 |
 |
 |
| |
Coagulation factor levels are not affected by tipranavir (TPV/r) capsules or Vitamin E-containing solution formulations nor by comparator protease inhibitors (PI)
|
| |
| |
Reported by Jules Levin
IAC Aug 3-8 2008 Mexico City
J Weitz1, V Kohlbrenner2, M Johnston3, D Neubacher4, P Robinson2
1McMaster University, Henderson Research Center, Hamilton, Ontario, Canada;
2Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT, USA; 3Hematology Reference Laboratory, Hamilton, Ontario, Canada;
4Boehringer Ingelheim Pharma GmbH & Co. KG, Biberach, Germany
ABSTRACT
Background: PIs may increase bleeding in hemophiliac HIV-infected patients (pts). In rats, TPV/r reduced levels of Vitamin K-dependent (vitK-dep) coagulation (coag) factors; an effect enhanced with co-administration of Vitamin E (vitE) and associated with bleeding. To examine whether these findings are relevant to humans, we measured levels of coag factors in stored plasma from pts randomized to TPV/r or comparator PI (CPI/r) in RESIST trials, and from children administered either vitE-containing TPV/r oral solution (TPV/r+VitE-OS) or TPV/r capsules.
Methods: Plasma samples (baseline [bsl], weeks 8, 24, and 48 of treatment) from randomly selected pts in RESIST and pediatric trial 1182.14 were assayed for levels of vitK-dep coag factors (prothrombin [FII] + factor VII [FVII] antigen and activity) and a non vitK-dep factor V(FV), as were PT/aPTT. Geometric mean on-treatment values (gMean) were compared to bsl. Analyses were powered to detect a clinically relevant ł30% reduction in the levels of FII and FVII.
Results: 25 TPV/r and 25 CPI/r pts in RESIST had samples assayed. Compared with bsl, gMean FII and FVII levels increased by 1.4% and 2.7%, respectively, in those given TPV/r and decreased by 1% and increased by 3.3%, respectively, in those receiving CPI/r. 25 TPV/r+VitE-OS children and 19 treated with TPV/r capsules had samples assayed. Compared with bsl, gMean FII and FVII levels
decreased by 3.8% and 4.1%, respectively, with TPV/r+VitE OS, and increased by 0.6% and 3.4%, respectively, with TPV/r capsules. There was no systematic change in PT/aPTT values nor change in FV levels in adults or children. None of these changes were statistically significant or clinically important. Additionally, in a subgroup of RESIST pts with bleeding events, there was no TPV/r effect on coag.
Conclusions: Neither TPV/r nor CPI/r produced decreases in levels or activity of vitK-dep coag factors or FV. TPV/r+VitE-OS had no effect on coag factors in children. Findings observed in rats appear to be species specific.
AUTHOR DISCUSSION
In Trial 1182.14, there was no difference in Vitamin K-dependent factor levels in pediatric patients treated with solution versus capsules. There was no difference between baseline and followup values for Vitamin K-dependent factors.
There was no difference in the bleeding markers PT and aPTT in patients treated with TPV OS versus TPV capsules.
There was no systematic effect of the Vitamin E containing TPV OS on blood coagulation.
There was no evidence of Vitamin K-dependent or non-Vitamin K-dependent mechanisms affecting factors II and VII or the other related coagulation markers.
Similarly, in the RESIST trials there was no difference in Vitamin K-dependent factor levels in patients treated with TPV/r or CPI/r. There was no effect amplified by patients treated with warfarin or Vitamin E, although the number of patients in this subgroup was small.
Evaluation of coagulation factor levels in patients with clinical bleeding events revealed no apparent effect of TPV/r on coagulation mechanisms as possible underlying causes of the bleeding episode. Rather these infrequent episodes remain primarily explained by the underlying concurrent conditions of patients with advanced HIV-disease.
