 |
 |
 |
| |
Intravenous silibinin for treatment of nonresponders to peginterferon/ribavirin therapy in chronic hepatitis C
|
| |
| |
Reported by Jules Levin
ASSLD No 4 2008 San Francisco, CA
P. Ferenci, T. Scherzer, K. Rutter, S. Beinhardt, H. Hofer, M. Schoniger-Hekele, P.E. Steindl-Munda, Internal Medicine 3, Medical University of Vienna, Wien, AUSTRIA; H. Kerschner, H. Holzmann, Clinical Virology, Medical University of Vienna, Wien, AUSTRIA;
ABSTRACT
Background: By chance we observed that iv. silibinin (SIL) had marked antiviral properties in patients with chronic hepatitis C, confirming in vitro findings that SIL inhibited viral replication. The aim of this investigation was a dose finding study and the assessment of safety.
Methods: 20 nonresponders (17 male, 3 female; age: 52.7±12.8 years, 17 genotype 1; 2=1; 4=2; F3/4: n=7, F1/2: n=10; n.a.:3) to full dose of peginterferon/ribavirin (PR) combination therapy were included. Nonresponse was defined by the lack of a > 2log drop of viral load after 12 weeks of full dose PR therapy and/or by not achieving an end of treatment response. Patients first received daily 5, 10 or 15 mg/kg 20mg/kg SIL (Rottapharm-Madaus, Koln, Germany) infused over 4 hrs for 14 consecutive days. On day 8 treatment with 180μg/wk PegIFNé-2a and 1-1.2 g/d ribavirin was started. After day 14 patients received 280 mg silymarin (Rottapharm-Madaus, Koln) TID per os. Viral load was determined daily preinfusion by the TaqMan PCR assay (Cobas Ampliprep/ Cobas TaqMan HCV Test; limit of detection, 15 IU/mL, Roche Diagnostics).
Results:
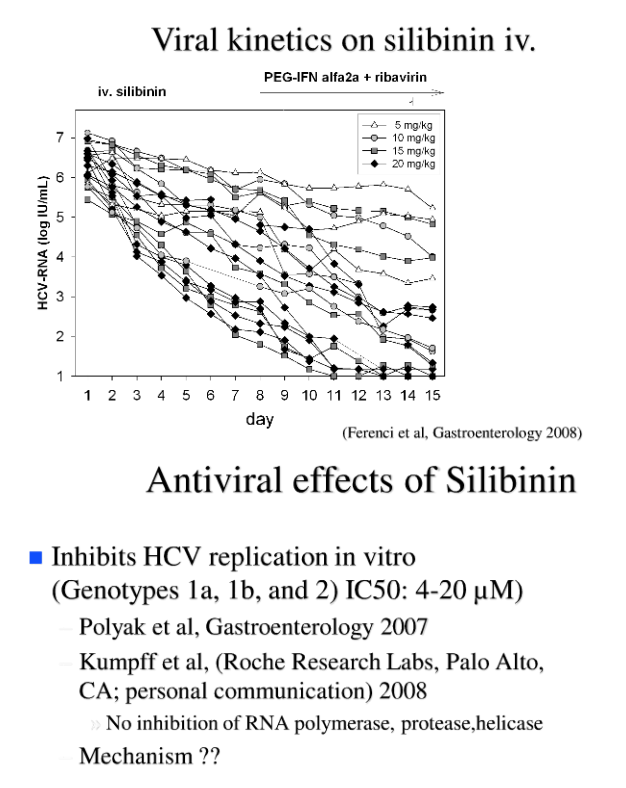
After 7 days of silibinin monotherapy the 5mg/kg dose was marginally effective (n=3, log drop 0.55±0.5), whereas the 10 mg/kg (n=3, log drop 1.41±0.59), 15 mg/kg (n=5, log drop 2.11±1.15) and 20 mg/day doses (n=9, 3.02±1.01) led to a significant decrease in viral load (p<0.001).
After 1 week of combined silibinin and PR therapy viral load decreased further (log drop: 5 mg/kg: 1.63±0.78; 10 mg/kg: 4.16±1.28; 15 mg/kg 3.69±1.29; 20 mg/kg 4.8± 0.89; all p<0.0001 vs. baseline).
