 |
 |
 |
| |
The Safety, Efficacy, and Steady State Pharmacokinetics of Atazanavir/Ritonavir (ATV/r)
Once Daily During Pregnancy: Results of Study AI424182
|
| |
| |
Reported by Jules Levin
5th International AIDS Society, July 19-22, 2009, Cape Town, South Africa
F. Conradie,1 C. Zorilla,2 D. Josipovic,3 M. Botes,4 Y. Osiyemi,5 M. Gomez,6 M. Mathew,7 E. Vandeloise,8 K. Grimm,9 T. Eley,10 M. Child,10 Y. Wang,10 R. Bertz,10 W. Hu,7 A. Collins,8 S. Hilaly,11 D. Mc Grath7 for the AI424182 Study Group
1HIV Clinical Trial Unit, Helen Joseph Hospital, University of Witswatersrand, Westdene, South Africa, 2UPR School of Medicine, San Juan, Puerto Rico, 3Perinatal Research Unit, University of The Witwatersrand,
Soweto, South Africa, 4Scion Clinical Research, Pretoria, South Africa, 5Triple O Research Institute, West Palm Beach, FL, USA, 6Bristol-Myers Squibb, Research and Development, Bedfordview, South Africa,
7Bristol-Myers Squibb, Research and Development, Wallingford, CT, USA, 8Bristol-Myers Squibb, Research and Development, Braine l'Alleud, Belgium, 9Bristol-Myers Squibb, US Medical Affairs, Plainsboro, NJ, USA
10Bristol-Myers Squibb, Discovery Medicine and Clinical Pharmacology, Hopewell, NJ, USA, 11Bristol-Myers Squibb, Research and Development, Rueil, France
AUTHOR DISCUSSION
Compared to ATV/r 300/100 mg in HIV-infected non-pregnant adults:
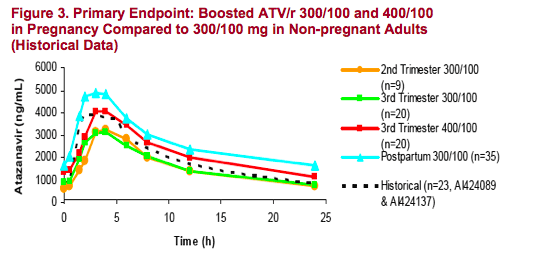
- ATV/r 300/100 mg has a lower third trimester AUC, but similar Cmin
- ATV/r 400/100 mg has a similar third trimester AUC but higher Cmin
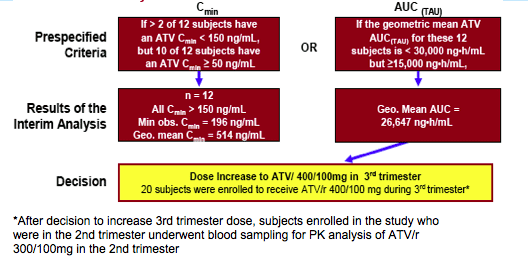
All Cmins observed were > 10X EC90 for ATV (EC90 = 14 ng/mL for wild-type virus). The lowest observed Cmin was 196 ng/mL
Levels of ATV are elevated in the post-partum period. This observation has been made for other PIs in the postpartum period. Levels of ATV appear to normalize by week 16 post partum2
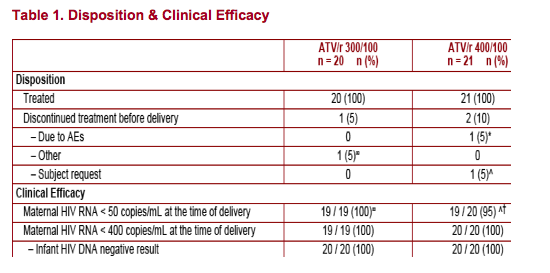
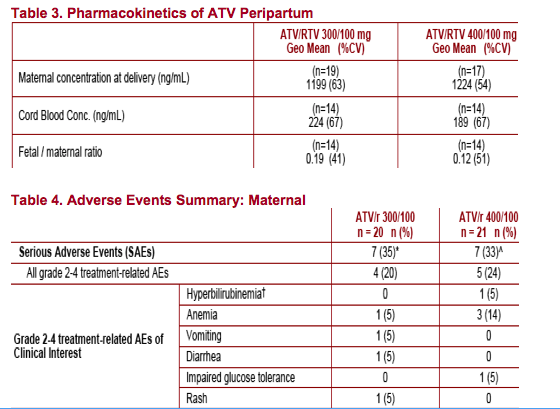
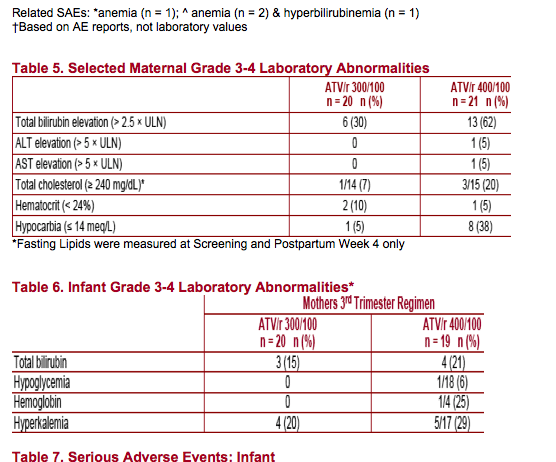
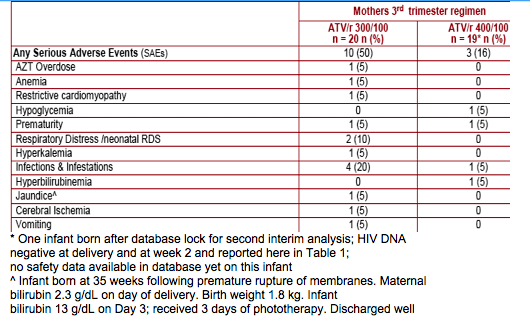
Both ATV/r 300/100 and ATV/r 400/100 were well tolerated with no unexpected, related AEs. However, ATV/r 400/100 dosing was associated with twice as much Grade 3-4 hyperbilirubinemia as ATV/r 300/100
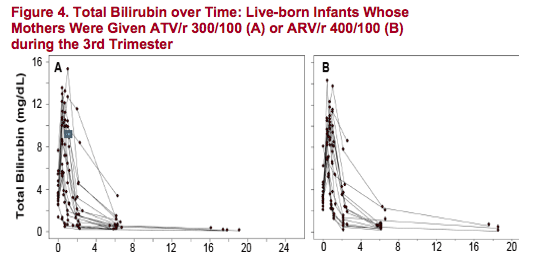
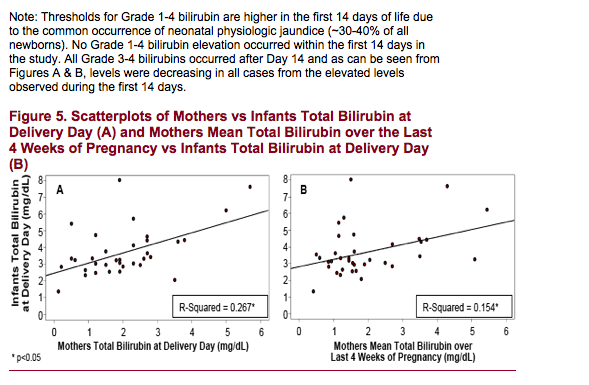