Neither TPV/r nor CPI/r treatment produced decreases in either the levels or the activity of the Vitamin K-dependent coagulation factors or Factor V. The Vitamin E-containing TPV OS had no effect on coagulation factors compared to TPV capsules in children. Therefore, the effects observed in the rat study would appear to be species specific and of no clinical relevance for patients taking TPV OS or TPV capsules.
INTRODUCTION
A consistent side effect of protease inhibitor (PI) treatment has been the development of an increased bleeding tendency in HIV-infected patients with hereditary bleeding disorders [1-5]. Two large case studies have been published which have helped to characterize the nature of PI-associated bleeding in patients with hereditary bleeding disorders [6,7]. However, the increased bleeding tendency associated with PI therapy does not appear to be restricted exclusively to patients with hereditary bleeding disorders. In one publication, 28 cases of bleeding were reported in HIV-infected patients with no bleeding disorders [6]. Among non-hemophilic patients, the mucous membranes and the gastrointestinal tract were the most common sites of bleeding. Skin, soft tissue, and intracranial bleeds were also reported [6]. The mechanism responsible for the increased bleeding associated with PI therapy is unknown. The reduced efficacy, or even failure, of clotting factor replacement therapy in non-hemophilic
individuals suggests that PIs do not directly exacerbate the inherent increased bleeding risk of low factor VIII and factor IX levels. Furthermore, investigation of patients with PI-associated bleeding has not revealed evidence of altered levels of coagulation factors, von Willebrand factor or fibrinogen [1,2,4,8].
Tipranavir, 500 mg twice daily, co-administered with 200 mg ritonavir twice daily (TPV/r), was approved by the FDA for use in treatment-experienced HIV-1 infected adult patients in June 2005. In May 2006, 13 TPV/r-treated patients with intracranial hemorrhage (ICH) were identified across the BI-sponsored clinical development program for an overall ICH rate estimated as 0.22/100 patient years of exposure (PEY). This compares with a rate of 0.07/100 PEY (95% CI: 0.05-0.10) in the VA Virtual Cohort and of 0.23/100 PEY (95% CI: 0.20-0.25) in the Medi-Cal cohort; there were a total of 49,610 individuals with HIV infection in these cohorts [9]. The product label was updated to note that TPV has been associated with reports of both fatal and non-fatal ICH. Many of these patients had other medical conditions or were receiving concomitant medications that may have caused or contributed to ICH. No abnormalities of the coagulation pathways were observed in patients in general or preceding the development of
ICH. Therefore, routine measurement of coagulation parameters is not currently indicated in the management of patients receiving TPV/r.
The TPV oral solution (OS) formulation contains an excipient: Vitamin E-TPGS (d-_-tocopherol polyethylene glycol 1000 succinate), which is a water-soluble Vitamin E preparation containing 25% Vitamin E. Vitamin E is known to affect Vitamin K-related coagulation factors in rats. A study was conducted to evaluate any potential interactions between TPV oral solution and Vitamin K-related
coagulation pathways in rats [data on file]. Rats were given TPV in doses ranging from 500/600 mg/kg to 3500/1200 mg/kg coadministered with Vitamin E-TPGS/TPV; or TPV alone at a dose of 600 or 1200 mg/kg. The prothrombin time (PT) and activated partial thromboplastin time (aPTT) were both increased in rats given TPV alone. However, the effects on aPTT and PT were exacerbated by the
co-administration of Vitamin E-TPGS and TPV. Rat mortality associated with a hemorrhagic diathesis was also noted. Hemorrhagic effects were observed within the first several days of dosing. The preliminary results from this study suggested that the coadministration of Vitamin E-TPGS exacerbates the anticoagulant effect of TPV in rats but the precise mechanism for the interaction between TPV and Vitamin E-TPGS in rats is unknown.
