 |
 |
 |
| |
Interim Results from HCV SPRINT-1: RVR/EVR from Phase 2 Study of Boceprevir Plus Peginterferon alfa-2b/Ribavirin in Treatment-Naive Subjects with Genotype-1 CHC
|
| |
| |
Reported by Jules Levin
43rd EASL
April 23-27, 2008
Milan, Italy
P. Kwo1, E. Lawitz2, J. McCone3, E. Schiff4, J. Vierling5, D. Pound6, M. Davis7, J. Galati8, S. Gordon9, N. Ravendhran10, L Rossaro11, F. Anderson12, I. Jacobson13, R. Rubin14, P. Mukhopadhyay15, E. Chaudhri15, L. Pedicone15, J. Albrecht15
1 Indiana University School Of Medicine, Indianapolis, Indiana, USA;
2 Alamo Medical Research, San Antonio, Texas, USA;
3 Mount Vernon Endoscopy Center, Alexandria, Virginia, USA;
4 University of Miami Center for Liver Diseases, Miami, Florida, USA;
5 Baylor College of Medicine, Houston, Texas, USA;
6 Indianapolis Gastroenterology Research Foundation, Indianapolis, Indiana, USA;
7 South Florida Center of Gastroenterology, Wellington, Florida, USA;
8 Liver Specialists of Texas, Houston, Texas, USA;
