 |
 |
 |
| |
Nevirapine Hypersensitivity is Different for Treatment-Experienced
|
| |
| |
Reported by Jules Levin
9th International Workshop on Adverse Drug Reactions and Lipodystrophy in HIV
July 19-21, 2007, Sydney Australia
Incidence of nevirapine-associated hypersensitivity reactions is different in patients with prior treatment-experience compared to treatment-naïve patients ATHENA cohort
Ferdinand Wit*, Anouk Kesselring, Luuk Gras,
Clemens Richter, Ineke van der Ende, Kees Brinkman,
Joep Lange, Frank de Wolf, Peter Reiss
Center for Poverty-related Communicable Diseases, Academic Medical Center, University of Amsterdam, Amsterdam, the Netherlands
ABSTRACT
Background: Current recommendations that nevirapine (NVP) should not be initiated in females with CD4 counts greater than 250 or in males with CD4 counts greater than 400 are based on findings in treatment-naive patients. The incidence ratio is unknown for treatment-experienced patients with low pre-ART CD4 counts are current CD4 counts above the mentioned threshold, who switch to NVP-containing regimens. Whether current guidelines are applicable in such patientr is unclear.
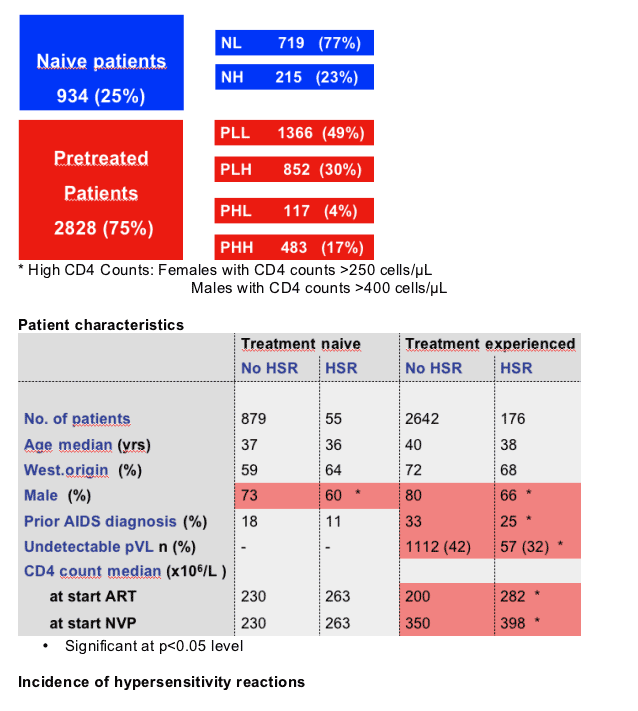
Methods: All patients in the Athena cohort who ever used NVP were included. We identified patients who discontinued NVP because of NVP-induced HSR within 18 months after starting NVP. We grouped patients according to CD4 counts at the start of NVP-based HAART, being either high (>250 for females and >400 for males) or low. Treatment-experienced patients were also subdivided according to the last available pre-ART CD4 count using the same criteria: treatment-naïve, low current CD4 (NL), treatment-naïve, high current CD4 (NH); pretreated, low pre-ART CD4 and low current CD4 (PLL); pretreated, low pre-ART CD4 and high current CD4 (PLH); pretreated, high pre-ART and low current CD4 (PHH). Risk factors for developing HSR were assessed using multivariate logistic regression.