In a separate 22-week study, TPV caused a time-related decrease in the concentrations of Vitamin K-dependent factors VII and IX in rats, concomitant with increases in aPTT [data on file]. These effects were prevented by the co-administration of Vitamin K. Tipranavir also inhibited arachidonic acid-induced platelet aggregation, an effect that was partially mitigated by co-administering Vitamin K. Thus, TPV appears to have an effect on the Vitamin K-dependent bleeding cascade in rats. Similarly, there are published studies that describe
increases in the PT and aPTT in rats exposed to high doses of Vitamin E, an effect associated with a decrease in Vitamin K-dependent clotting factor function [10-13]. Collectively, these data suggest that the hepatic Vitamin K cycle is the most likely point of interaction between TPV and Vitamin E.
Although there is no clinical evidence to suggest an increased risk of bleeding in patients who take TPV and Vitamin E, further elucidation of the mechanisms observed in rats was undertaken by evaluating the levels of Vitamin K-dependent coagulation factors in stored plasma samples from patients who took part in TPV clinical trials. In this poster, we report the results of analyses of clinical samples in a retrospective study designed to investigate potential mechanisms that could contribute to an increased risk of bleeding in patients taking TPV/r.
METHODS
Patients
Trial 1182.14 was an open label, multiple dose, randomized study of the safety and pharmacokinetics of TPV/r, administered as capsules or oral solution, in pediatric patients infected with HIV-1. The RESIST trials were randomized, open-label, comparative safety and efficacy studies of TPV/r versus genotypically-defined comparator protease inhibitor/ritonavir (CPI/r) in multiple antiretroviral drug-experienced adult patients. During the studies, serial plasma samples were
obtained from patients and frozen for subsequent safety investigations. Serial samples were selected for patients from the oldest age groups in Trial 1182.14 and for randomly chosen patients in RESIST for Vitamin K-dependent coagulation factor determination. Additionally, data from subsets of patients (those who took concomitant warfarin, or Vitamin E (_-tocopherol), or who were in the amprenavir arm [AGENERASE capsules contain _-tocopherol]) and
patients who had clinical bleeding events were analyzed. Patients who fulfilled the inclusion criteria (Table 1) were selected using the following procedures.

Trial 1182.14:
25 patients treated with TPV oral solution and 19 patients treated
with TPV capsules.
RESIST:
· Twenty five patients treated with TPV/r and 25 patients treated with CPI/r (lopinavir or saquinavir) without severe or serious bleeding events, and without use of concomitant warfarin or Vitamin E;
· Five patients treated with TPV/r taking warfarin at baseline and/or concomitantly throughout study treatment;
· Six patients treated with TPV/r taking Vitamin E (__tocopherol) at baseline and/or concomitantly throughout study treatment
· Within the amprenavir treatment stratum, eight patients treated with TPV/r and eight patients treated with amprenavir
· Ten patients with clinical bleeding events reported as severe or serious who had been treated with TPV/r
Laboratory methods
The retrospective laboratory study had two primary objectives:
1. To investigate whether there is an effect of the formulation of TPV/r (oral solution versus soft gel capsule) on Vitamin K-dependent coagulation factors in the pediatric Trial 1182.14.
2. To investigate whether there is a TPV/r effect on Vitamin Kdependent
coagulation factors in comparison to an active PI control treatment in the RESIST trials.
Secondary objectives were:
1. To compare the effect of TPV/r on the study endpoints relative to baseline in the treatment groups from the 1182.14 and RESIST trials.
2. To explore the presence of a trend over time in the study endpoints in the treatment groups from the 1182.14 and RESIST trials.
3. To evaluate Factor V levels in the treatment groups from the 1182.14 and RESIST trials, as a control of each of the measures listed above.
4. To explore whether Vitamin K-dependent coagulation factors II and VII change when warfarin or Vitamin E (__tocopherol) is coadministered with TPV/r.
5. To explore a potential association between treatment dependent changes in Vitamin K-dependent coagulation factors II and VII and changes in prothrombin time in RESIST patients.
