 |
 |
 |
| |
RANDOMIZED DOUBLE BLIND PLACEBO-CONTROLLED TRIAL OF NITAZOXANIDE IN THE TREATMENT OF PATIENTS WITH CHRONIC HEPATITIS C GENOTYPE 4
|
| |
| |
Reported by Jules Levin
43rd EASL, April 208, Milan Italy
Jean-Francois Rossignol1,2, Samir M. Kabil3, Yehia El-Gohary4, Emmet B. Keefe1,2
1The Romark Institute for Medical Research, Tampa, FL, USA, 2Division of Gastroenterology and Hepatology, Department of Medicine, Stanford University School of Medicine, Palo Alto, California; 3Department of Hepatology, Gastroenterology and Infectious Diseases, University of Benha, Faculty of Medicine, Benha, Egypt; 4Department of Tropical Medicine and Infectious Diseases, University of Alexandria, Faculty of Medicine, Alexandria, Egypt
AUTHOR SUMMARY
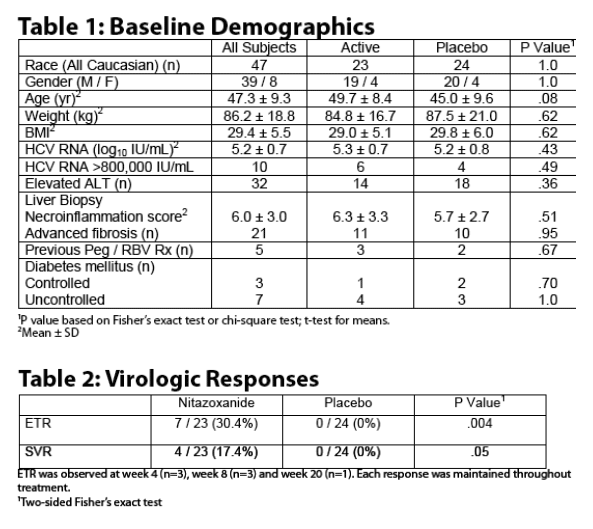
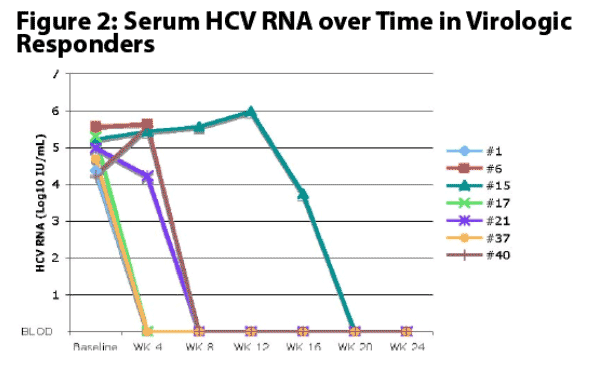
Treatment of patients with chronic hepatitis C genotype 4 with nitazoxanide 500 mg twice daily for 24 weeks was associated with an ETR in 7 of 23 patients (30.4%) and SVR in 4 of 23 patients (17.4%) compared to zero of 24 patients (0%) treated with placebo (P=0.0039).
Serum HCV RNA was the most significant independent predictor of virologic response (P=0.009). All virologic responders had baseline serum HCV RNA less than 385,000 IU/mL.
No safety concerns were identified during the study. Adverse events in the nitazoxanide and placebo treatment groups were similar.
AUTHOR CONCLUSIONS
Nitazoxanide monotherapy for 24 weeks is safe and effective in achieving SVR in some patients with chronic hepatitis C genotype 4, if baseline serum HCV RNA levels are low and there is no advanced liver disease or comorbidities, especially uncontrolled diabetes.
Second generation thiazolides with an improved pharmacokinetic profile may increase virologic response rates when administered as monotherapy for treatment of chronic hepatitis C.
INTRODUCTION
Nitazoxanide (Alinia, Romark Laboratories, Tampa, Florida) is licensed in the United States for the treatment of diarrhea caused by Cryptosporidium parvum and Giardia lamblia, and has a favorable toxicology profile over a short duration of therapy.
The mechanism of action of nitazoxanide against anaerobic bacteria and protozoa is attributed to interference with anaerobic energy metabolism via pyruvate:ferredoxin oxidoreductase.
Nitazoxanide is also active against a range of viruses in cell culture models, and is currently in phase II clinical development for the treatment of chronic hepatitis C and chronic hepatitis B.
The mechanism of action of nitazoxanide against viruses involves enhancement of the intracellular activity of interferon by inducing eIF2alfa phosphorylation. Recent laboratory studies suggest that nitazoxanide does not induce antiviral resistance.
In patients with chronic hepatitis C genotype 4, nitazoxanide combined with peginterferon alfa-2a and ribavirin for nine months compared to standard peginterferon alfa-2a and ribavirin for twelve months increased the sustained virologic response to 79% versus 50%. In this trial, nitazoxanide was confirmed to be safe during long-term administration.
