 |
 |
 |
| |
Safety of the HCV Protease Inhibitor TMC435350 in Healthy Volunteers and Safety and Activity in Chronic Hepatitis C Infected Individuals: A Phase I Study
|
| |
| |
Reported by Jules Levin
43rd EASL Conference, April 23-27, 2008, Milan, Italy
H. Reesink,1 R. Verloes,2 K. Abou Farha,3 A. Van Vliet,3
C. Weegink,1 G. van Ôt Klooster,2 F. Aharchi,2 K. Marien,2
P. Van Remoortere,2 H. de Kock,2 F. Broeckaert,2 G. Fanning,2
P. Meyvisch,2 E. Van Beirendonck,2 K. Simmen2
1Academic Medical Center, Department of Gastroenterology and Hepatology, Amsterdam, The Netherlands; 2Tibotec BVBA, Mechelen, Belgium and Tibotec Pharmaceuticals Ltd., Ireland; 3PRA International, Zuidlaren, The Netherlands
Author Conclusions and future directions
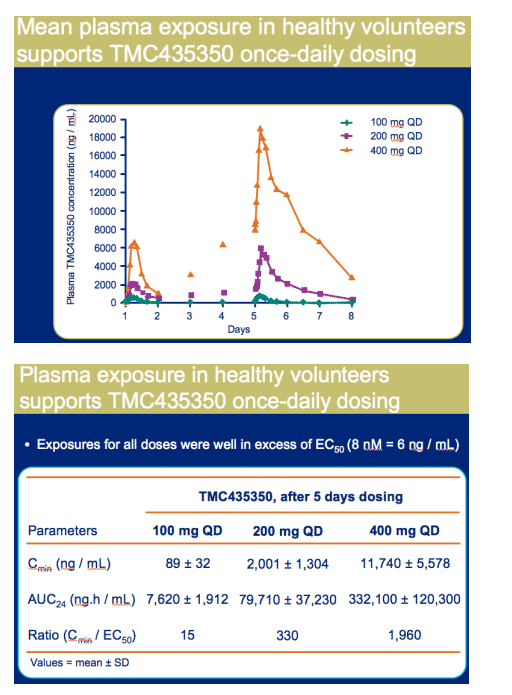
In healthy volunteers TMC435350 was well tolerated and PK supports QD dosing
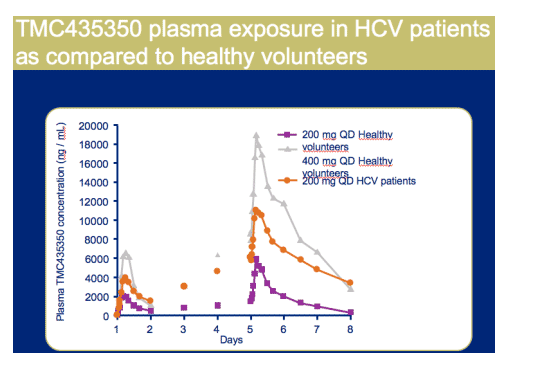
In HCV treatment experienced patients, 5 days QD TMC43530 dosing was well tolerated
Once daily dosing resulted in robust antiviral activity in all patients with a maximal median decrease in HCV RNA of 3.9 Log10, at day 6
TMC435350is currently in a Phase IIA 4-week comination trial with Peg-IFN/ribavirin HCV genotype 1-infected patients.