6. To explore whether Vitamin K-dependent coagulation factors II and VII change over time in RESIST patients with severe or serious clinical bleeding events.
Frozen plasma samples obtained at baseline and at serial timepoints (Weeks 8, 24, and 48) from patients in Trial 1182.14 and the RESIST trials were assayed for Vitamin K-dependent coagulation factors (II and VII), and Factor V (not Vitamin K-dependent, as control). Prothrombin time measurements were determined as part of the original clinical trial. As an additional control, levels of Ecarin factor II were measured. In order to explore a possible mechanism for bleeding in patients with severe or serious bleeding events, levels of matrix gla protein were measured.
All samples were analyzed at the Hemostasis Reference Laboratory, Ontario, Canada. The normal reference ranges for these assays performed by this laboratory are as follows:

Statistical methods
The first set of primary null-hypotheses was that the effect of the TPV/r oral solution on Vitamin K-related coagulation factors II and VII is equal to that of the TPV/r capsule in 1182.14. The endpoint tested was the proportional change from baseline to the average on-treatment value obtained on the assigned formulation.
The second set of primary null-hypotheses tested was that TPV/r has an effect equal to the comparator treatment in RESIST on Vitamin K-related coagulation factors II and VII. The endpoint tested was the proportional change from baseline to the average on-treatment value. The primary analyses were performed using two-sided t-tests.
Secondary analyses applied descriptive statistical methods and explorative statistical tests to explore potential trends over time, to assess the impact of warfarin and Vitamin E on Vitamin K-related coagulation factors and to explore the level of Vitamin K-related coagulation factors in patients with severe or serious bleeding events.
The coagulation markers were chosen to give a robust evaluation of Vitamin K dependent and non-Vitamin K-dependent mechanisms. The study was well powered to be able to detect clinically relevant changes of 30%.
RESULTS
Overall, 541 adults and 44 children treated with TPV/r had baseline and serial on-treatment stored frozen plasma samples assayed retrospectively for coagulation factor levels. The 44 pediatric patients took either TPV oral solution, which contains Vitamin E as TPGS, or TPV capsules, which do not contain Vitamin E. This allowed comparisons to be performed to examine the effect of Vitamin E (OS versus capsule), or TPV (pre-dose versus on-treatment with TPV) on coagulation. For the adults, comparisons were made for the effect of TPV/r on coagulation compared to other PIs. Thirty-three (33) adults treated with comparator PIs had samples assayed.
Trial 1182.14
In the 12- to 18-year age group in Trial 1182.14, 14 eligible patients using TPV OS and 18 eligible patients using TPV capsules had samples selected for coagulation factor analyses. In the 6- to <12-year age group, 11 patients using TPV OS and an additional patient using TPV capsules were selected. Overall, 25 patients using TPV OS and 19 patients using TPV capsules had samples included for coagulation factor analyses.
There were only minor changes in coagulation factor levels and coagulation times when comparing the TPV/Vitamin E-containing OS formulation with TPV capsules (Table 2). Average on-treatment Factor II levels decreased by 3.8% from baseline in children receiving the OS, compared with an increase of 0.6% for capsules. Average on-treatment functional Factor VII levels were decreased by 4.1% from baseline for the OS compared with an increase of 3.4% for capsules. Factor V levels were unaffected. None of the changes were statistically significant or considered clinically important.
