 |
 |
 |
| |
FACTORS PREDICTIVE OF RELAPSE IN GENOTYPE 2 AND 3 PTS TREATED FOR 12 WEEKS WITH PEGIFN ALFA 2B AND WEIGHT BASED RIBAVIRIN COMBINATION
|
| |
| |
Reported by Jules Levin
43rd EASL Conference, April 23-27, 2008, Milan, Italy
A. Mangia1, N. Minerva2, D. Bacca3, R. Cozzolongo1, E. Agostinacchio4, F. Sogari5, F. Vinelli6, G. Scotto6, G.L. Ricci7, M. Romano7, V. Carretta9, D. Petruzzellis1, V. Piazzolla1, F. Spirito1, A. Andriulli1
1 Gastroenterology IRCCS, San Giovanno Rotondo, Italy;
2 Internal Medicine Hospital Canosa, Canosa, Italy;
3 Internal Medicine Hospital Casarano, Casarano, Italy;
4 Internal Medicine Hospital, Italy;
5 Internal Medicine, Italy;
6 Gastroenterology And Infectious Disease Ospedali Riuniti Foggia, Italy;
7 Clinical Science Department, Italy;
8 Liver Unit, Italy;
9 Internal Medicine Hospital Venosa, Italy
Mangia and others have reported that 12 weeks of peg-RBV therapy is successful therapy in patients with genotype 2 or 3 with HCV monoinfection. She reported in the NEJM that 85% could achieve SVR after viral undetectability (RVR) at week 4; in her study the relapse rate was 10%. Relapse rate represents the main concern about short-term treatment as it has been reported higher after short treatment than after 24 weeks. The purpose of this trial was to delineate baseline features which may help identify RVR patients most likely to relapse after a short course of treatment. The secondary objective of this study is to assess SVR rates after re-treating with standard duration patients who relapsed after an initial course of antiviral therapy.
In her study in Italy all consecutive patients with HCV 2 and 3 genotype are currently enrolled in an ongoing program of treatment according to RVR:
--patients with RVR get 12 weeks therapy
--patients without RVR get 24 weeks.
The Multicenter Italian study from "Al-Liver Group"; enrollment: Jan 2005 - June 2007; 11 Italian centers; 970 new patients; Treatment: Peg-IFN alfa-2b 1.5 ug/kg plus RBV 1000-1200 mg/day.
970 patients evaluated; 718 enrolled:
222 without RVR (31%)
496 with RVR (68%): 17 withdrew by wk 12; 3 HCV RA+ at week 12
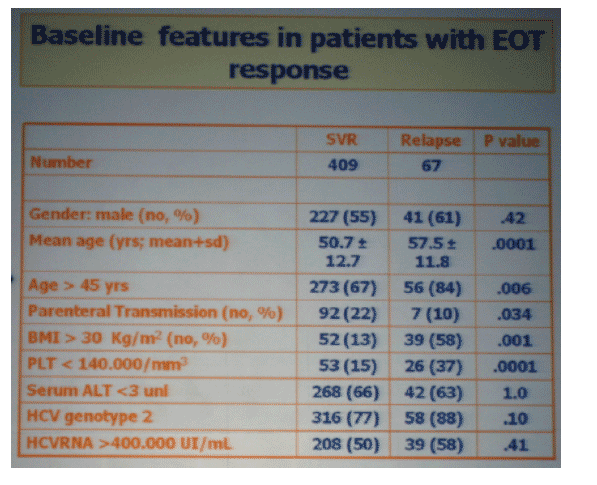
476 completed: 409 with SVR (85^), 67 relapsed (14%)
TREATMENT RESPONSE ACCORDING TO GENOTYPE
561 HCV-2: 69% achieved RVR (n=387); EOT response 97%; SVR 82%
157 HCV-3: 69% achieved RVR (n=107); EOT response 94%; SVR 84%
AUTHOR SUMMARY
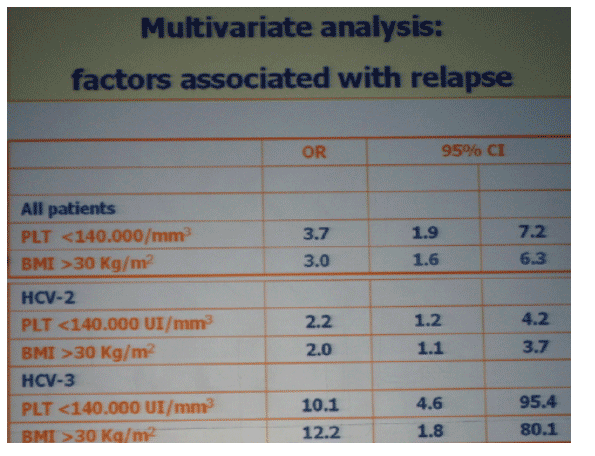
Patients with BMI > 30 kg/M2 and/or Platelet counts <140,000 mm3 have higher likelihood of relapse after short treatment.