Most infants had some elevation of bilirubin. The level and pattern of bilirubin elevation in infants was generally consistent with physiologic jaundice, with the highest elevations around Day 3 with subsequent reductions to normal
levels over the next several weeks. Bilirubin levels in infants on the day of birth are only partly explained by maternal levels at the time of delivery. One infant with risk factors for neonatal jaundice (premature birth and low birth weight)
had phototherapy
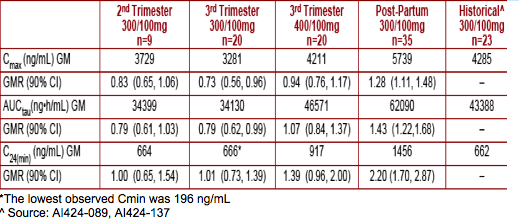
The ratio of maternal to cord blood ATV indicates that, as with other PIs, ATV does not freely cross the placenta, however, the levels achieved in the cord blood may provide some anti-viral protection to the fetus
Full suppression of HIV viral replication (HIV RNA < 50 copies/mL) was achieved in all mothers, and no mother-to-child HIV transmission occurred
AUTHOR CONCLUSIONS
ATV/r 300/100mg once daily dosing has a 27% lower AUC in the 3rd trimester of pregnancy compared to the AUC in non-pregnant adults, however, Cmins are similar. Increasing the dose to ATV/r 400/100mg achieves a similar AUC to
non-pregnant adults and higher Cmins, but with more frequent grade 3-4 hyperbilirubinemia
ATV/r 300/100mg once daily dosing appears to provide adequate ATV exposure throughout pregnancy and was well tolerated in these HIV-infected pregnant women. Adequate Cmins with reduced maternal hyperbilirubinemia compared
to ATV/r 400/100mg, and no need for dose modification in the 3rd trimester or post-partum, suggests that ATV 300/r in combination with a SOC backbone could be a potential treatment option for HIV-infected pregnant women in need of
HAART
The clinical outcomes from this phase I study suggest that ATV 300/100mg once daily was efficacious in the suppression of HIV RNA in these patients, and prevented the transmission of mother-to-child HIV-1 infection when used in combination with AZT/3TC twice daily
Treatment with ATV/r in the mothers appeared to be well tolerated by the newborn infants in this study. Bilirubin elevation was common among infants and is likely mostly physiologic in nature with only partial correlation with maternal levels at the time of delivery
Introduction
There are very limited options for the use of protease inhibitors as a component of HAART in HIV-infected pregnant women
There remains an unmet medical need for a once-daily, safe, efficacious and well tolerated protease inhibitor (PI)
for use during pregnancy
