 |
 |
 |
| |
Screening for HIV Tropism Using Population-Based V3 Genotypic Analysis: A Retrospective Virological Outcome Analysis Using Stored Plasma Screening Samples From MOTIVATE 1
|
| |
| |
Reported by Jules Levin
18th HIV Drug Resistance Workshop
June 9-12
Ft Myers Florida
P.R. Harrigan1, R. McGovern1, W. Dong1, A. Thielen2, M. Jensen3, T. Mo1, D. Chapman4, M. Lewis5, I. James5, J. Heera6, H. Valdez41BC
Valdez41BC Centre for Excellence in HIV/AIDS, Vancouver, Canada; 2Max-Planck Institute for Informatics, Saarbrucken, Germany; 3Fortinbras Research, Buford, GA; 4Pfizer, Inc., New York, NY; 5Pfizer Global R&D, Sandwich, UK; 6Pfizer Global R&D, New London, CT
Author Conclusions
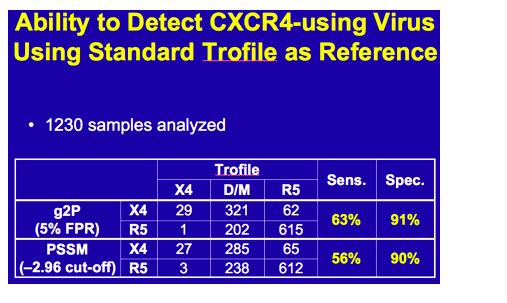
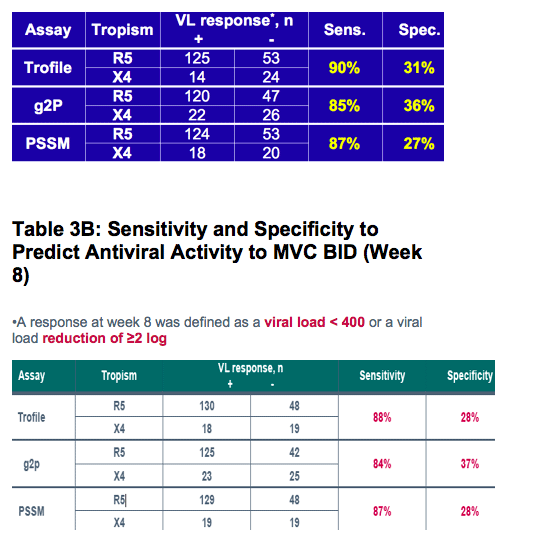
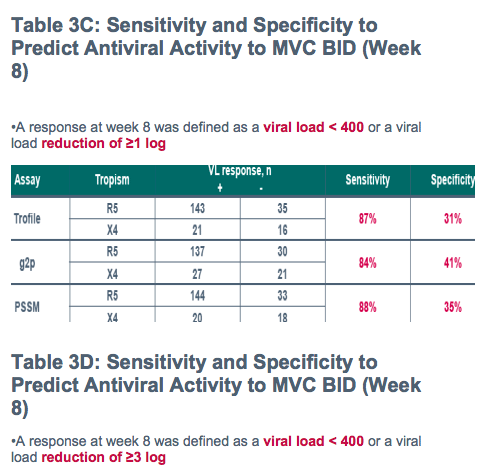
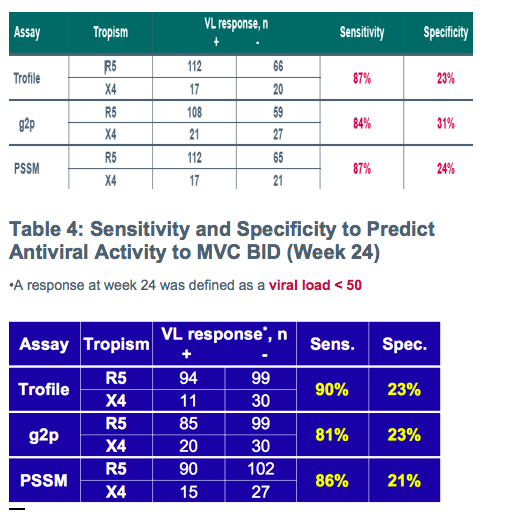
Despite apparently poor sensitivity of population V3 genotyping for predicting non-R5 HIV by standard Trofile:
-All methods discriminated between short-term responders and non-responders to maraviroc with broadly similar performance in this treatment-experienced population