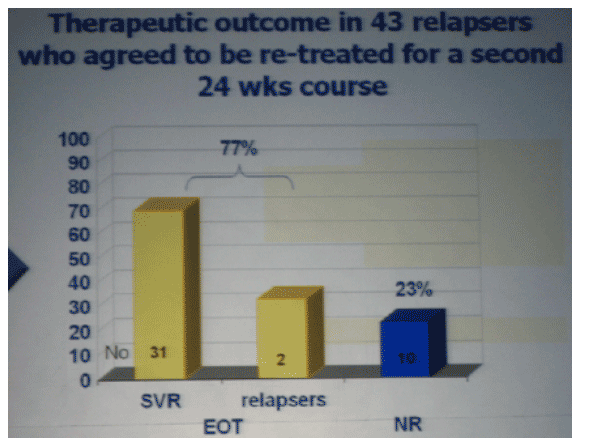
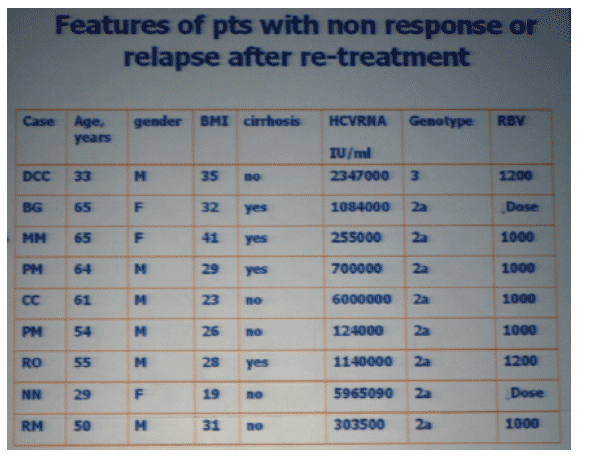
Re-treatment of patients who relapsed allows 72% rate of SVR.
A limited number of patients (4%) with HCV 2 and 3 genotype after RVR do not respond to current schedules of treatment.
Almost all HCV 2 and 3 genotype patients who achieve RVR may be cured with only 12 weeks of Peg-IFN alfa-2b and weight-based doses of BV.
We do not recommend short treatment only in HCV 2 and 3 genotype patients with BMI >30 kg/m2 and features of advanced liver disease.
This is program abstract. A key number that was different in the final presentation by Mangia was that BMI > 30 kg/m2 is the level of BMI that is predictive that a short course of peg/RBV therapy may not be effective.
Background and aims: The relatively high rate of relapse after a 12-16 weeks course of treatment is the main concern on shortening treatment duration for HCV genotype 2 and 3 patients. To delineate predictors of relapse after short treatment duration in patients with HCV clearance at treatment week 4 (RVR) and to report sustained virological response (SVR) rates after a second 24 weeks course in RVR patients who experienced a relapse.
Methods: After RVR, naive patients were treated with PegIFN alpha 2b (1.5 µg/Kg) and ribavirin (1000-1200 mg) for 12 weeks. Patients who cleared the virus at the end of treatment (EOT) but had a detectable HCVRNA at the end of 24 weeks follow up underwent re-treatment with the same dosages of both PegIFN alfa 2b and ribavirin but for an extended duration of 24 weeks. Logistic regression analysis was applied to delineate baseline features of SVR to short treatment course by using age, gender, route of transmission, BMI, serum ALT, HCV genotypes, serum HCVRNA levels and platelets counts as covariates.
Results: Of 790 patients with genotype 2 and 3 who consecutively underwent treatment from January 2005 at our centers, 496 (mean age 51.6 yrs, M/F 3:2) tested HCVRNA negative at treatment week 4 (63%): EOT response rate was observed in 96% of them and SVR in 82%. Of patients with EOT response, 14% relapsed off-therapy.
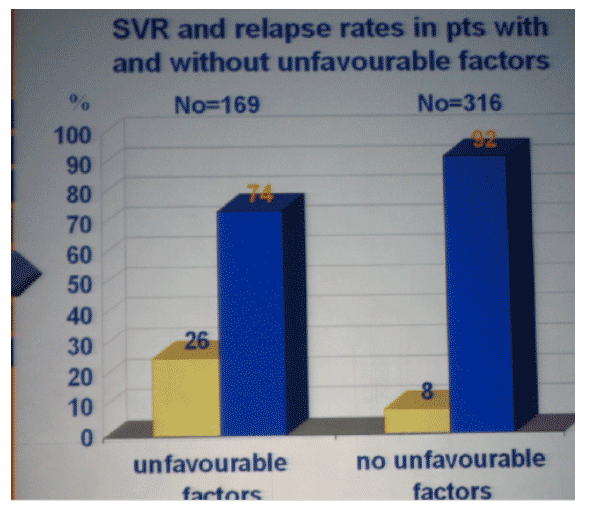
Factors predictive of relapse were age older than 45 yrs (p=0.004), BMI > 27 Kg/m2 (p=0.0001), platelets counts <140.000 mm3 (p=0.0001). At logistic regression analysis, only platelets counts (p=0.001, OR 2.5, 95% CI 1.49-4.20) and BMI (p=0.036, OR1.7, 95% CI 1.03-2.7) were independently associated with SVR. In the subset of patients without these unfavourable predictors, relapse rate was only 7%. Forty three of 67 patients with relapse agreed to be re-treated. SVR was achieved in 47 (70%)of them.
Conclusions: Almost all genotype 2 and 3 patients who achieves RVR maybe cured with only 12 weeks of PegIFN alfa 2b and weight based ribavirin. Standard (24 weeks) treatment duration should be reserved only to patients with PLT < 140.000 mm3 and/or BMI > 27 Kg/m2.
|
| |
|
 |
 |
|
|