 |
 |
 |
| |
Safety, Tolerability and Efficacy of Darunavir/Ritonavir in Treatment-Experienced Women With HIV Infection: 24-Week Interim Analysis of GRACE (Gender, Race, And Clinical Experience)
|
| |
| |
Reported by Jules Levin
Judith Currier, MD, MSc1; Kathleen Squires, MD2; Dawn Averitt Bridge3; Robert Ryan, MS4; Joseph Mrus, MD,
MSc5; Alan Tennenberg, MD, MPH5; Ron Falcon, MD5; on behalf of the GRACE Study Group
1University of California, Los Angeles, School of Medicine, Los Angeles, CA, USA; 2Jefferson Medical College of Thomas Jefferson University,
Philadelphia, PA, USA; 3The Well Project, Inc., Atlanta, GA, USA; 4Tibotec, Inc., Yardley, PA, USA; 5Tibotec Therapeutics, Bridgewater, NJ, USA
AUTHOR CONCLUSIONS
GRACE is the largest treatment trial to date in North America to focus on women with HIV
The GRACE study is fully enrolled (287 women, 142 men), demonstrating that HIV-infected women from North America, including women of color, can be successfully recruited to participate in clinical trials of antiretrovirals
In this 24-week interim analysis of the first 203 patients (180 women and 23 men):
- Response rates were within the expected range from previous clinical trials of DRV/r in treatment-experienced patients
- AEs among women were generally similar to those seen in other studies of DRV/r in treatment-experienced patients
- Approximately one-quarter of women discontinued the study prior to Week 24; reasons were generally unrelated to AEs or virologic failure
Sex- and race-based comparisons will be available at Week 48 primary analysis for all 429 patients
INTROUCTION
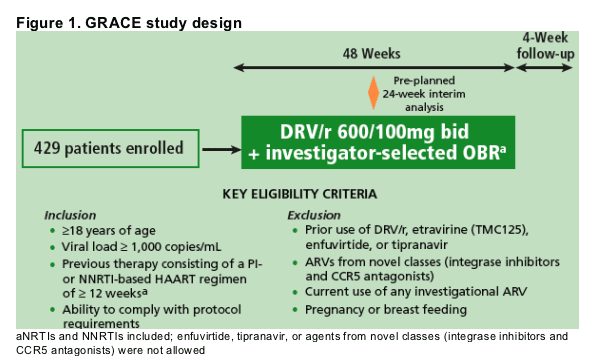
GRACE (Gender, Race, And Clinical Experience) is a multi-center, open-label phase IIIb trial designed to assess sex and race differences in efficacy, safety, and tolerability of darunavir/ritonavir (DRV/r; PREZISTA�/r) 600/100mg bid
plus an optimized background regimen (OBR) over 48 weeks in treatment-experienced patients
GRACE is the largest treatment trial in North America to date to focus on women with HIV, and the first in North America to focus on antiretroviral (ARV) treatment-experienced women
The study was designed to enroll a predominantly female, racially diverse population. The 65 study sites in North America were selected to meet these objectives
We report a pre-planned interim analysis of the first 203 patients to complete Week 24 or discontinue sooner
-- The cutoff date for the interim analysis was February 4, 2008, at which time 203 patients reached Week 24 or discontinued
METHODS
GRACE is a phase IIIb, non-inferiority study with a primary endpoint of difference in virologic response (HIV-1 RNA <50 copies/mL) between men and women at Week 48 (Figure 1)
- Maximal allowable difference in virologic response of 15%, one-sided significance level of 0.025 and 80% power
Patients with HBV/HCV were allowed to enroll if condition was clinically stable and not requiring treatment during the study
Patients were allowed to enter the study on treatment interruption of 34 weeks
All patients received DRV/r 600/100mg bid plus physician-selected OBR