OBJECTIVE
The primary objective of this study was to evaluate the safety and efficacy of 24 weeks of nitazoxanide monotherapy for the treatment of chronic hepatitis C genotype 4.
Secondary objectives included evaluation of the effect of nitazoxanide monotherapy on sustained virologic response rates, reduction in serum HCV RNA levels, and changes in ALT levels.
Study
· Multicenter, randomized, double-blind, placebo-controlled study (RM01-3027)
· 50 patients 18 years of age with chronic hepatitis C genotype 4 and active disease on liver biopsy enrolled at two centers in Egypt
· Patients randomized to either one nitazoxanide 500 mg tablet or matching placebo twice daily for 24 weeks
· Patients followed every 4 weeks during the 24-week treatment period and for 24 weeks after therapy
· Serum HCV RNA measured by PCR (lower limit of detection = 10 IU/mL)
Efficacy Parameters
· ETR: HCV RNA undetectable at end of treatment
· SVR: HCV RNA undetectable 24 weeks after completion of treatment
Statistical Methods
· Analysis of data was intent to treat
· Proportions were compared using Fisher's exact tests (two-sided) and means using a student's t-test.
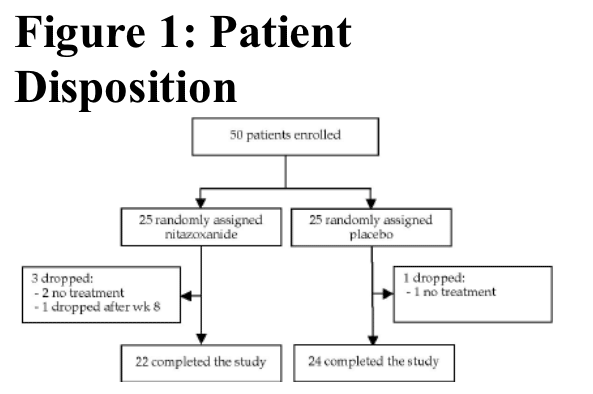
Protocol deviations: one patient in the nitazoxanide group was coinfected with HBV. Serum HBV DNA was 1000
copies/mL at baseline and then undetectable throughout therapy.
PREDICTORS OF RESPONSE TO THERAPY
Lower HCV RNA levels at baseline predicted virologic response on treatment (p = .0086). All responders had baseline serum HCV RNA <385,000 IU/mL.
Other predictors of virologic response were indicators of less severe liver disease:
- Higher platelet counts (p = .04)
- Lower prothrombin time (p = .06)
- Lower alpha-fetoprotein level (p = .07)
-- Absence of bridging fibrosis or cirrhosis
Absence of uncontrolled diabetes mellitus

Changes in ALT
Mean changes in ALT from baseline to end of treatment were not significantly different in two treatment arms.
Adverse Events
Seventeen patients (11 from the nitazoxanide group, and 6 from the placebo group) reported a total of 33 adverse events.
Two were serious adverse events.
The remaining adverse events were mild to moderate and transient in nature, with none requiring modification of discontinuation of treatment.
Adverse events were similar in the nitazoxanide and placebo groups
No laboratory serious adverse events were reported during the study.
REFERENCES
1. Anderson VR, Curran MP. Nitazoxanide: a review of its use in the treatment of gastrointestinal infections. Drugs. 2007;67:1947-1967.
2. Kamal SM, Nasser IA. Hepatitis C genotype 4: what we know and what we don't yet know. Hepatology. 2008;47:1371-1383.
3. Korba BE, Montero AB, Farrar K, Gaye K, Mukerjee S, Ayers MS, Rossignol JF. Nitazoxanide, tizoxanide, and other thiazolides are potent inhibitors of hepatitis B virus and hepatitis C virus replication. Antivir Res. 2008;77:56-63.
4. Korba BE, Elazar M, Lui P, Rossignol JF, Glenn JS. Studies of the potential for nitazoxanide or tizoxanide resistance in hepatitis C virus replicon-containing cell lines. Antimicrob. Agents. Chemother. In press.
5. Rossignol JF, Elfert A, El-Gohary Y, Kee_e EB. Randomized controlled trial of nitazoxanide-peginterferon-ribavirin, nitazoxanide-peginterferon and peginterferon-ribavirin in the treatment of patients with chronic hepatitis C genotype 4 [abstract]. J. Hepatol. In press. (Presented at the European Association for the Study of the Liver (EASL), Milan, Italy, 2008).
6. Rossignol JF, Elfert A, Kee_e EB. Evaluation of a 4-week lead-in phase with nit-azoxanide prior to nitazoxanide-peginterferon in treating chronic hepatitis C [abstract]. J. Hepatol. In press. (Presented at the European Association for the Study of the Liver (EASL), Milan, Italy, 2008).
|
| |
|
 |
 |
|
|