 |
 |
 |
| |
DEVELOPMENT OF NOVEL HYPERGLYGOSYLATED TYPE 1 INTERFERONS: A STRATEGY TO IMPROVE PK PERFORMANCE WITHOUT LOSS OF BIOLOGICAL POTENCY
|
| |
| |
Reported by Jules Levin
43rd EASL
April 23-27, 2008
Milan, Italy
There are several alternative interferons in development including this one and Albuferon which is now in phase III.
AUTHOR CONCLUSIONS
⇒ A need still exists for IFN-based products with novel biological properties
⇒ Addition of Glycosylation Sites
- Improved PK
- Maintained Biological Potency
- May Address HCV NR
- Additional applications in HBV and possibly HIV
⇒ Continuing optimization of glycosylation
- Optimization cell culture conditions will improve carbohydrate incorporation
- More glycosylation sites can be added if needed
⇒ Further Study is Warranted


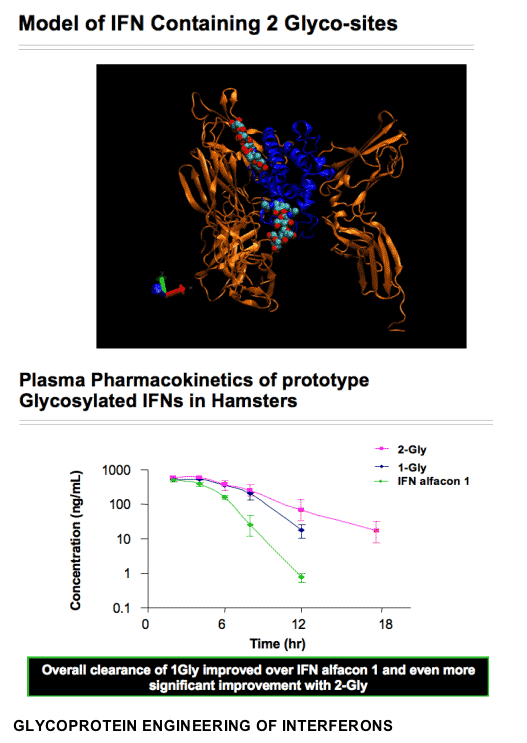
GLYCOPROTEIN ENGINEERING OF INTERFERONS
This statement is from Alios BioPharma
Interferon (IFN) alpha based products are approved worldwide for the treatment of various malignancies as well as viral diseases such as chronic hepatitis B hepatitis C, and human papillomavirus.
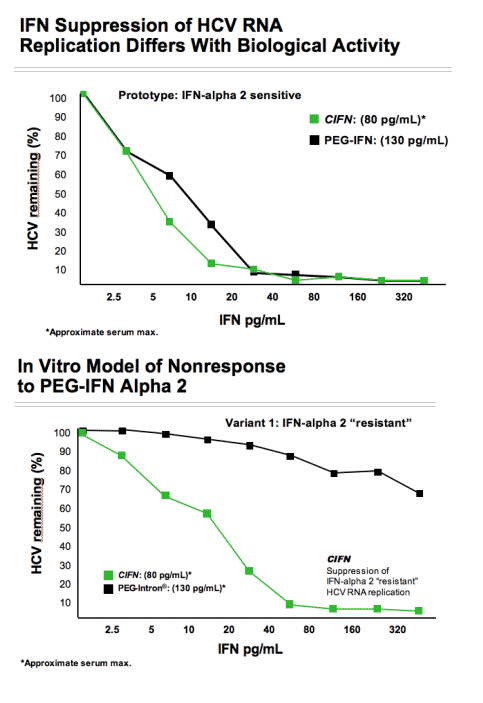
Although the clinical utility of both alpha and beta IFN has been clearly demonstrated, there is a need for new molecules that address the disadvantages of pegylated-IFN (PEG-IFN) and albuferon (interferon alpha 2b/albumin fusion protein) products which have improved pharmacokinetic (PK) characteristics but with significant loss of biological potency. The need for new IFNs is most urgent for the treatment of chronic hepatitis C where recent data show that the addition of direct antiviral agents to treatment regimes still requires the use of PEG-IFN and that patients who are null-responders (NR) to PEG-IFN + ribavirin are unlikely to benefit from retreatment with PEG-IFN + ribavirin and a direct antiviral agent. This lack of response may in part be due to the fact that the addition of the PEG moiety to the IFN molecule reduces the biological potency of the resultant molecule by creating steric hindrance of the receptor ligand interaction. These patients may however benefit from a more potent IFN included in the drug cocktail.
Alios founding scientists have discovered, and hold exclusive worldwide rights to, a method enabling the creation of highly potent IFN protein therapeutics with improved PK characteristics without loss of biological potency resulting in molecules with the potential for improved therapeutic indices. This proprietary technology does not require post-fermentation chemical modification of the IFN, a process that dramatically reduces potency and increases cost of goods of existing PEG-IFN products. The Alios technology involves introduction of glycosylation sites into the IFN protein by modification of the primary amino acid sequence of the IFN molecules thus increasing the molecular weight and hydro-dynamic radius of the protein resulting in improved PK characteristics with no loss of biological potency. It is important to note that the glycosylation sites that can be engineered into one IFN molecule can also be engineered into additional IFN species. Thus, the Company is applying this technology to several IFN alpha and beta species with the goal of developing individual products optimized for the treatment of: viral diseases, cancer and MS. The prototype molecule under development is called Glycoferon¨.
