 |
 |
 |
| |
A Study of Telaprevir Combined with Peginterferon Alfa-2a and Ribavirin in Patients with Well-documented Non-response or Relapse After Previous Peginterferon Alfa-2a and Ribavirin Treatment: Interim Analysis
|
| |
| |
Reported by Jules Levin
59th Annual Meeting of the American Association for the Study of Liver Diseases, San Francisco, CA, USA, October 31-November 4 2008
Mitchell L Shiffman,1 Thomas Berg,2 Fred Poordad,3 Jean-Pierre Bronowicki,4 Andrew J Muir,5 Stuart C Gordon,6 Shelley George,7 Nathalie Adda,7 John G McHutchison5
1Virginia Commonwealth University Medical Center, Richmond, VA, USA; 2Charite Virchow Klinikum, Berlin, Germany; 3Cedars-Sinai Medical Center, Los Angeles, CA, USA; 4CHU Nancy, Vandoeuvre-les-Nancy, France;
5Duke Clinical Research Institute and Duke University, Durham, NC, USA; 6Henry Ford Hospital, Detroit, MI, USA; 7Vertex Pharmaceuticals Inc., Cambridge, MA, USA
Author Conclusions
In this interim analysis of telaprevir-based treatment in HCV genotype 1-infected patients with well-characterized null response, partial response or relapse to prior Peg-IFN plus RBV treatment:
-- 65% were HCV RNA <10 IU/mL at Week 4
-- 68% were HCV RNA <10 IU/mL at Week 12
-- 59% were HCV RNA <10 IU/mL at Week 24.
(From Jules: At week 24, 43% of null-responders (n=42) achieved undetectable HCV viral load, 82% of partial responders did also (n=22), and 71% of relapsers (n=7) did as well. See tables in this article below)
As of the data cut-off point, incidence of viral breakthrough through
Week 24 was:
-- prior relapser = 0 patients, prior viral breakthrough = 1 patient, prior partial responder = 2 patients (6%), null responder = 10 patients (20%) (5 patients within the first 4 weeks).
The safety profile was consistent with that previously described in the PROVE trials. Four subjects discontinued due to rash, and one due to anemia.
Continuing follow-up of these patients will be required to evaluate the ultimate clinical significance of these promising early findings.
A Phase 3 study in patients who previously failed Peg-IFN and RBV (REALIZE) is ongoing.
Introduction
Many hepatitis C virus (HCV) genotype 1 patients do not achieve sustained virologic response (SVR) after a 48-week regimen of peginterferon alfa-2a/2b (Peg-IFN, P) and ribavirin (RBV, R).1-3
Retreatment of prior PR non-responders (null and partial responders) and relapsers with PR achieved SVR rates of less than 10% and 40%, respectively.4-7
Telaprevir (TVR, VX-950) is a specifically targeted antiviral therapy for HCV (STAT-C) that potently and selectively inhibits the HCV NS3-4A protease.8
This is an ongoing, open-label study of TVR combined with Peg-IFN and RBV (T/PR) in patients with HCV genotype 1 infection who did not achieve SVR in PR control arms of the Phase 2 PROVE (investigation of HCV PROtease inhibition for Viral Evaluation) studies.
Methods
Primary objectives:
-- Provide access to TVR-based treatment to patients enrolled in the Peg-IFN alfa-2a plus RBV control groups of VX05-950-104 (Study 104, PROVE1), VX05-950-104EU (Study 104EU, PROVE2) and VX06-950-106 (Study 106, PROVE3) who stopped treatment due to inadequate response to treatment, or who relapsed after treatment
-- Demonstrate the efficacy of T/PR in well characterized, HCV genotype 1-infected patients who did not achieve SVR with prior Peg-IFN and RBV treatment
-- Evaluate the safety of T/PR in these patients.
Secondary objectives:
-- Perform intrapatient comparisons of antiviral response to T/PR with the antiviral response to prior PR treatment
-- Determine the antiviral response to T/PR in patients characterized as null responders, partial responders, having virologic breakthrough, or relapsers during prior PR treatment in PROVE1, PROVE2 and PROVE3.
