 |
 |
 |
| |
Estimating the Likelihood of Sustained Virological Response in Chronic Hepatitis C (CHC) Therapy
|
| |
| |
Reported by Jules Levin
43rd EASL
April 2008
Milan, Italy
Mauss S1, Hueppe D2, John C3, Goelz J4, Meyer U5, Heyne R6, Moeller B6, Link R7, Teuber G8, Sworszt S9, Herrmann A10,
Spelter M11, Wollschlaeger A12, Steffens H5, Baumgarten A13, Simon K-G14, Dikopoulos N15, Witthoeft T16
1Center for HIV and Hepatogastroenterology, Duesseldorf; 2Center of Gastroenterology, Herne; 3Center of Gastroenterology, Berlin ; 4Practice Centre Kaiserdamm, Berlin; 5Center of Internal Medicine, Berlin ; 6Center of Gastroenterology and Livercenter, Berlin; 7Internal Medicine, St. Josef Clinic, Offenburg; 8Johann Wolfgang Goethe University, Frankfurt/Main; 9General Practice, Gelsenkirchen; 10Friedrich Schiller University, Jena; 11Center of Gastroenterology, Wuppertal; 12Hospital Dresden Friedrichstadt, Dresden; 13General Practice, Berlin; 14Center of Gastroenterology, Leverkusen; 15Department for Internal Medicine I, University of Ulm, Ulm; 16Medical Department I, Div. of Gastroenterology, University Hospital Schleswig-Holstein Campus Luebeck, Luebeck; all Germany
This research was supported by Roche Pharma AG, Grenzach-Wyhlen, Germany.
INTRODUCTION
For patients and physicians the likelihood of a treatment success is one of the most relevant factors for a decision to start or defer therapy of chronic hepatitis C (CHC). For this decision only baseline variables are important.
The current evaluation is based on PRACTICE (Pegylated Interferons and Ribavirin: Analysis of CHC Treatment In Centres of Excellence), a nationwide retrospective observational study on routine clinical practice including centres with large expertise in the treatment of chronic hepatitis C (CHC).
OBJECTIVE
Aim of this analysis is to identify predictive factors for the success of treatment of cHC-patients under real life conditions.
METHODS
This evaluation is part of a large retrospective German multicentre, open-label observational study including anti-HCV positive adults with detectable HCV RNA. According to the nature of this study, all treatment decisions concerning peginterferon and ribavirin (selection, dosing and treatment duration) were at the discretion of the physician.
The data set includes patients who completed treatment with peginterferon alfa-2a or alfa-2b plus ribavirin. The data collection was performed via an e-CRF online via the internet.
The documented data should refl ect the clinical routine as intended by the physicians in charge. Therefore, the statistical analysis remains descriptive.
Treatment
Patients were treated either with:
- pegylated interferon alfa-2a (40KD) (Pegasys) mostly plus ribavirin (Copegus) or
- pegylated interferon alfa-2b (PegIntron) mostly plus ribavirin (Rebetol).
Matched Pairs
To achieve comparability regarding interferon therapy, patients were matched acc. to the following criteria:
- age difference ≦3 years,
- same HCV genotype (only main type),
- same category of viral load: LVL or HVL (cut-off: ≦400.000 IU/ml),
- BMI difference ≦2 kg/m2,
- same anamnesis of hepatitis C incl. sub-categories (monotherapy, IFN-RBV-combi na tion therapy, PEG-RBV-combination therapy, virological non-response, not adequate pretreatment),
- presence of opioid maintenance (yes / no),
- presence of HIV-coinfection (yes / no).
Baseline predictive factors
In order to identify predictive factors for the response to treatment, the following factors were estimated:
- gender,
- age categories: <30, 30-<40, 40-<50, 50-<60, 60 or more years,
- BMI: <22, 22-28, >28 kg/m2,
- HCV genotype: 1, 2, 3 or 4,
- HCV RNA: cut-off ≦400 000 IU/ml for High viral load (HVL) and Low viral load (LVL),
- ALT: cut-off ULN,
- GGT: cut-off ULN,
- bilirubin: cut-off ≦1 mg/dl,
- platelets (PLT): cut-off ≥150 000 /μl as surrogat marker for advanced fi brosis,
- opioid maintenance: absence vs. presence,
- treatment: PEG-IFN alfa-2a vs. -2b (plus ribavirin).
RESULTS
Patients
3470 patients treated with pegylated interferon plus ribavirin under real life conditions were documented between 2000 and 2007 from 23 large outpatient clinics.
Matched data sets were available for 1204 pairs of patients (N=1204 PEG-IFN alfa-2a; N=1204 PEG-IFN alfa-2b).
In order to evaluate only patients with chronic hepatitis C, patients with acute hepatitis C were excluded from the analysis.
The resulting CHC-data set included 1189 matched pairs (N=1189 PEG-IFN alfa-2a; N=1189 PEG-IFN alfa-2b).
The demographic data for the 2378 CHC-patients and the total group of 2408 patients are shown in Table 1.
The following analysis is based on matched pairs of 2378 CHC-patients.
