive value;
RVR = rapid virologic response; SVR = sustained virologic response.
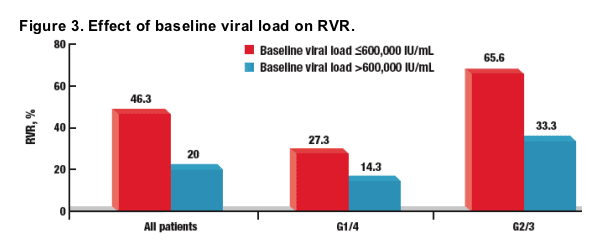
RVR According to Viral Load at Baseline
Patients with baseline viral load ≦600,000 IU/mL were significantly more likely to attain RVR than those with a baseline HCV RNA level >600,000 IU/mL (Figure 3)
Overall, 46.3% (25/54) of patients with baseline viral load ≦600,000 IU/mL attained RVR
- More patients infected with HCV G2/3 (21/32) than patients infected with HCV G1/4 (6/22) with baseline viral load ≦600,000 IU/mL attained SVR
Overall, 20% (8/40) of patients with baseline viral load >600,000 IU/mL attained RVR
- More patients infected with HCV G2/3 (4/12) than patients infected with G1/4 (4/28) with baseline viral load >600,000 IU/mL attained SVR
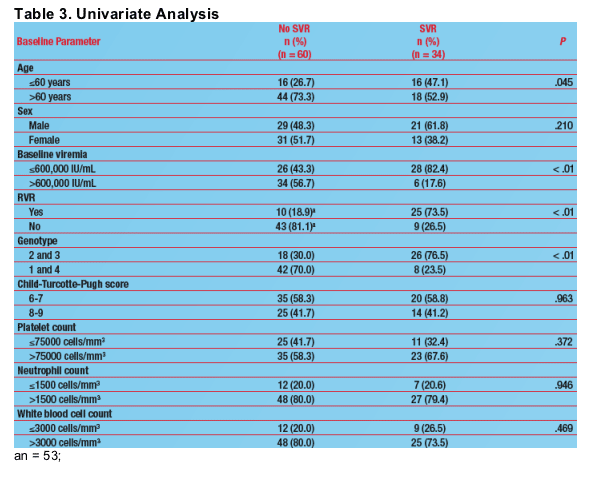
Predictors of Virologic Response
Univariate (Table 3) and multivariate (Table 4) analyses revealed that baseline viremia, RVR, and genotype were significant predictors of SVR

References
1. Navasa M et al. J Hepatol. 2007;46:185-188.
2. Everson GT et al. Hepatology. 2005;42:255-262.
3. Iacobellis A et al. J Hepatol. 2007;46:206-212.
|
| |
|
 |
 |
|
|