9 Henry Ford Hospital, Detroit, Michigan, USA;
10 Digestive Disease Associates, Baltimore, MD, 11_Univ Of California-Davis, Sacramento, CA, USA ,12_The Liver And Intestinal Research Center, Vancouver, Canada, 13_Weill Medical College Of Cornell Univ, New York NY, 14_Digestive Healthcare Of Georgia,, Atlanta, GA, 15_schering-Plough Research In
Summary of Interim Results
Boceprevir with P/R for 28 weeks
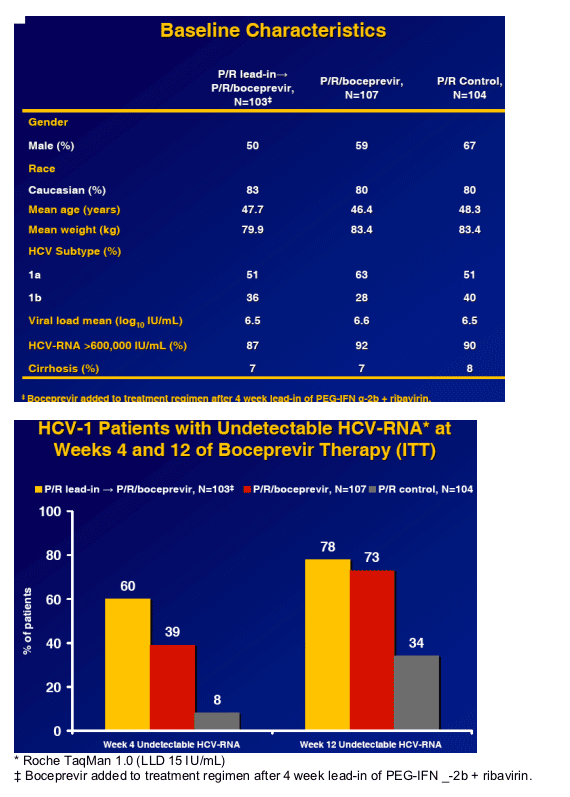
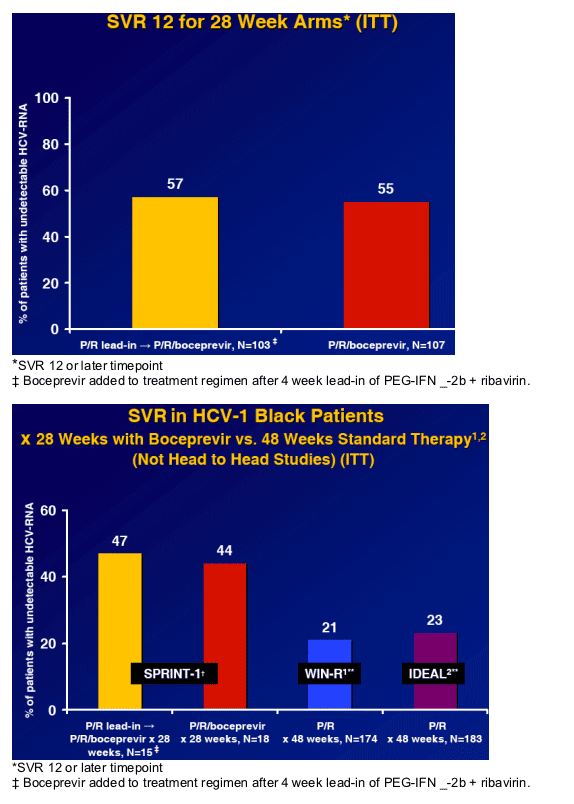
Greater SVR than P/R alone for 48 weeks in HCV-1 naive patients.1-4 (56% 12week SVR in whites and 45% in Blacks) [patients in study receive Boceprvir for entire length of study while in Telaprevir studies patients receive protease for limited shorter duration]
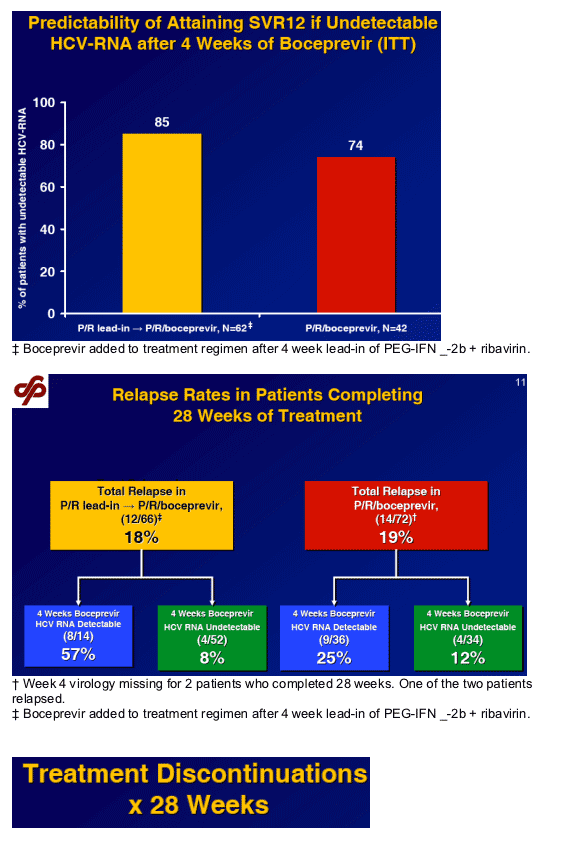
Predictability of response after 4 weeks of boceprevir
Up to 85% with undetectable HCV-RNA achieve SVR 12.
Higher relapse rate in subjects with detectable HCV-RNA after 4
weeks boceprevir.