TMC435350: a novel and potent HCV
protease inhibitor
In vitro profile
Reversible NS3/4A protease inhibitor (non-covalent)
Molecular weight = 750
EC50 = 8 nM in genotype 1 replicon
Ki <1 nM for genotype 1 enzymes
Synergy with IFN_ and polymerase inhibitors; additive with ribavirin
In vivo preclinical profile
High oral bioavailability: 45-80%
High liver to plasma ratio: >30
No cardiovascular alert
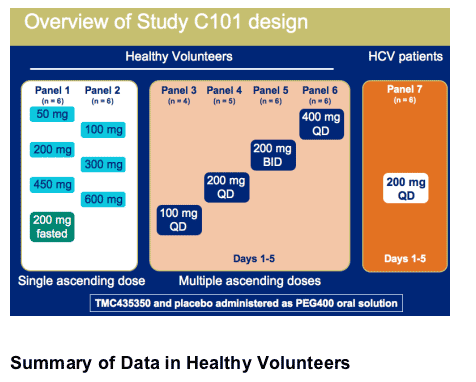
Objectives of TMC435350 Phase I study C101
Evaluate the safety and pharmacokinetic (PK) profile of single and multiple ascending doses of TMC435350 in healthy volunteers1,2
Evaluate safety, PK and antiviral activity of TMC435350 in HCV-infected patients
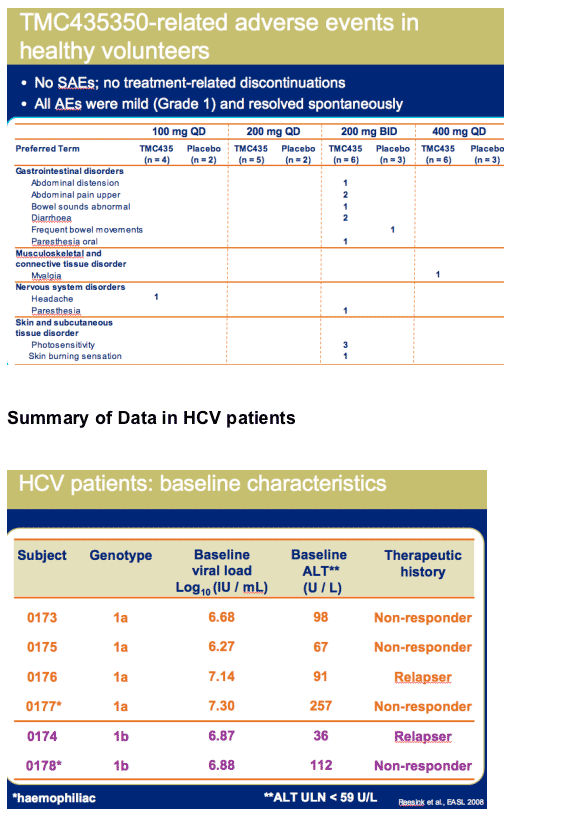
HCV patients: minimal adverse events reported
No SAEs were observed
All adverse events were mild (Grade 1)
--Headache (n=3)
--Fatigue (n=2)
--Other AEs were observed in no more than 1 patient
No treatment-related discontinuations
No clinically relevant changes in vital signs, ECG and echocardiography parameters
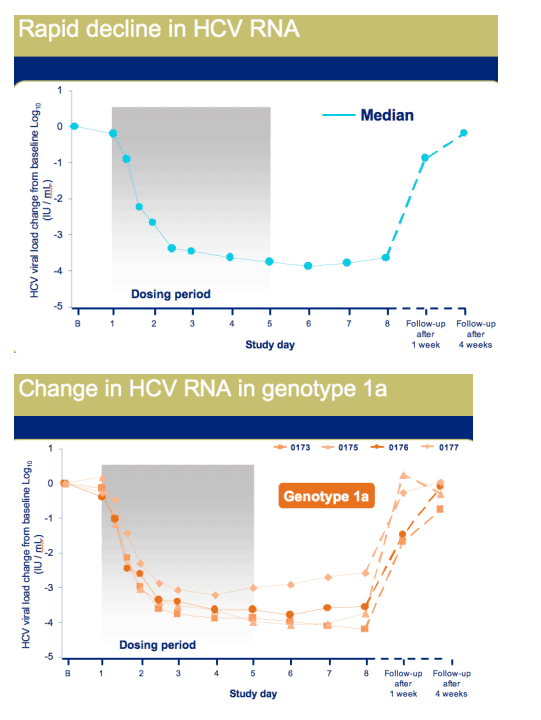
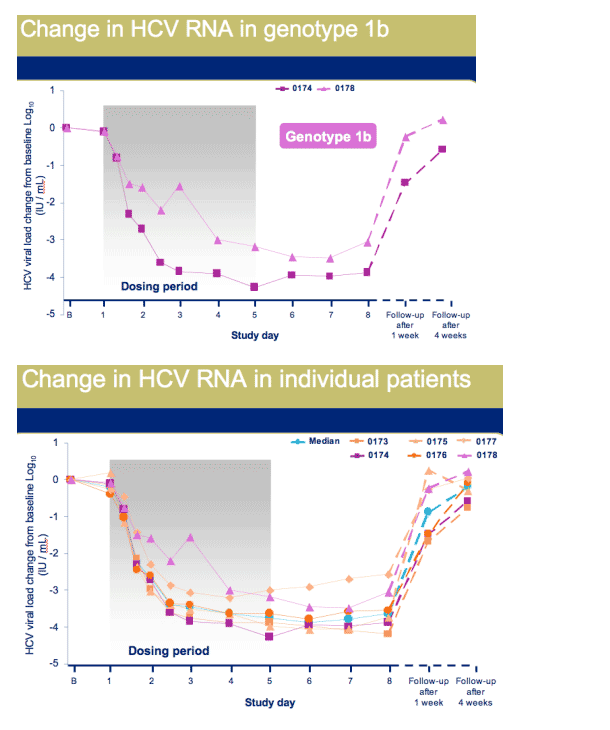
|
| |
|
 |
 |
|
|