-Observation was consistent in sub-analyses
-Similar responses were maintained over the 24 weeks
INTRODUCTION & OBJECTIVES
CCR5 antagonists require a test for HIV tropism before use
Tropism tests typically utilized include phenotypic tests such as the Monogram Trofile and the enhanced sensitivity Trofile assays
Sequencing of the V3 loop provides a genotypic alternative
Objectives:
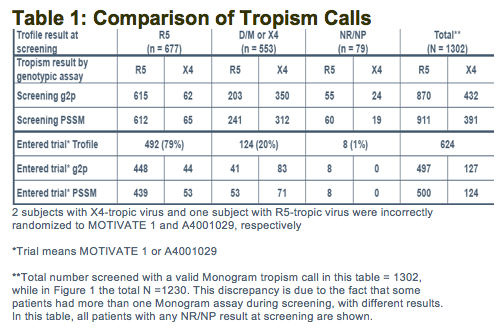
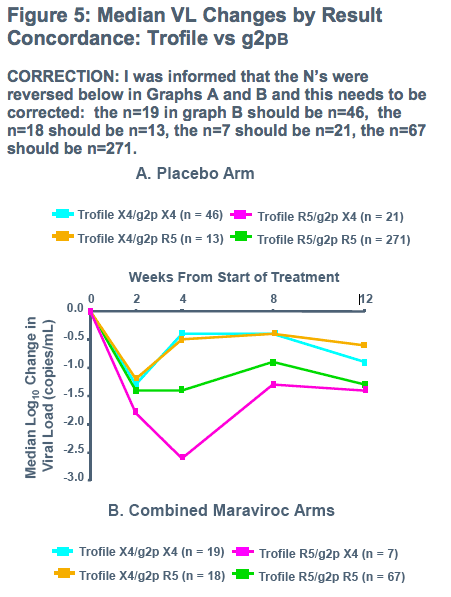
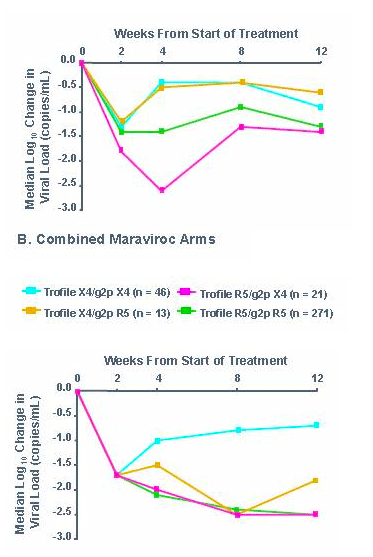
-To determine concordance between screening Trofile and screening genotyping of V3 loop
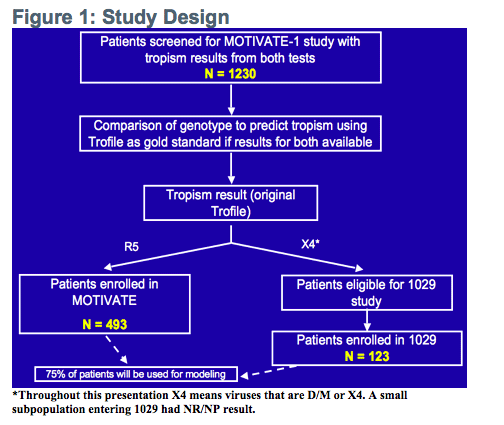
(500 people from 1027 MOTiVATE [X4 screened out] with R5 and 123 from study 1029
-To determine the ability of Trofile and V3 loop genotyping to predict responses to Maraviroc (MVC) in treatment-experienced patients
METHODS
Population-based sequencing of V3 on frozen, stored screening plasma samples from the MOTIVATE 1 study (MOTIVATE 1 participants and study 1029 participants)