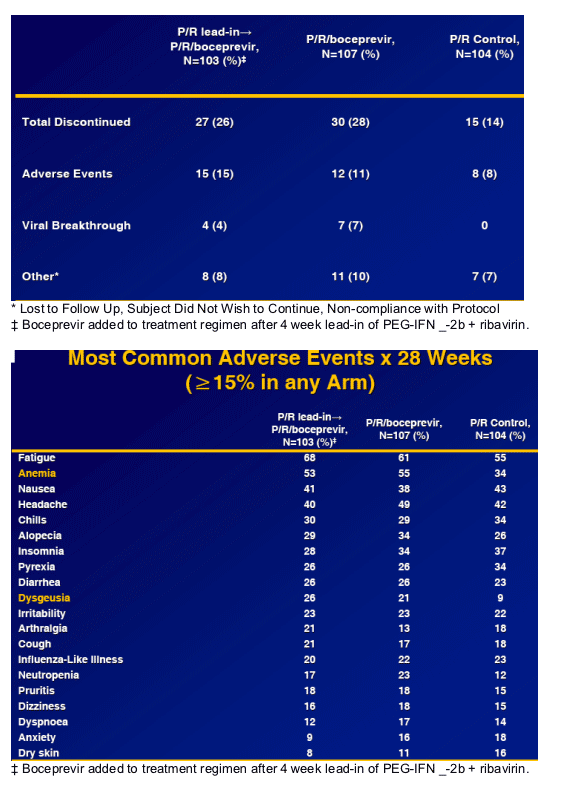
Safety
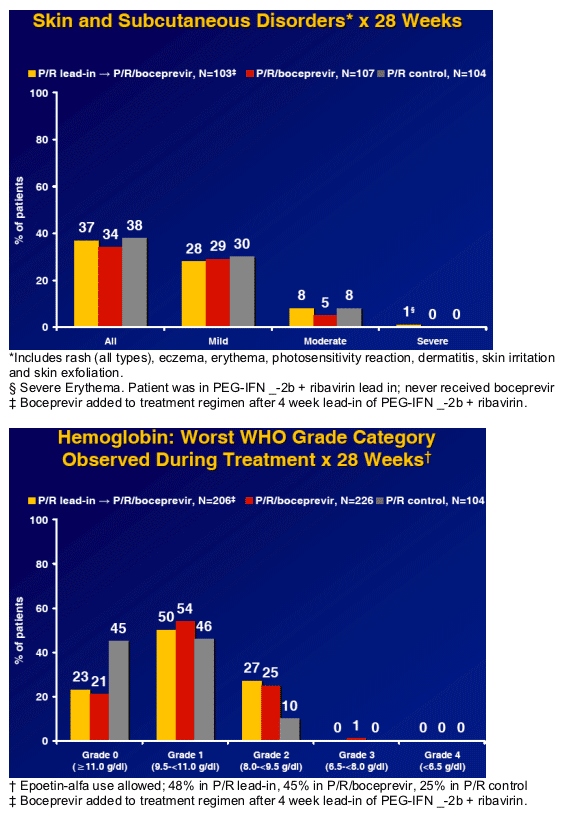
Boceprevir containing regimens appear to be well-tolerated.
Incidence of rash and pruritis comparable to Control.
No boceprevir-defining toxicity responsible for treatment discontinuation.
1 Fried M, et al. N Engl J Med 2002;347:975-82. 2 Manns M, et al. Lancet 2001; 358: 958Đ65.
3 Sulkowski M, et al. Abstract #LB 2, EASL 2008. 4 Jacobson I, et al. Hepatology 2007;46:982-990.
Rationale for New Therapies
Sustained virologic response with PEG-IFN _ + ribavirin x 48 weeks in genotype 1 treatment naive patients is ~40%1-3
Sustained virologic response with PEG-IFN _-2b + ribavirin x 48 weeks in genotype 1 treatment naive, black patients is ~20%4
1 Fried M, et al. N Engl J Med 2002;347:975-82.
2 Manns M, et al. Lancet 2001; 358: 958Đ65.
3 Sulkowski M, et al. Abstract LB #D2, EASL 2008.
4 Jacobson I, et al. Hepatology 2007;46:982-990.
Aims of the Study
To evaluate the most effective treatment strategy in HCV-1 treatment naive patients
Duration of therapy
28 weeks vs. 48 weeks
Lead-in strategy
with Peginterferon alfa-2b + ribavirin followed
by boceprevir 800 mg TID vs. Triple therapy from Day 1
Standard vs low dose ribavirin
800-1400 mg/day vs 400-1000 mg/day
To evaluate the predictability of Week 4 and 12 HCV-RNA undetectability on SVR

|
| |
|
 |
 |
|
|