 |
 |
 |
| |
Abacavir & MI (myocardial infarction) Risk in D.A.D Study
|
| |
| |
'Big Cohort Study Finds Evidence That Abacavir Boosts Heart Attack Risk'
15th Conference on Retroviruses and Opportunistic Infections
February 3-6, 2008
Boston
Mark Mascolini
From Jules Levin: below is a report of the data from Mark Mascolini as reported by the DAD Study authors. But here is my take. The DAD Study is a cohort study of multiple cohorts. Interpreting findings from a cohort study is not straightforward. In fact findings from a cohort study should sometimes be taken with a grain of salt. It is often difficult to be firm regarding findings from a cohort study because it is not as straight forward as a randomized study. A randomized study is prospective, that is, they would randomize patients to either abacavir or another nuke and follow them prospectively to see who developed MIs. In this study and cohort studies they look back retrospectively. When considering the findings of a cohort study, and DAD is a cohort study of cohorts, it is more difficult to adjust for confounding factors. For example, causes of a MI could be lipids elevations, smoking, blood pressure elevated or hypertension, diabetes, fat loss/gain, elevated glucose, and other potential factors. The DAD Study authors did in this case say they adjusted for these potential risk factors and found abacavir itself was associated with MI. But it is more difficult for a cohort study to adjust for these risk factors than it is for a prospective study. Several things don't make sense in this study. The authors said using abacavir for 3 years was not any more of a risk, according to their findings, than using abacavir for 6 months. They said that as soon as abacavir was stopped the risk for MI disappeared. They said that prior use of d4T was not a confounding factor; so another words if a patient used d4T for several years and then switched to abacavir, the prior use of d4T did not affect their finding of MI being associated with abacavir use. The study authors referred to potential bias from 'channeling'. This means that patients who had higher risks for heart disease were put on abacavir by docs because the perception was that abacavir was safe because it was not associated with lipids elevations. By looking at Table 1 in the poster patients with recent abacavir use, and it was recent abacavir use that was associated with MI in this study, had a higher percentage (28%) of high 10-year CVD risk as defined by Framingham equation for risk while patients in this study who had no recent abacavir or ddI use had a lower 10-year risk (21%). More patients on recent use of abacavir than not on recent abacavir or ddI use were current smokers (46% vs 40%). Again, the study authors said they accounted for all these potentially confounding risk factors and found abacavir was associated with an increased risk for MI. This study created quite a lot of discussion and drew attention at this meeting so I think there will be future studies to examine this question and try to answer it. I will wait to see the results from future studies before putting much stock into the DAD Study findings reported here. In speaking with the study researchers they felt confident of their findings but said since it's a cohort study they can't be sure of their findings. By the way I spent quite a lot of time today speaking to the study authors and to a good number of other researchers about this study in order to evaluate this study finding.
Back to report of the data by Mark Mascolini
Abacavir raised the risk of myocardial infarction (MI) 90% in the multicohort D:A:D study, and didanosine (ddI) upped the risk 49% [1]. But thymidine nucleoside analogs, zidovudine (AZT) and stavudine (d4T), did not make MI more likely in the D:A:D cohorts.
As earlier research consistently shows, HIV-infected people with classic cardiovascular risk factors stand the greatest chance of suffering a heart attack. But abacavir and ddI independently compounded that risk. Although the absolute (versus relative) risk of MI with abacavir or ddI was small, the researchers "recommend that patients receiving abacavir or ddI should consult their doctor and discuss whether a modification of their anti-HIV drug regimen is appropriate." They warn people not to stop abacavir or ddI without talking to their clinician first.
D:A:D investigators began this inquest with the hypothesis that thymidine analogs hoist MI risk because their use correlates with high lipids and insulin resistance. But to be thorough, the researchers also assessed the potential impact of the nonthymidine analogs abacavir, ddI, and lamivudine (3TC) on MI risk. Already-published D:A:D results show that protease inhibitor (PI) regimens make heart attacks 16% more likely for every year of treatment [2].
During 157,912 person-years of follow-up in the new analysis, 517 people had a heart attack. The investigators figured MI risk with cumulative nucleoside use, recent use (still using or stopped in past 6 months), or past use (last used more than 6 months ago). They adjusted this analysis for demographic factors, standard cardiovascular risk factors, and cumulative exposure to other antiretrovirals, including tenofovir and the major PIs and nonnucleosides.
Cumulative or recent use of AZT, d4T, or 3TC hardly budged MI risk, but cumulative abacavir or ddI use did make MI more likely (table, model 1). In a statistical analysis including cumulative and recent use (model 2), recent abacavir use almost doubled MI risk but cumulative use did not. And in a model melding cumulative, recent, and past use (model 3), cumulative abacavir use did not affect MI risk, but again recent use almost doubled MI chances and past abacavir use raised MI odds 29%. For ddI, cumulative or past use did not affect risk in models 2 or 3, but recent use raised MI risk about 50%.