-94% of patients had Clade B HIV-1
Triplicate PCR with automated sequence analysis
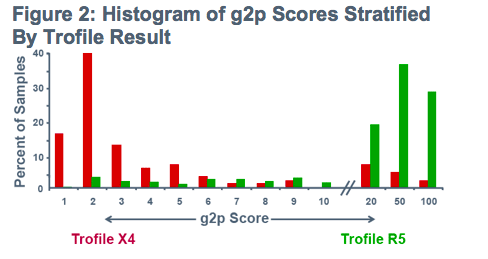
Tropism prediction using 2 algorithms:
-PSSM (-2.96 cutoff) -or-Geno2pheno (g2p; 5% FPR)
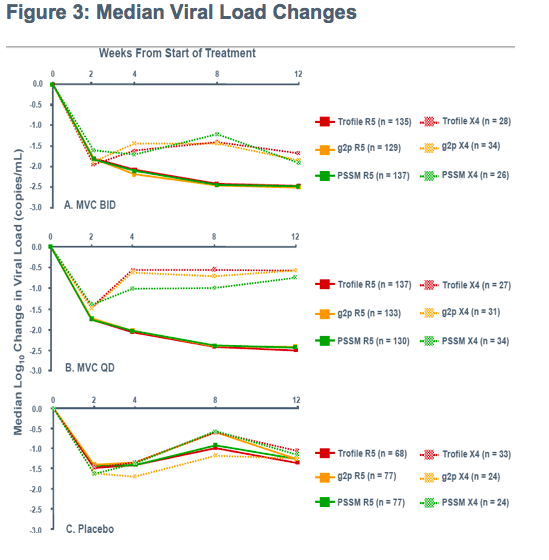
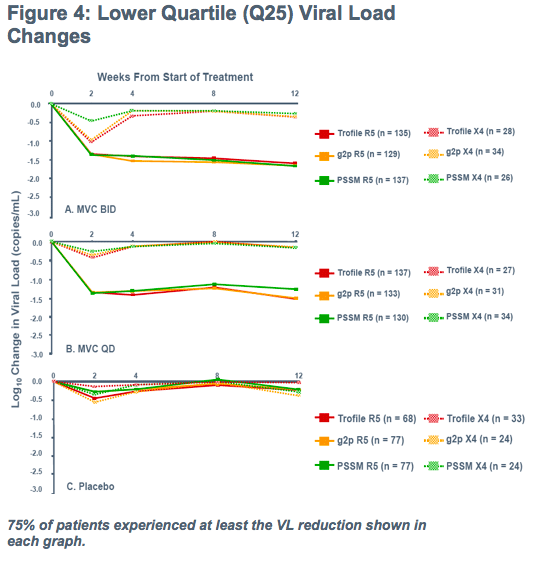
Initial endpoint of 8 week change in viral load
-Patients from both studies (MOTIVATE 1 and 1029) were followed for treatment outcome
-Allowing time for suppression with few drop-outs and few suppressed below 50 copies/mL
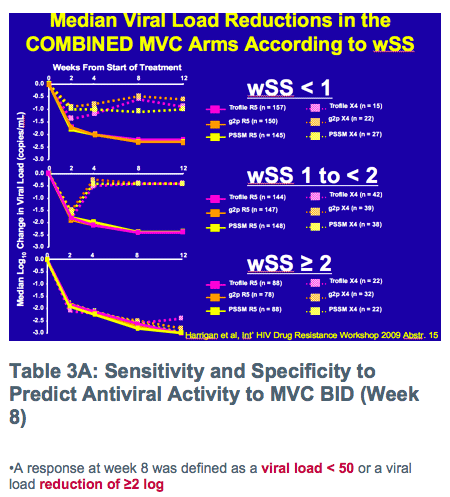
Weighted Sensitivity Score (wSS) of the background therapy
-Method to determine antiviral activity of background therapy according to susceptibility of the patient's HIV isolate to a givendrug and antiviral potency of the drug
-Drugs in continuous use from screening to the treatment phase of the trial were scored as 0
-Viral resistance was defined by phenotype only (Monogram) except by genotype for enfuvirtide (BC CfE)
-Active NRTIs score 0.5 each, other active agents score 1.0, inactive agents scored 0

|
| |
|
 |
 |
|
|