- Zidovudine, emtricitabine, tenofovir, tenofovir/emtricitabine and etravirine were provided in the study, but not mandated for use
-- Enfuvirtide, tipranavir, integrase inhibitors and CCR5 antagonists were not allowed as part of the OBR
This pre-planned interim analysis was conducted primarily to assess safety and tolerability
Safety analysis was based on all available data collected through last study visit prior to interim analysis cutoff date; efficacy analysis was based on data collected through Week 24 or early termination
Virologic failure (VF) was defined as HIV-1 RNA >50 copies/mL at Week 24 or any visit beyond this time point, and confirmed by two consecutive measurements taken at least 14 days apart
We present preliminary efficacy results using:
- ITT-TLOVR: time to loss of virologic response algorithm; intent-to-treat population
-- TLOVR-non-VF: TLOVR algorithm; population excluded patients who discontinued for reasons other than VF
Baseline Demographics, Disease
Characteristics and Study R
287 women and 142 men enrolled in GRACE between November 2006 and November 2007
180 women and 23 men were included in this 24-week interim analysis
- Results for the 23 men included in this analysis are not presented separately due to small sample size
- Sex-based comparisons will be presented for the Week 48 primary analysis
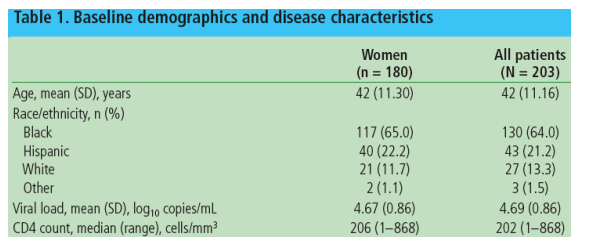
Baseline demographics and disease characteristics are presented for the 180 women and all 203 patients included in the interim analysis (Table 1)
-- The racial/ethnic composition of women in GRACE (65% black, 22% Hispanic) is reflective of HIV demographics among women in the US1eg
Women in this interim analysis of GRACE had a wide range of treatment experience (Table 2)
- At baseline, 17% of women had no active ARVs in the OBR; more than half had 2 or more active ARVs in their OBR
Approximately one-third of women (32.8%) and patients overall (32.5%) entered the study on a treatment interruption of >/=4 weeks.
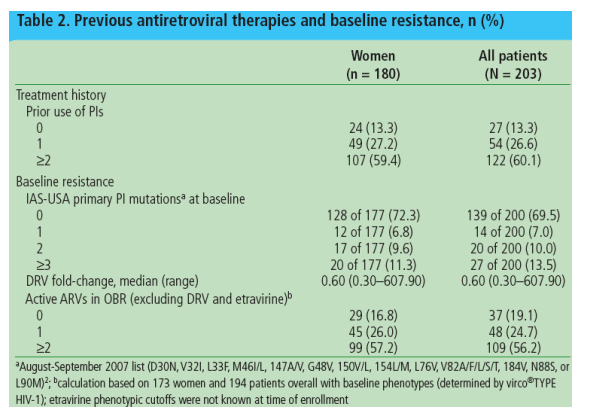
Etravirine was used in 40% of patients.
The NRTIs most commonly used in the OBR were tenofovir (81%) and emtricitabine (72%).
Patient Disposition
The mean exposure to study drug for the first 180 women enrolled was 33.4 weeks (SD 15.6)
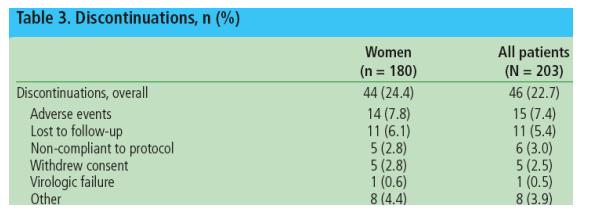
Approximately one-quarter (24.4%) of women discontinued the study by Week 24 in the interim analysis (Table 3)
The majority of discontinuations among women were due to reasons other than adverse events (AEs) (7.8%)
or VF (0.6%)
Examples of discontinuations for reasons other than AE or VF among women included:
- Withdrew consent (n=5; 2.8%)
- Non-compliant to protocol: use of unapproved medications (n=5; 2.8%)
- Pregnancy (n=1; 0.6%)
- Other (n=8; 4.4%)
Study site closed (n=3; 1.7%)
Moved out of geographic study area (n=1; 0.6%)
Cancelled medical appointments (n=1; 0.6%)
Patient taking too many medications; removed from study for safety
reasons (n=1; 0.6%)
Collection of patient- and site-level information is currently in progress to provide further details on reasons for discontinuation.
