 |
 |
 |
| |
Effect of Tesamorelin (TH9507), a Growth Hormone-Releasing Factor (GRF) Analogue, on Visceral Adipose Tissue (VAT) in HIV-Infected Patients with Excess Abdominal Fat: Impact of Antiretroviral Therapy Regimen
|
| |
| |
Reported by Jules Levin
10th International Workshop on Adverse Drug Reactions and Lipodystrophy in HIV, in London, England Nov 6-8, 2008
Julian Falutz1, Jean-Claude Mamputu2, Christian Marsolais2, Diane Potvin2, Monika Zoltowska2, Dana Aeschliman2, Donald Kotler3, and Steven Grinspoon4
1Montreal General Hospital, McGill University Health Centre, Montreal, Quebec, Canada; 2Theratechnologies Inc., Montreal, Quebec, Canada; 3St. Luke's-Roosevelt Hospital Center, Columbia University College of Physicians and Surgeons, New York, NY, USA; 4Massachusetts General Hospital and Harvard Medical School, Boston, MA, USA
AUTHOR CONCLUSIONS
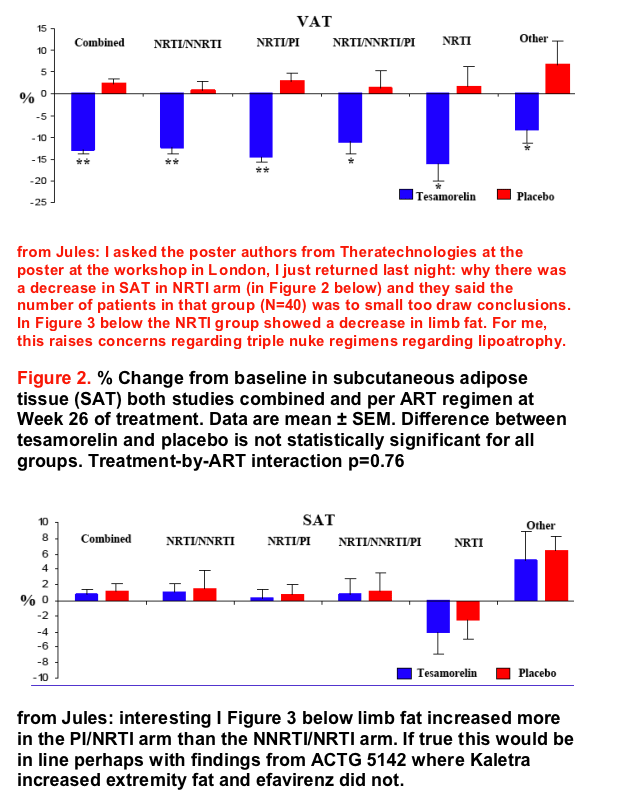
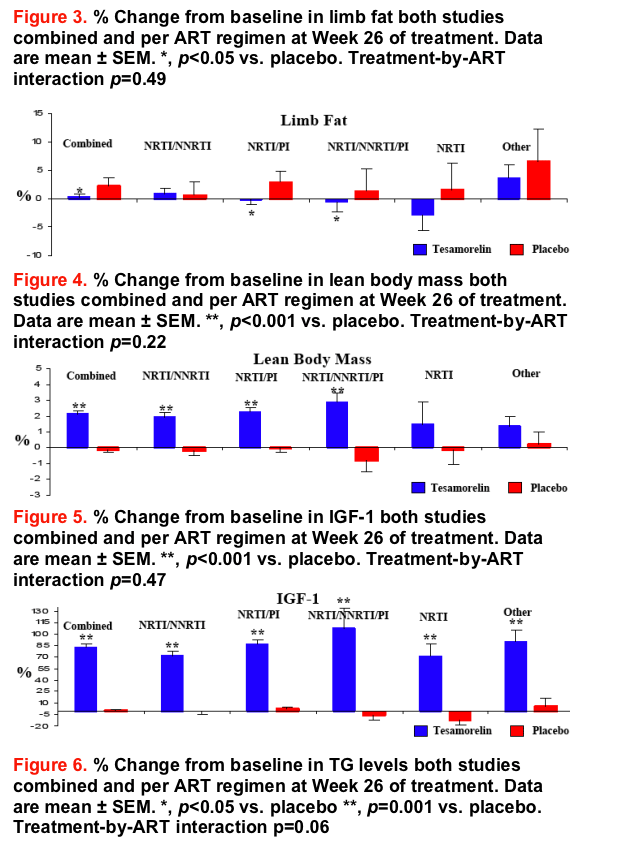
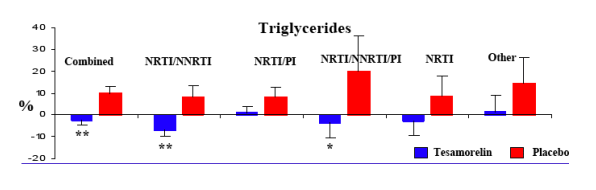
The results of this study indicate that regardless of the type of ART regimen, daily administration of a 2 mg dose of tesamorelin for 26 weeks significantly reduces VAT, without affecting SAT, in HIV-infected patients with excess abdominal fat on ART. Moreover, an improvement in triglycerides as well as an increase in lean body mass and IGF-1 are observed regardless of ART regimen.