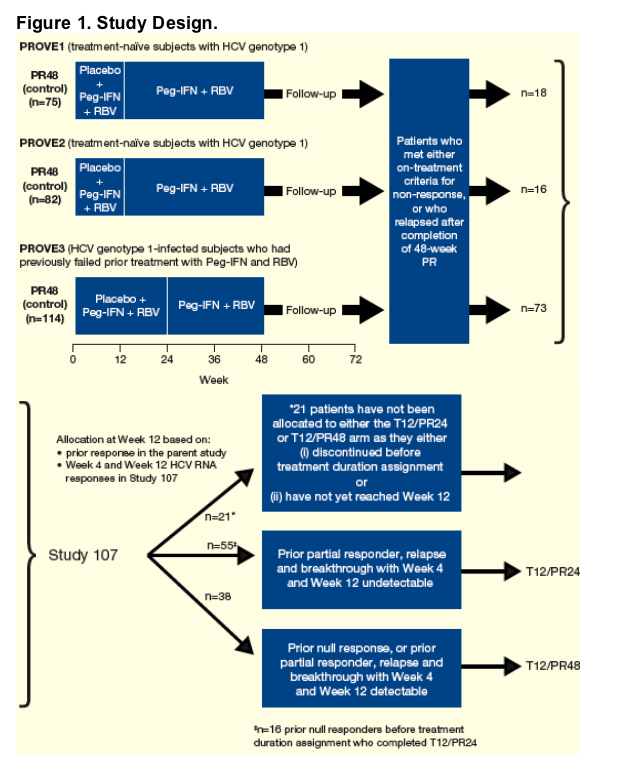
Study 107 Design
Open-label treatment of patients from control arms of PROVE1, PROVE2 and PROVE3 who met either on-treatment criteria for non-response or who relapsed after completion of 48 weeks of Peg-IFN + RBV (Figure 1).
Patients who discontinued therapy because of adverse events (AEs) in the PROVE studies were not eligible for inclusion.
Stopping rule criteria in Study 107 is shown in Table 1.

Methods
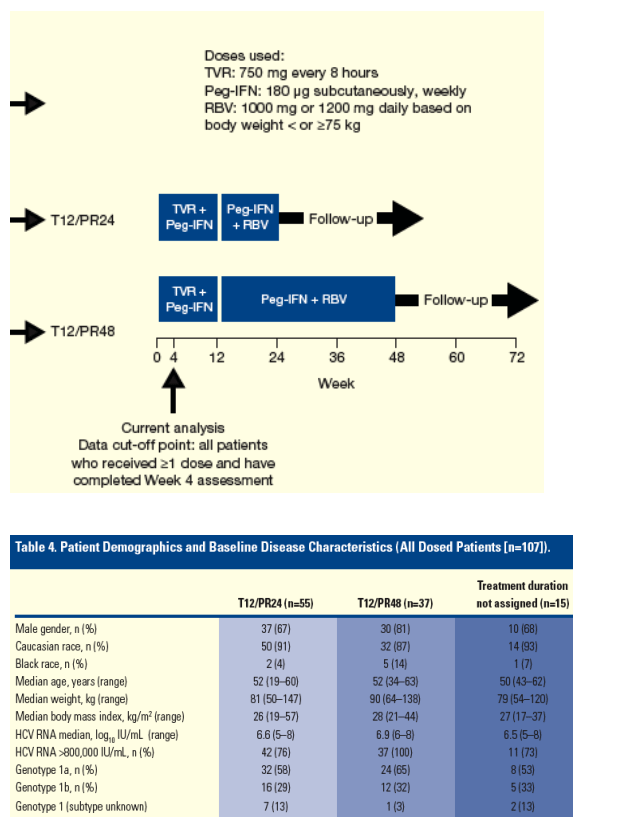
Preliminary Analysis
This pre-planned preliminary analysis included all enrolled patients who received at least one dose of study regimen and who completed at least the Week 4 assessment up to the data cut-off point.
Virologic Responses
This preliminary analysis is based on the patients who reached at least Week 4 assessment (intent-to-treat [ITT] analysis; Table 5).
Table 5. Week 4 HCV RNA by Prior Response to Control Peg-IFN + RBV Arm in the PROVE Studies (ITT Analysis).
From Jules: 75% of null-responders (36/48) had <25 IU/mL at week 4; 97% of partial responders ((32/33) had <25 IU/mL at week 4; 100% of relapsers (22/22) had undetectable at week 4.