Glycoferon
The first product the Company is developing an optimized hyperglycosylated consensus interferon (Glycoferonń) as a broad spectrum antiviral for treatment of HCV, HBV, HIV, avian influenza virus and other viruses. The non-glycosylated form of consensus interferon is currently being sold by Three Rivers Pharmaceuticals under the trade name Infergen and generated approximately $50 million in annual revenues during 2007. Consensus Interferon has also demonstrated clinical efficacy in patients with SARS in a clinical trial that was completed during the 2003 Toronto SAR outbreak. While Infergen is more potent than either Pegasys or PegIntron, the two leading interferon therapies, each generate revenues in excess of $1 billion per year, InfergenŐs three times per week and daily dosing schedule puts it a commercial disadvantage to competing therapies. The Company believes that Glycoferon will have equivalent if not improved dosing over these leading therapies (at least once per week dosing) with superior potency.
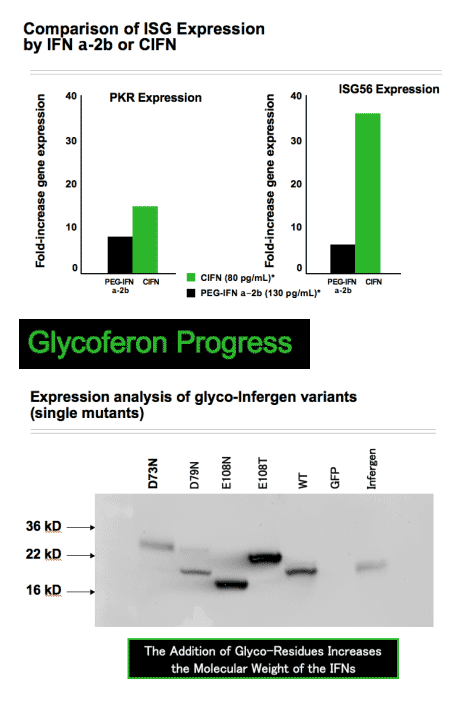
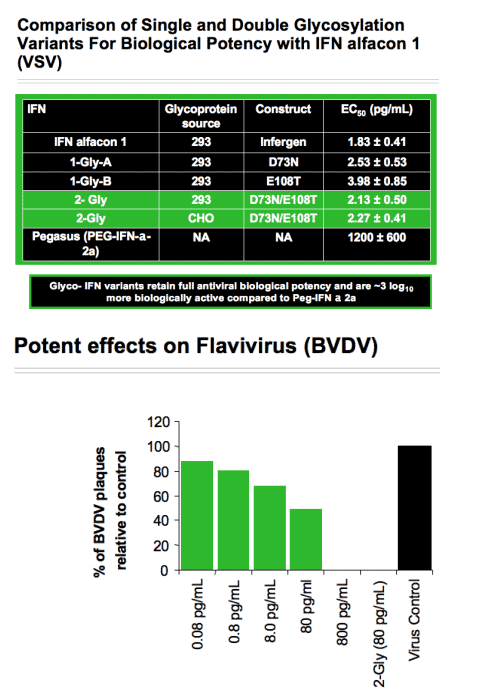
To produce the Glycoferon candidates, Alios scientists took the DNA encoding sequence for consensus IFN and made amino acid modifications to encode for several glycosylation sites throughout the protein. Two sites were identified that were quantitatively modified by addition of carbohydrate moieties. A single variant carrying both sites was quantitatively modified at both sites. Glycosylation substantially increased molecular weight with no resultant loss of biological potency. In cell culture models, consensus IFN demonstrates increased potency when compared to naturally occurring type 1 IFNs. Comparisons of the antiviral potency of glycol-variants with the parent non-glycosylated consensus IFN and PEG-IFN alfa 2a using the VSV on A549 cells are shown in the table below:
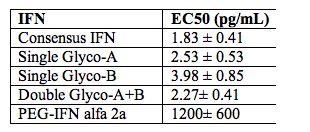
Statistical analysis of the EC50 values obtained demonstrated that all glycol-variants retain similar biological potency when compared to the consensus IFN molecule and all are approximately 3 logs10 more potent when compared to PEG-IFN alfa 2a (P<0.001 for all observation). Preliminary assessment of the pharmacokinetic characteristics of the glycosylated variants demonstrates an approximate 4-fold improvement in plasma exposure for AUC and a significant improvement in serum half-life. These data indicate that hyperglycosylation is a viable strategy to create a novel type 1 IFN with improved PK characteristics without subsequent loss in biological potency.
Based on the stage of development for Glycoferon, the Company estimates the time to entering the clinical for the treatment of chronic hepatitis C is approximately 18 months. It is important to note that clinical development in this setting is well established for IFN-based therapeutics and early clinical trials can demonstrate the utility of drug candidates. Due to these factors the company plans to move rapidly into a population of chronic Hepatitis C patients who are PEG-IFN plus ribavirin NR to demonstrate the potential for viral kinetic effects in these patients who previously failed to have log10 reductions of HCV RNA following PEG-IFN + ribavirin combination therapy. Demonstration of significant viral kinetic reductions in this patient population would be a value creating event for the Glycoferon product as early viral load reductions in chronic hepatitis C patients are predictive of sustained virological responses (SVR).
|
| |
|
 |
 |
|
|