ABSTRACT
Background: HIV-infected patients on antiretroviral therapy (ART) often show increased visceral adipose tissue (VAT), a known cardiovascular risk factor. Previous data showed that administration of a daily 2 mg dose of tesamorelin, a Growth Hormone-Releasing Factor analogue, to HIV-infected patients with excess abdominal fat for 26 weeks resulted in a significant decrease in VAT over placebo and improvements in lipids as well as patient-reported outcomes related to body image. Here we report the percent change from baseline to Week 26 in VAT per type of ART regimen for the combined Phase 3 studies of tesamorelin in HIV-infected patients with lipohypertrophy.
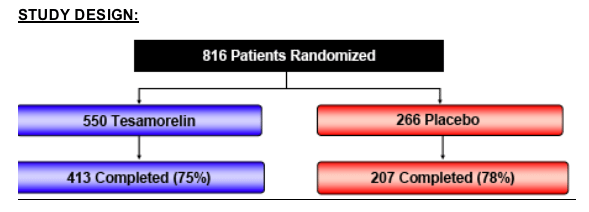
Methods: 816 HIV-infected patients with abdominal fat accumulation in the context of HIV treatment were randomized in two independent Phase 3 studies to receive daily subcutaneous injection of either tesamorelin 2 mg (n=550) or placebo (n=266) for 26 weeks. The primary endpoint of these studies was the percent change from baseline to Week 26 in VAT, as assessed by computerized tomography (CT) scan, using the intent-to-treat population with the last observation carried forward for patients not completing the study.
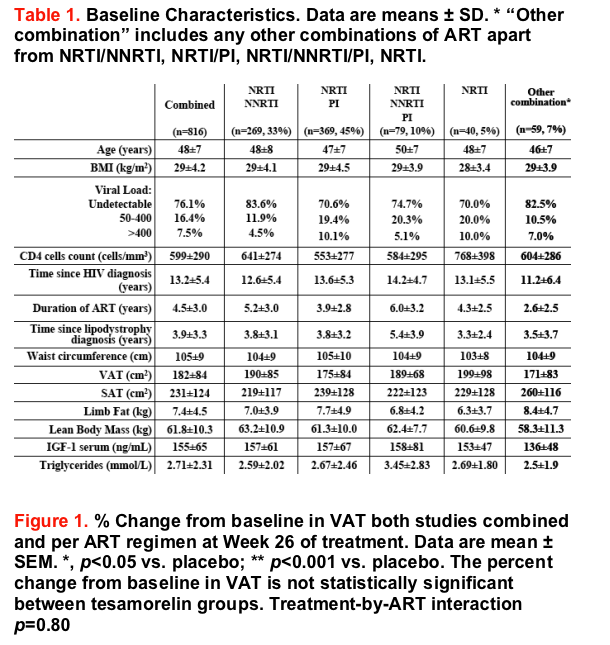
Results: Mean age was 48±7 (SD) years, waist circumference 105±9 cm, BMI 29±4 kg/m2, CD4 cell count 599±290 cells/mm3, and 76% of patients had undetectable HIV viral load at baseline. Mean time since initial diagnosis of HIV infection was 13.2 years. Mean duration on ART was 4.5 years, while mean time since initial diagnosis of lipodystrophy syndrome was 3.9 years.