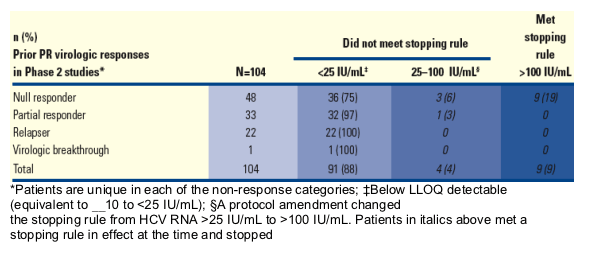
Table 6 shows the virologic responses obtained by prior response to control Peg-IFN + RBV arm in the PROVE studies at Week 12 (ITT analysis).
Table 6. Week 12 HCV RNA by Prior Response to Control Peg-IFN + RBV arm in the PROVE Studies (ITT Analysis).
From Jules: 58% of null-responders (28/48) had <10 IU/ml (undetectable viral oad) at week 12 79% of partial responders had <10 Iu/mL at week 12; 73% of relapsers (16/22) had <10 Iu/mL at week 12.
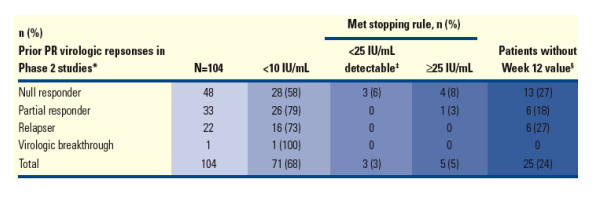
Figure 3 shows the proportion of patients achieving undetectable HCV RNA through Week 24 (modified ITT)
-- Includes only in the denominator by visit, patients who have either reached that visit, or discontinued before, or were detectable. All patients who were still on treatment but haven't yet reached the designated visit are not counted in the denominator.
Figure 3. Proportion of Patients Achieving Undetectable HCV RNA Through Week 24 by Prior Response to
Peg-IFN + RBV Arm in the PROVE Studies (Modified ITT Analysis).
From Jules: At week 24, 43% of null-responders (n=42) achieved undetectable HCV viral load, 82% of partial responders did also (n=22), and 71% of relapsers (n=7) did as well.
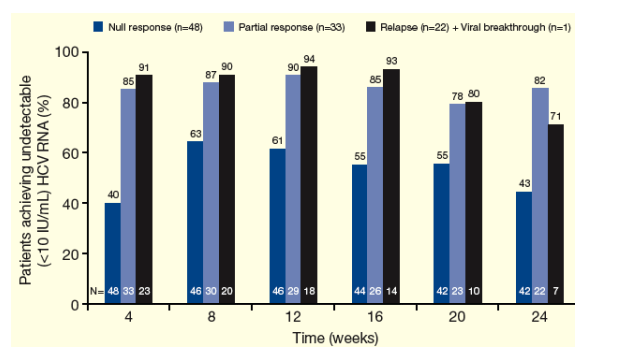
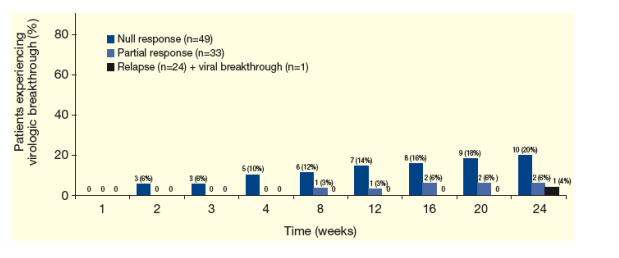
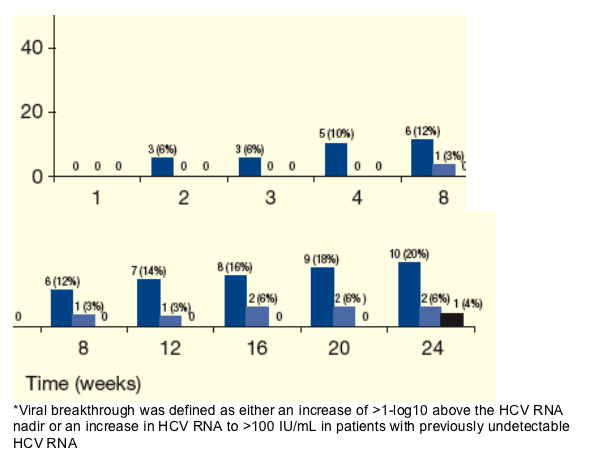
Figure 4 shows the proportion of patients with virologic breakthrough following T/PR treatment.