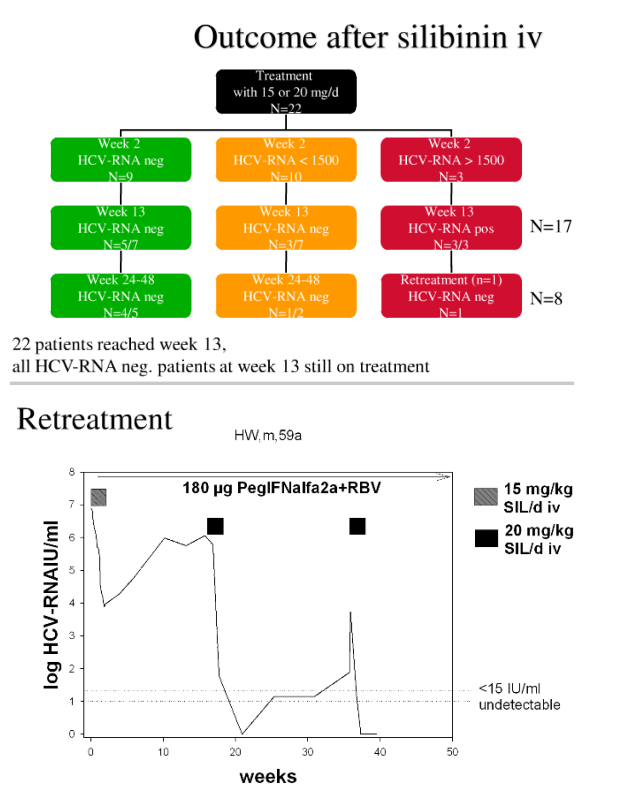
Two of the 5 and 4 of the 9 patients in the 15 mg/kg and 20 mg/kg groups, respectively had HCV-RNA<15 IU/mL at day 15. Further 4 had a HCV-RNA < 50 IU/mL. 7 of these patients had a rapid virologic response (week 5 = after 4 weeks of PR combination therapy), and 8 (57%) had HCV-RNA <15 IU/mL at week 13, one relapsed at week 9.
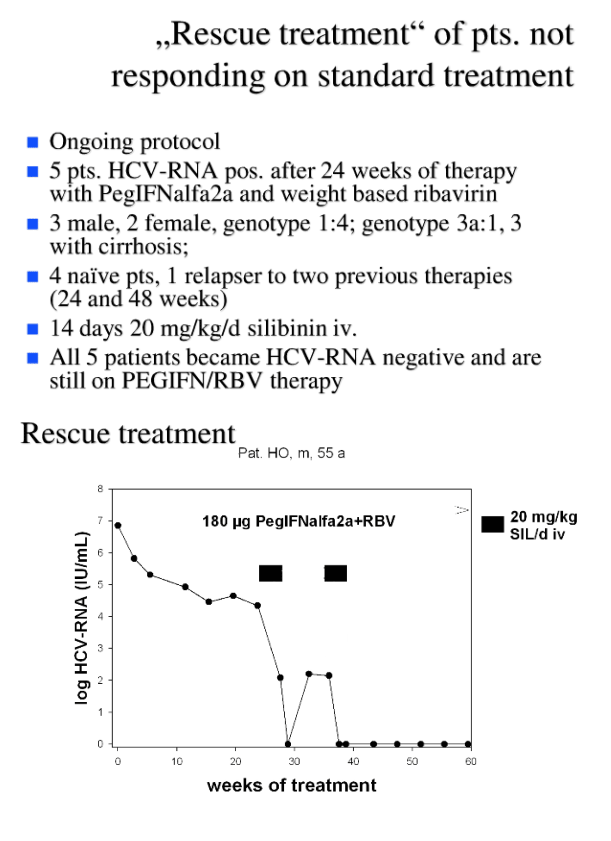
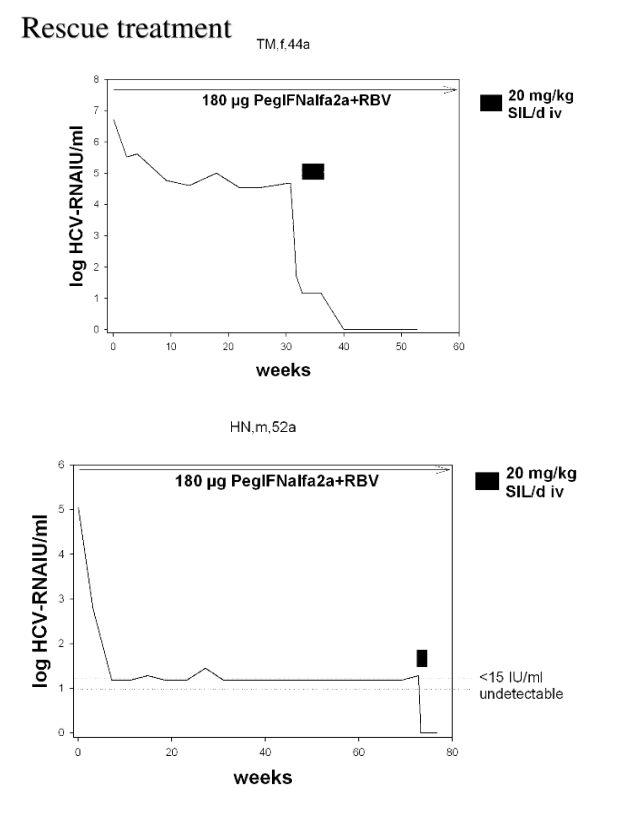
HCV-RNA increased in all patients with HCV-RNA > 50 IU/mL at the end of the infusion period. HCV-RNA became undetectable in 3 of them after a second cycle of SIL infusions given after week 13. All responders are still on PR therapy. SIL treatment was well tolerated.
Conclusion: Intravenous SIL is a potent antiviral agent and may be a treatment option for treatment of PR nonresponders.




|
| |
|
 |
 |
|
|