SAFETY
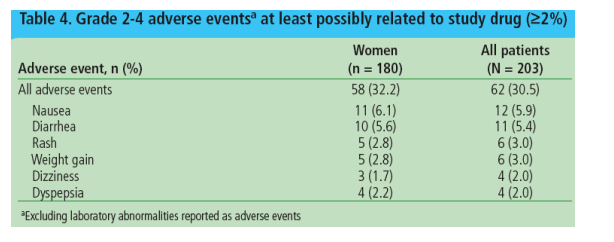
The most common (32%) grade 2-4 AEs at least possibly related to study drug are shown in Table 4
Serious AEs (SAEs) were reported in 16.7% (n=30) of women
- The most common SAE was pneumonia, reported in 8 (4.4%) women and no men
AEs led to discontinuation in 7.8% of women.
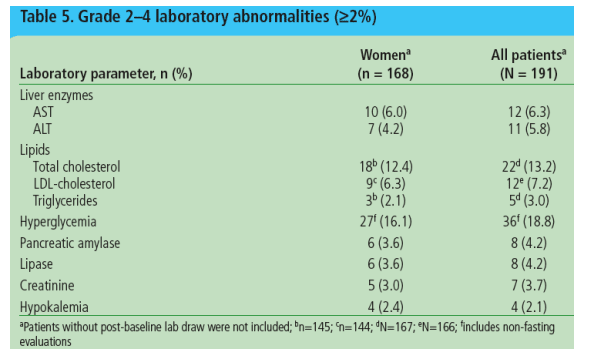
Grade 2-4 laboratory abnormalities (32%) are described in Table 5
EFFICACY
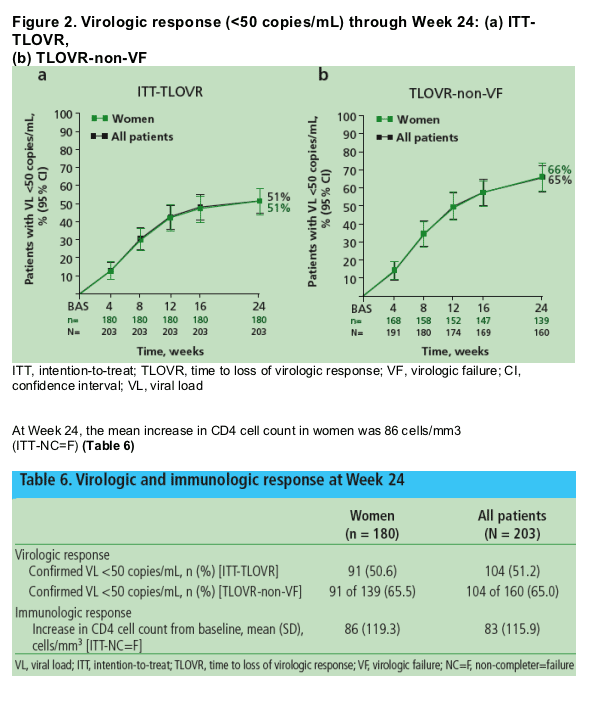
At Week 24, 51% (ITT-TLOVR) and 66% (ITT-non-VF) of women achieved HIV-1 RNA <50 copies/mL (Figure 2a and 2b, and Table 6)
On average, patients in GRACE were more PI-experienced than patients in the TITAN study and less PI-experienced than patients in the POWER studies
-- In the TITAN and POWER (1, 2 and 3) studies, 73.5% and 45% (ITT-TLOVR) of patients achieved HIV-1 RNA <50 copies/mL by Week 24, respectively
|
| |
|
 |
 |
|
|