-- No virologic breakthrough was observed in patients who previously relapsed in the PROVE studies (on control Peg-IFN + RBV arm).
Figure 4. Cumulative Virologic Breakthrough* Following T/PR Treatment According to Prior Response to Control Peg-IFN + RBV Arm in the PROVE Studies.
From Jules: Null-responders had the most virologic breakthroughs: 12% (n=6) at week 8, and 20% (n=10) at week 24. Partial responders had very low breakthrough rates, 6% at week 24 (n=2).
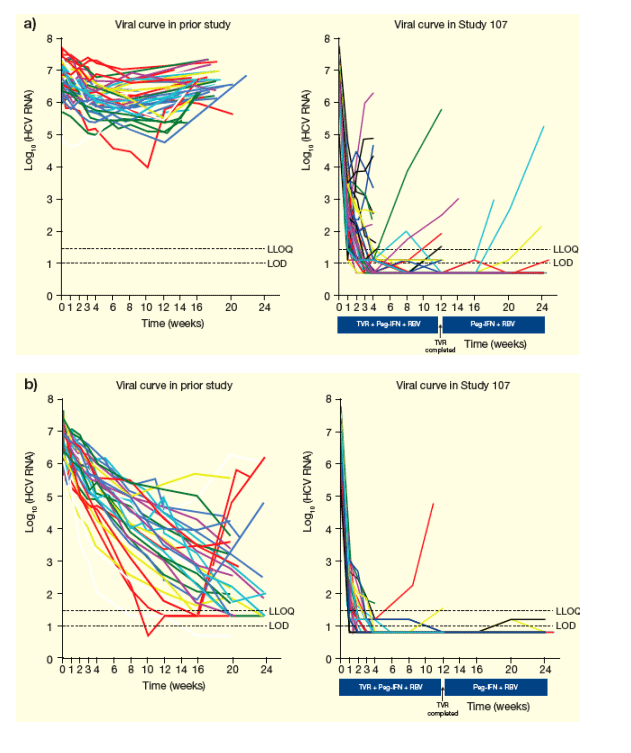
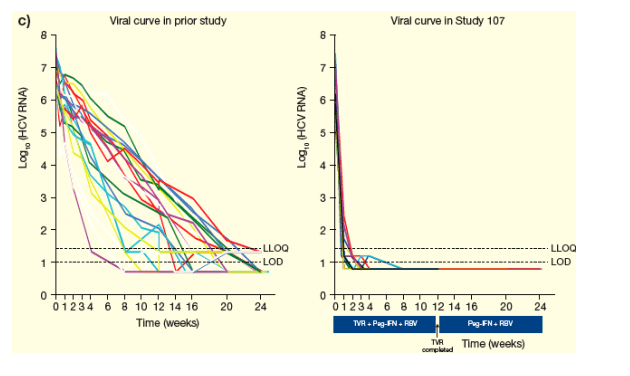
Viral curves through Week 24 are shown in Figures 5a-c.
Figure 5. Viral curves through Week 24 for A) Null Responders, B) Partial Responders and C) Relapsers.
From Jules: I expect null-responders would do well with 2 oral drugs on board, as you can see in the graph on the right in Study 107 when receiving telaprevir patients had a good initial decline in viral load but some patients started to rebound, so with 2 oral drugs on board I they would respond well and not rebound. Roche is just starting the first study of 2 oral drugs now, with R7128+ITMN191. But this first study is an initial study.
Safety and Tolerability
The types of AEs encountered were those that are expected with Peg-IFN + RBV, and are consistent with those previously reported in patients treated with TVR-based regimens in the PROVE studies (Table 7).

REFERENCES
1. Manns MP, et al. Lancet 2001;358:958-65.
2. Fried MW, et al. N Engl J Med 2002;347:975-82.
3. Hadziyannis SJ, et al. Ann Intern Med 2004;140:346-55.
4. Shiffman ML. Hepatology 2002;36:S128-34.
5. Shiffman ML, et al. Gastroenterology 2004;126:1015-23.
6. Ahmed F, et al. Gastroenterol Clin North Am 2004;33:513-26.
7. Poynard T, et al. 43rd EASL 2008, Milan, Italy, April 23-27 2008.
8. Lin K, et al. Antimicrob Agents Chemother 2004;40:4784-92.
|
| |
|
 |
 |
|
|