Atazanavir (ATV) is a potent, well-tolerated, once-daily HIV-1 PI with established efficacy in both treatment-naïve and treatment-experienced adult, non-pregnant HIV-infected patients
HIV protease inhibitor exposures are generally reduced during pregnancy, especially during the 3rd Trimester
There are some data to suggest that atazanavir levels, while reduced during pregnancy, may nonetheless be adequate to recommend dosing at the usual non-pregnant dose of atazanavir 300mg boosted with low-dose ritonavir 100mg (ATV/r 300/100mg)1
However, further data are needed before a recommendation can be made regarding adequate ATV/r dosing in pregnancy
Objectives
Primary objective: To determine what dosing regimen of ATV/r produces adequate drug exposure during pregnancy compared to historical data in HIV-infected subjects
Secondary objectives
- Measure maternal:infant ATV level ratio
- Safety of ATV/r in pregnant women
- Safety in infants born to women exposed to ATV/r during pregnancy
- Antiviral efficacy
Suppression of HIV RNA in mothers
Prevention of mother-to-child transmission of HIV-1
Methods
Multicenter, open-label, prospective, single-arm phase 1 study
- Enrollment in South Africa, Puerto Rico, and the USA
Study Population: HIV-1 infected pregnant women between 12- 32 weeks gestation; CD4 ≥ 200 cells/mm3
Treatment: ATV/r 300 or 400/100 mg once-daily and ZDV/3TC 300/150 mg twice-daily
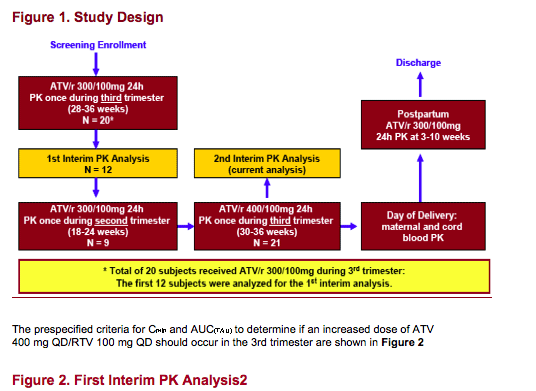
Planned First Interim PK analysis after 12 subjects received ATV/r 300/100mg during third trimester with prespecified criteria for increasing dose to ATV/r 400/100mg
Second Interim Analysis after primary endpoint data available (all third trimester PK data at both ATV/r 300/100 mg and 400/100mg)
Second trimester PK data for ATV/r 300/100 mg was collected in a limited number of subjects
Post-partum PK was assessed between 3-10 weeks after delivery
Infants were assessed by HIV DNA testing on the date of delivery and at week 2, 6, 16 and 24

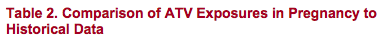
REFERENCES
1.Ripamonti et al. Atazanavir plus low-dose ritonavir in pregnancy: pharmacokinetics and placental transfer. AIDS 2007, 21:2409-2415.
2. Eley T et al. Steady state pharmacokinetics and safety of atazanavir after treatment with ATV 300 mg once daily/ritonavir 100 mg once daily +
ZDV/3TC during third trimester in HIV + women. Poster presented at 15th Conference on Retroviruses and Opportunistic Infections (CROI), February 3-6, 2008; Boston, MA, USA.
|
| |
|
 |
 |
|
|