Patients received the following types of ART regimen during the studies: "NRTI/NNRTI" (33%), "NRTI/PI" (45%), "NRTI/NNRTI/PI" (10%), "NRTIs alone" (5%), and "Other" (7%).
At baseline, VAT values were 190, 175, 189, 199, and 171 cm2 for the "NRTI/NNRTI", "NRTI/PI", "NRTI/NNRTI/PI", "NRTIs alone", and "Other" groups, respectively.
Overall, VAT decreased from baseline by 13% in tesamorelin-treated patients after 26 weeks of treatment (p<0.001 vs. placebo). The mean percent changes from baseline to Week 26 in VAT were -12.3, -14.5, -10.9, -16.0, and -8.2% for tesamorelin-treated patients in the groups "NRTI/NNRTI", "NRTI/PI", "NRTI/NNRTI/PI", "NRTIs alone" and "Other", respectively, whereas the mean percent changes from baseline in VAT were 0.6, 2.9, 1.4, 1.6, and 6.6% for placebo-treated patients in the same groups (p<0.05 for tesamorelin vs. placebo for each type of ART regimen).
Conclusion: The results of this study indicate that daily administration of a 2 mg dose of tesamorelin is useful for reducing VAT in HIV- infected patients with excess abdominal fat on ART, regardless of type of ART regimen.
NRTI: Nucleoside Reverse Transcriptase Inhibitor; NNRTI: Non-Nucleoside Reverse Transcriptase Inhibitor; PI: Protease Inhibitor
INTRODUCTION
A significant number of HIV patients on ART develop excess abdominal fat and metabolic abnormalities including dyslipidemia and insulin resistance (1). Accumulating evidence indicates that these patients are at increased risk of cardiovascular diseases (2, 3, 4).
Body changes have a negative impact on patient-reported outcomes (PRO), including body image, and may thereby decrease adherence to ART (5).
Results from two Phase 3 studies in HIV-infected patients with lipohypertrophy demonstrate that treatment with tesamorelin (TH9507), a stabilized analogue of growth hormone-releasing factor (GRF) or growth hormone-releasing hormone (GHRH), for 26 weeks led to a significant reduction in VAT, improvement in triglycerides as well as PRO related to body image (6, 7).
Here we report the percent change from baseline to Week 26 in VAT and other efficacy endpoints per type of ART regimen for the combined Phase 3 studies.
SUBJECTS AND METHODS
STUDY DESIGN:
OBJECTIVES:
Phase 3 trials
Primary: To demonstrate a reduction in VAT from baseline vs. placebo by computerized tomographic (CT) scan at L4-L5.
Secondary: To demonstrate the efficacy of tesamorelin vs. placebo in: (i) patient reported outcomes related to body image, (ii) total cholesterol/HDL-cholesterol ratio and triglycerides levels, and (iii) serum IGF-1 levels. To characterize the safety profile of tesamorelin (glucose, insulin, adverse events and others).
Analyses of the combined studies
The objective of the current combined analyses was to evaluate tesamorelin efficacy by ART regimen.
STATISTICAL ANALYSES:
Arithmetic means and Standard Deviations (SD) were used in Table 1 and arithmetic means and Standard Error to the Means (SEM) were used in all graphs. Quoted p-values for VAT are from linear models where the response is the natural log of (VAT at Week 26 over VAT at baseline) and the explanatory variables are the treatment, natural log of VAT at baseline, study, ART category and the interaction termtreatment-by-ART category. The model presenting the combined results excludes the treatment-by-ART category interaction and keeps only the main effects. For the other parameters, the statistical models used were the change from Baseline to Week 26 as the dependant variable with the same explanatory variables as described above, but with baseline value as covariate. No adjustments for multiplicity of tests were performed. A type I error of 5% was used for all comparisons.
RESULTS
|
| |
|
 |
 |
|
|