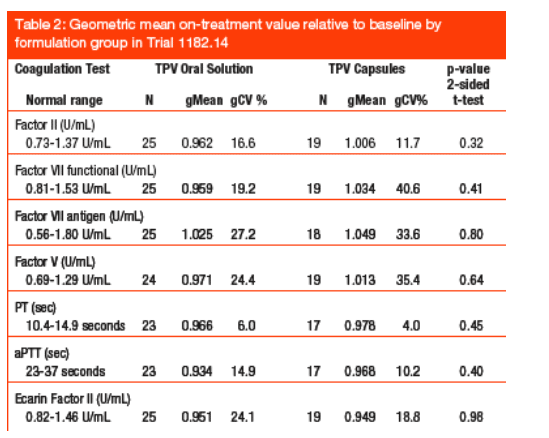
There was no prolongation of PT or aPTT in any of the treatment groups. On the contrary, coagulation times were shorter on-treatment relative to baseline in both treatment formulation groups. No patients had PT or aPTT outside of the normal range at any time during the study except for one patient. This patient who developed clinical bruising had an underlying coagulopathy, which could not be characterized. However, the coagulopathy was shown not to be due to Vitamin K deficiency alone and was not exacerbated by TPV/r administration.
A series of comparisons were conducted by each formulation (Ecarin Factor II, Factor II, Factor V, Factor VII antigen, Factor VII functional, Ratio of Factor VII functional/antigen, PT, and aPTT). The coagulation factor level and coagulation time changes over the course of treatment were assessed. A similar series of analyses for this set of coagulation parameters was performed comparing values within formulations relative to baseline at discreet timepoints on treatment. No significant or clinically relevant changes were observed in both analyses.
RESIST
In total, samples from 87 patients from the RESIST trials were tested for coagulation factors. Demographic data for these patients are shown in Table 3.
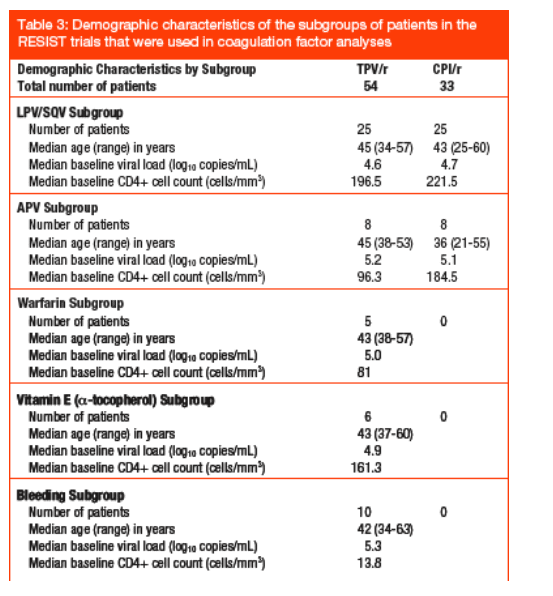
Compared with baseline, geometric mean Factor II and functional Factor VII levels increased by 1.4% and 2.7%, respectively, in those given TPV/r and decreased by 1.0% and increased by 3.3%, respectively, in those receiving CPI/r. Neither TPV/r nor CPI/r had any effect on PT values. None of these changes were statistically significant or considered clinically important (Table 4).
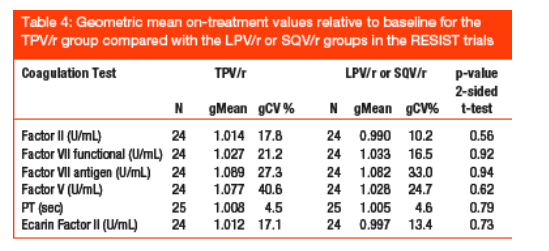
The following analyses were conducted: a non-parametric analysis of geometric mean on-treatment coagulation parameter values relative to baseline by treatment group (TPV/r and CPI/r); an analysis of average values for these treatment groups at discreet timepoints on treatment; and a comparison of coagulation parameter values relative to baseline at discreet timepoints on treatment for each of the two treatment groups. No significant or clinically relevant changes were observed.
Although there were three individuals in the LPV/SQV subgroup who had Vitamin K-dependent coagulation factor levels below the reference range, no coagulation time changes were noted. There were no patients in either the TPV/r or CPI/r group with prolonged PT values.