Virological response
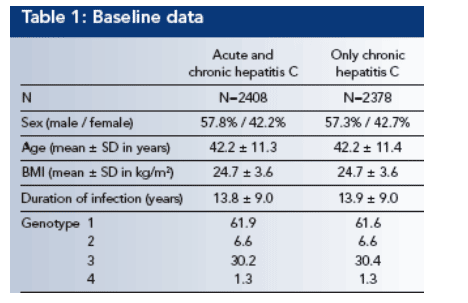
Over all, a Sustained virological response (SVR) was achieved in 1377 of 2378 patients (57.9%).
Univariate analyses
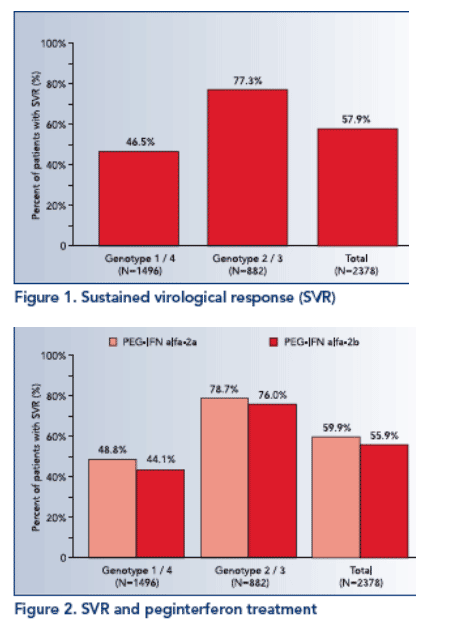
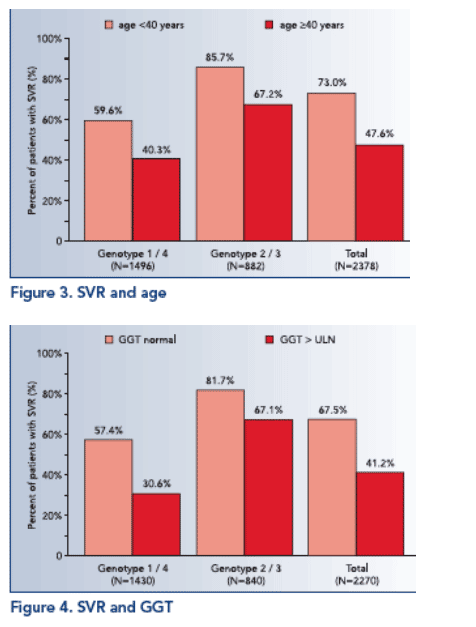
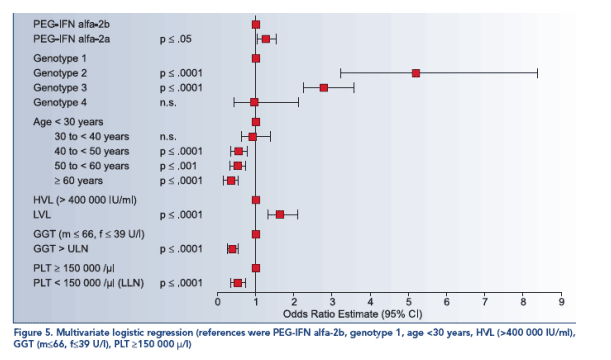
In univariate analyses SVR was significantly associated with genotype (p≦.0001; see Figure 1), pegylated interferon (p≦.05; see Figure 2), viral load (p≦.0001), age (p≦.0001; see Figure 3), BMI (p≦.0001), GGT (p≦.0001; see Figure 4) and platelets (p≦.0001).
No association was observed with gender, ALT and opioid maintenance.
Multivariate analysis
Positive predictive factors being significantly associated with SVR in multivariate logistic regression (MLR) are (see Figure 5):
- genotype 2 (p≦.0001; OR=5.214; 95% CI: 3.252-8.358),
- genotype 3 (p≦.0001; OR=2.871; 95% CI: 2.276-3.621),
- LVL (p≦.0001; OR=1.710; 95% CI: 1.397-2.093),
- PEG-IFN alfa-2a (p≦.05; OR=1.276; 95% CI: 1.051-1.550).
Negative predictive factors being significantly associated with SVR in multivariate logistic regression (MLR) are (see Figure 5):
- age 40-<50 yrs (p≦.0001; OR=.550; 95% CI: .397-.762),
- age 50-<60 yrs (p≦.001; OR=.521; 95% CI: .359-.756),
- age ≥60 yrs (p≦.0001; OR=.349; 95% CI: .220-.552),
- increased GGT (p≦.0001; OR=.428; 95% CI: .349-.526),
- decreased platelets (p≦.0001; OR=.571; 95% CI: .434-.752).
The BMI was not significantly associated with SVR (data not shown).
CONCLUSIONS
Genotype 2, genotype 3, low viral load and treatment with Peginterferon alfa-2a were identified as positive predictive factors for treatment success in this well controlled large cohort.
Higher age (≥ 40 years), elevated GGT and platelets <150 000 /μl were identifi ed as negative predictors for treatment success.
|
| |
|
 |
 |
|
|