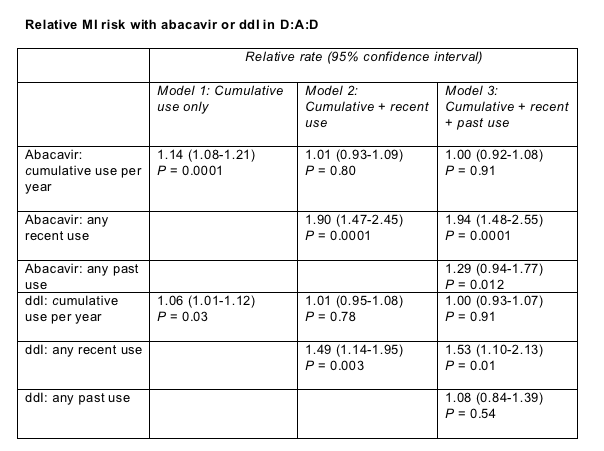
Higher MI odds with abacavir and ddI held true regardless of how long people used these nucleosides and after statistical adjustment for viral load, CD4 count, out-of-line lipids, and other metabolic factors.
D:A:D statisticians considered that physicians may prescribe abacavir more often for people with a higher heart attack risk when starting antiretrovirals. So they tried to correct their analysis to compensate for this potential bias. But the heightened risk changed little after statistical adjustment for a panoply of standard heart risk factors--gender, age, body mass index, current or former smoking, personal or family heart disease history, diabetes, hypertension, bad lipids, or a moderate or high 10-year predicted risk by the Framingham score.
On top of that, the D:A:D team found that MI risk waned after people stopped abacavir. "Had the drugs merely served as surrogates for a high underlying cardiovascular risk," the D:A:D report says, "this increased risk would have been expected to remain even after discontinuation of the drugs."
Perhaps because these results seem likely to stir controversy, D:A:D investigators took the unusual step of offering a 4-page explanation with their data-packed poster. They drew a careful distinction between absolute MI risk in this population, which was low, and the higher relative risk with abacavir or ddI. Absolute risk equals the total number of patients (33,400) divided by the 517 heart attacks, which yields a 1.6% MI risk over the 5 years of follow-up. People taking abacavir had a 90% higher relative risk--in other words almost a doubled risk--of this low absolute MI risk. For someone with a low absolute risk of MI because they have few traditional risk factors, doubling that risk poses a much lower threat than it would for someone with a high absolute risk because they have several traditional risk factors.
D:A:D researchers also warned that they cannot rule out undiscovered biases against abacavir in this type of analysis. But they added that they "extensively investigated" the possibility of bias "and could not find any evidence of appreciable bias." If, indeed, abacavir or ddI regimens actually cause MI, they noted, "the unknown biological mechanism(s) appear reversible upon cessation of these drugs."
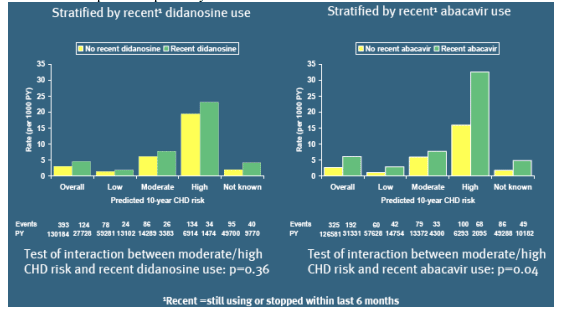
From Jules: They aso reported this table in the poster:
Rates of MI for recent use of ddI and of abacavir by predicted 10-year coronary heart disease (CHD) risk
In this table they report finding that patients with high risk for CHD had a much higher risk for MI as defined by the Framingham definition if they were taking abacavir than patients taking abacavir with moderate risk. Patients with moderate CHD risk had higher risk for MI compared to patients at low risk for CHD. The risk they reported for MI in patients with low predicted 10-year CHD risk by Framingham was by observation of this tabe about 2.5 per 1000 patient/years.
Reference
1. Sabin C, Worm S, Weber R, et al. Do thymidine analogues, abacavir, didanosine and lamivudine contribute to the risk of myocardial infarction? The D:A:D study. 15th Conference on Retroviruses and Opportunistic Infections. February 3-6, 2008. Boston. Abstract 957c.
2. D:A:D Study Group. Class of antiretroviral drugs and the risk of myocardial infarction. N Engl J Med. 2007;356:1723-1735.
|
| |
|
 |
 |
|
|