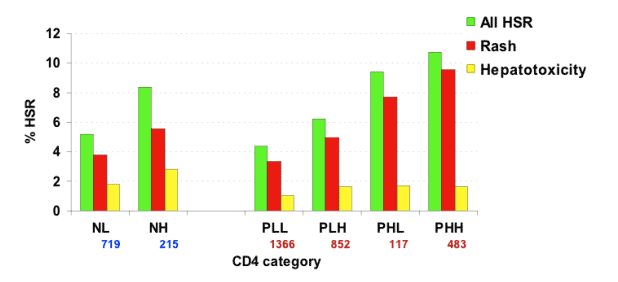
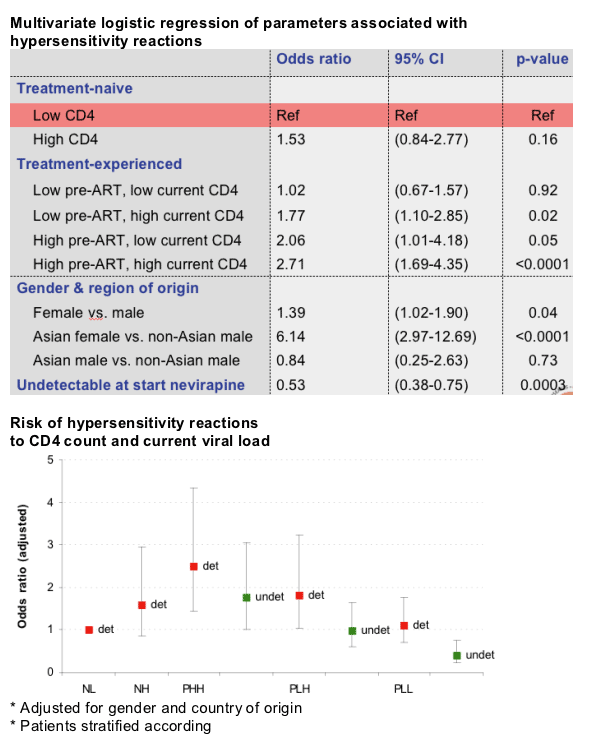
Results: In patients that ever used NVP (n=3,752), HSR occurred in 231 (6.2%). We found no difference in HSR rate between the NL and PLL groups (5.2% vs 4.4%, p=0.44). Similarly, NH patients had a HSR rate comparable to the HSR rate in PHH patients (8.4% vs 10.8%, p=0.33). The HSR rate in the PLH group (6.2%) was higher than in the PLL (4.4%, p=0.057), but lower when compared with the rate in the PHH group (10.8%, p=0.003). Using PLH as the reference group, the multivariate adjusted odds ratios (OR) for developing HSR were significantly lower for NL (OR 0.54; 95% CI % 0.35-0.82) and PLL (OR 0.55; 95% CI 0.38-0.78). Other independent risk factors include female gender and Asian race. Having an undetectable viral load at the start of NVP use was also associated with reduced risk for developing HSR (OR 0.52; 95% CI 0.38-0.71). Using NL as the reference group, PLH patients with a detectable viral load at the start of NVP use had a significantly higher risk for HSR (OR 1.87; 95% CI 1.11-1.32, whereas PLH patients with undetectable viral load did not (OR 1.03; 95% CI 0.66-1.61).
Conclusions: Treatment-experienced patients with low pre-ART CD4 counts, high current CD4 counts and undetectable viral load have a similar risk for developing HSR when they switch to NVP compared with treatment-naïve patients with low CD4 counts. This suggests that NVP may be safely initiated in such patients. However, in similar patients with a detectable viral load, it is prudent to continue adhering to current guidelines.
ORAL PRESENTATION
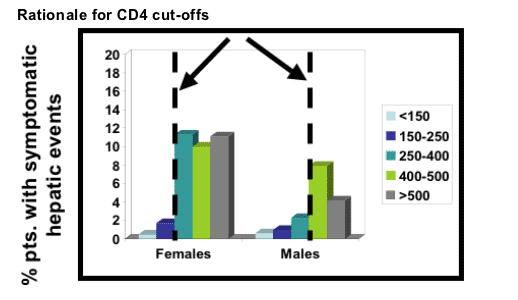
Females with CD4 counts >250 cells/μL
Males with CD4 counts >400 cells/μL
Mainly derived from patients starting NVP while treatment naïve
AIM OF THE STUDY
Focus on treatment experienced patients
--Incidence of HSR
--Relationship with CD4 counts prior to starting cART and current CD4 counts
--Relationship with viral load
--Identify other independent risk factors
METHODS
--Retrospective cohort analysis
--Selection of HIV-1 infected patients who ever used NVP with available pre-ART and current CD4 counts
--Identification of patients who had to stop NVP due to AE compatible with HSR within 18 weeks

Statistical analyses--
Logistic regression model
Potential other risk factors considered:
--Gender
--Country of origin
--Plasma HIV RNA levels at start NVP
--Age
--Prior AIDS diagnosis
--HBV or HCV coinfection
--BMI, weight
--Prior exposure to EFV
--Nucleosides/nucleotides used with nevirapine
CONCLUSIONS
Majority of NVP-HSR consisted of skin rashes without concomitant liver enzyme elevations
Independent factors associated with NVP HSR
-- Female gender (and Asian females >6 fold odds!)
-- High pre-ART and current CD4 cell count
-- Detectable HIV RNA load at start NVP: odds
almost doubled!
Compared to treatment-naive patients starting NVP with low CD4 counts:
--Patients in PLH group are at increased risk for HSR
--Patients in PLH group with undetectable load do not have a significantly different risk for HSR
--Compared to treatment-naive patients starting NVP with low CD4 counts:
Consider switch to NVP in PLH group if benefit outweighs the risk
LIMITATIONS OF STUDY
Predominantly white males in ATHENA cohort:
Results should be interpreted with caution for other groups of patients
|
| |
|
 |
 |
|
|