A mixed linear model with factors treatment group, visit and patient, as well as baseline as a linear covariate, was fitted to each of the endpoints for an integral assessment of potential influential variables. The only significant variable was the baseline value. In all models, the other factors remained non-significant. The non-significance of the factor visit provides evidence for the absence of a trend over time.
A second model added the interaction between treatment group and visit to explore potential differential effects over time between the treatment groups. Baseline values were significant for all endpoints. A significant impact on Factor II was seen in the visit by treatment group interaction (p=0.0442). The most extreme estimate was 0.06706, seen for CPI/r at Visit 11 (Week 48). This estimate is equivalent to a 7% increase in Factor II. All other estimates were smaller and no consistent trends over time were seen for the two treatment groups.
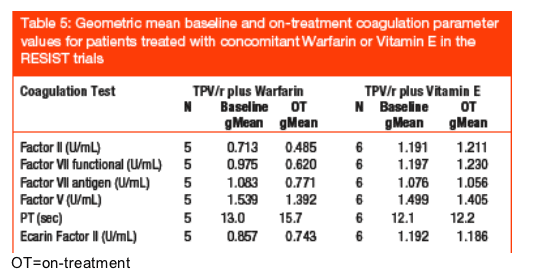
Patients taking warfarin or concomitant Vitamin E
Samples from five patients taking concomitant warfarin and six patients taking concomitant Vitamin E supplements during the course of TPV/r treatment in the RESIST trials were assayed. In patients taking concomitant warfarin, compared with baseline, geometric mean Factor II, functional Factor VII, and Factor VII antigen levels each decreased while the PT increased slightly. Factor V levels
remained unchanged. For patients taking concomitant Vitamin E (__tocopherol), coagulation parameters did not change (Table 5). None of these changes were considered clinically important; on the contrary, the level of anticoagulation achieved by warfarin was low.
Patients taking amprenavir
The amprenavir capsule formulation contains approximately 1,700 IU of Vitamin E per daily dose of amprenavir. Samples from eight TPV/r and eight APV/r patients in the RESIST trials were assayed. Compared with baseline levels, the geometric mean Factor II and functional Factor VII levels remained essentially the same in TPV/r patients whereas the Factor VII antigen levels increase slightly. Similarly for APV/r patients, the geometric mean Factor II and function
Factor VII levels remained the same on-treatment as at baseline but Factor VII antigen levels decreased slightly. None of these minor shifts had any effect on PT or were clinically important.
Exploratory analysis of coagulation factor effects in RESIST
patients who had bleeding events
During the course TPV/r treatment, 10 patients in the RESIST trials had bleeding events classified as severe in intensity or meeting serious adverse event reporting criteria. Three of these events were ICH. The others were: rectal bleeding (N=2); cerebro-vascular accident (N=1); fecal occult blood (N=1); post-operative hemorrhage (N=1); intra-abdominal hemorrhage (N=1); Intestinal hemorrhage (N=1). In the majority of cases, these 10 patients had medical
conditions in addition to their HIV infection and/or were receiving concomitant medications that may have contributed to the development of bleeding events. Evaluation of the patientsŐ coagulation factors was performed as exploratory analyses. Stored frozen plasma samples closest to the time of the bleeding event were analyzed, as well as samples preceding and following the clinical event, if available.
Low levels of Vitamin K-dependent factors were only detected in two patients, neither of whom had prolongation of PT. The course of Factor II levels over time for the 10 patients is shown in Figure 1.
