 |
 |
 |
| |
Resistance Mutations in the Viral Protease Alter
Bevirimat Resistance Patterns in vitro
|
| |
| |
Reported by Jules Levin
18th Intl HIV Drug Resistance Workshop
June 9-12
Ft Myers Florida
A Fun, NM van Maarseveen, REM Maas and M Nijhuis
Department of Medical Microbiology, University Medical Center Utrecht, Utrecht, The Netherlands
Background
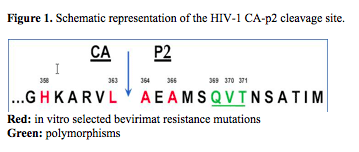
Bevirimat is the first maturation inhibitor in clinical trials. It inhibits HIV-1 replication by blocking viral protease-mediated cleavage of capsid/p2 (CA/p2). In vitro bevirimat resistance mutations were selected in the CA/p2 CS (Figure 1, aa 358, 363, 364, 366), indicated in red). In vivo studies revealed that naturally occurring polymorphisms in the QVT-motif (Figure 1, aa 369-371) just downstream of the CA/p2 CS, also impair bevirimat activity. Because of the functional interaction between the viral protease (PR) and the bevirimat target region, we investigated the impact of protease inhibitor (PI) resistance mutations on the selection of bevirimat resistance.
Methods:
We selected a panel of eight recombinant viruses containing PI resistance mutations in the viral protease and/or gag NC/p1 cleavage site and displaying a broad range of replicative capacities (RC). Multiple in vitro selections were performed in which viruses were cultured in SupT1 cells in the presence of increasing bevirimat concentrations. After five passages complete gag and protease were sequenced.
Results
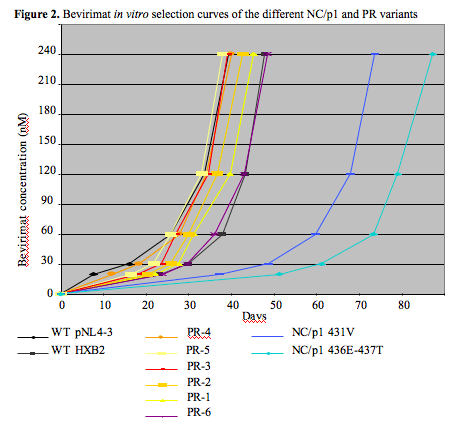
The rate of bevirimat resistance development appeared not to be affected by resistance mutations in the viral protease or differences in viral RC. However, the emergence of bevirimat resistance was delayed in the context of NC/p1 mutations only (Figure 2).

Results
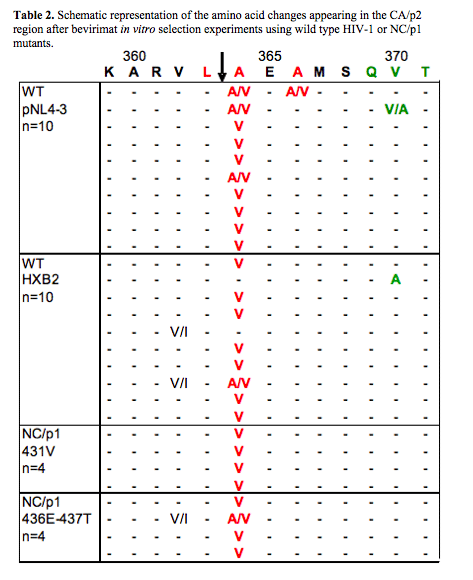
A difference was observed in the mutations selected when comparing wild type (HXB2 and pNL4-3) and gag CS variants with viruses harbouring PI resistant proteases. Whereas both wild type and gag CS variants almost exclusively selected for resistance mutations in the CA/p2 CS (A364V) (|Table 2), the mutational pattern in viruses with PI resistant proteases was far more diverse (Table 3). Particularly, mutations in the QVT-motif were significantly more selected in viruses with protease mutations. Until now, these mutations have only been described as naturally occurring polymorphisms. This study clearly demonstrates that QVT substitutions can also become selected under bevirimat pressure, especially in the presence of PI resistant proteases.
Summary
This study shows that the rate of selection of bevirimat resistance is unaffected by PI resistance mutations. However, PI mutations in the viral protease shift the bevirimat resistance pathway towards selection of mutations in the QVT-motif. These data, suggest a strong interaction between HIV protease and the gag QVT-motif, affecting bevirimat susceptibility.
Acknowledgements
This work was financially supported by European Union grant LSHP-CT-2007-037693.
|
| |
|
 |
 |
|
|