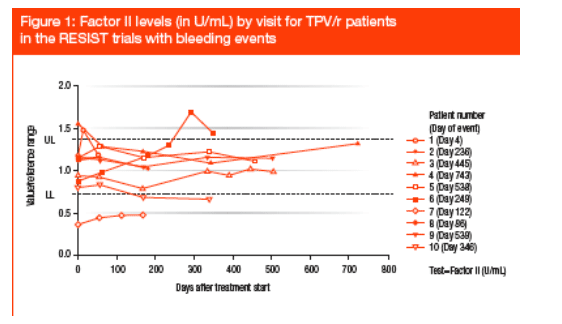
The differences between on-treatment values and baseline for the coagulation parameters were trivial. There was no systematic decrease in Vitamin K-dependent coagulation factor levels; Factor II, Factor VII functional and Factor VII antigen all increased slightly. PT remained unchanged. Measurements before and after the bleeding event were available for four patients. Coagulation factors showed average increases between 5% and 38%, while average matrix gla protein decreased by 0.8%. The individual patientsŐ bleeding events are not explained by an effect of TPV/r on Vitamin K-dependent coagulation factors but by the complex pathology of advanced HIV disease and underlying conditions.
REFERENCES
1. Mandalaki T; Katsarou O; Panagiotopoulou C; Karafoulidou A. Does protease inhibitor treatment induce increased bleeding tendency in haemophilia? Haemophilia 4, 766-767 (1998)
2. Yee TT; Amrolia PJ; Lee CA; Mir N; Giangrande PL.Protease inhibitors and unusual bleeding in haemophiliacs. Haemophilia 3, 220-221 (1997)
3. Wilde JT. Protease inhibitor therapy and bleeding. Haemophilia 6, 487-490
(2000)
4. Ginsburg C; Salmon-Ceron D; Vassilief D; Rabian C; Rotschild C; Fontenay- Roupie M; Stieltjes N; Sicard D.Unusual occurrence of spontaneous haematomas in three asymptomatic HIV-infected haemophilia patients a few days after the onset of ritonavir treatment. AIDS (Phila) 11 (3), 388-389
(1997)
5. Perez R; Martinez ML; Cisneros JM; Sosa R. Incidence of increased number of bleeding episodes in HIV positive haemophiliacs treated with protease inhibitors. Mtg of the World Federation of Haemophilia, 17 - 21 May 1998, Haemophilia 4, 276 Abstr 479 (1998)
6. Racoosin JA; Kessler CM. Bleeding episodes in HIV-positive patients taking
HIV protease inhibitors: a case series. Haemophilia 5, 266-269 (1999)
7. Wilde JT; Lee CA; Collins P; Giangrande PLF; Winter M; Shiach CR.
Increased bleeding associated with protease inhibitor therapy in HIVpositive
patients with bleeding disorders. Br J Haematol 107, 556-559 (1999)
8. Stanworth SJ; Bolton MJ; Hay CRM; Shiach CR. Increased bleeding in HIV-positive haemophiliacs treated with antiretroviral protease inhibitors.
Haemophilia 4, 109-114 (1998)
9. Fultz, S. L., D. Zingmond, et al. Evaluation of Intracranial Hemorrhage in 49,610 HIV-infected Veterans and California Medicaid Recipients. 14th CROI, Los Angeles, USA. Abs. 819. (2007)
10. Abdo KM; Rao G; Montgomery CA; Dinowitz M; Kanagalingam K. Thirteen-week toxicity study of d-alpha tocopheryl acetate (Vitamin E) in Fischer 344
rats. Food Chem Toxicol 24 (10/11), 1043-1050 (1986)
11. Krasavage WJ; Terhaar CJ. d-alpha-tocopheryl poly(ethylene glycol) 1000
succinate. Acute toxicity, subchronic feeding, reproduction, and teratologic studies in the rat. J Agric Food Chem 25 (2), 273-278 (1977)
12. Takahashi O; Ichikawa H; Sasaki M. Hemorrhagic toxicity of d-alphatocopherol in the rat. Toxicology 63, 157-165 (1990)
13. Takahashi O. Haemorrhagic toxicity of a large dose of alpha-, beta-,
gamma- and delta-tocopherols, ubiquinone, beta-carotene, retinol acetate
and L-ascorbic acid in the rat. Food Chem Toxicol 33 (2), 121-128 (1995)
|
| |
|
 |
 |
|
|