 |
 |
 |
| |
48-Week Results From the Phase 3 Study A4001026 (MERIT) -"Time to Loss of Virologic Response" Virology Analysis of Failures in the Enhanced Trofile®-Censored Subpopulation
|
| |
| |
Reported by Jules Levin
June 9-12
18th HIV Drug Resistance Workshop
Ft Myers Florida
Craig C1, Lewis M1, Simpson P1, Delogne C1, van der Ryst E1, Heera J2, Goodrich J2, Perros M1, and Westby M11. Pfizer Global Research & Development, Sandwich, England, UK; 2. Pfizer Global Research & Development, New London, CT, USA


INTRODUCTION
The MERIT study is an ongoing, randomized, double-blind clinical study designed to compare the safety and efficacy of maraviroc (MVC) 300 mg BID vs efavirenz (EFV) 600 mg QD, both administered with zidovudine/lamivudine (ZDV/3TC), in treatment-naive patients with R5 HIV-1 by the original Trofile® tropism assay (Monogram Biosciences)1
A retrospective re-analysis was performed using an enhanced sensitivity Trofile assay (ESTA), the version of Trofile currently available2
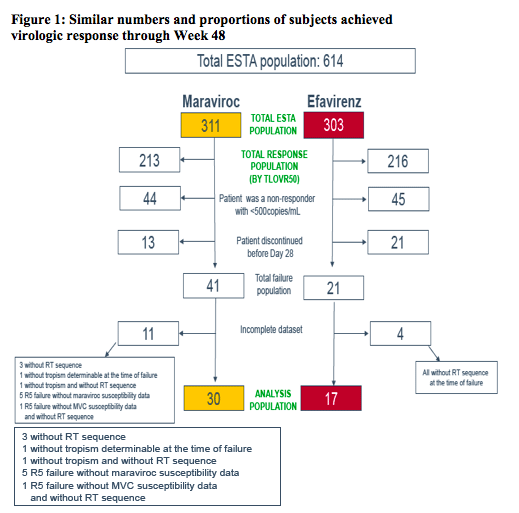
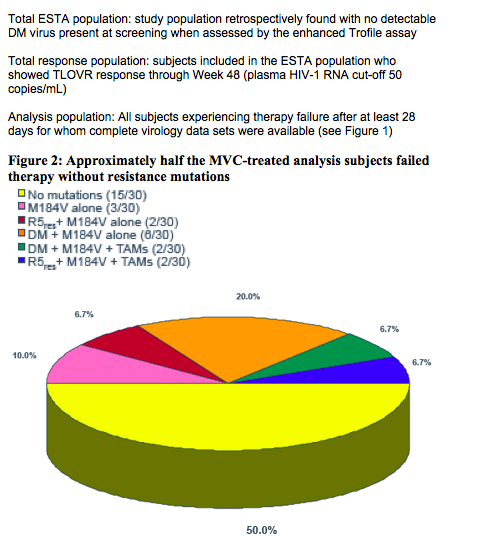
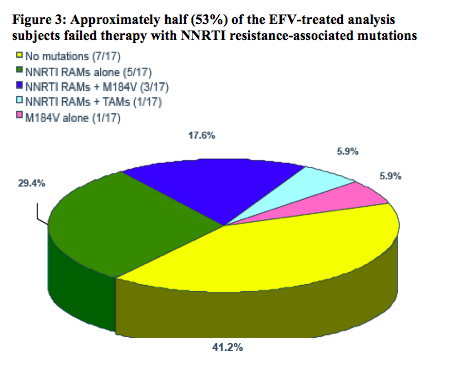
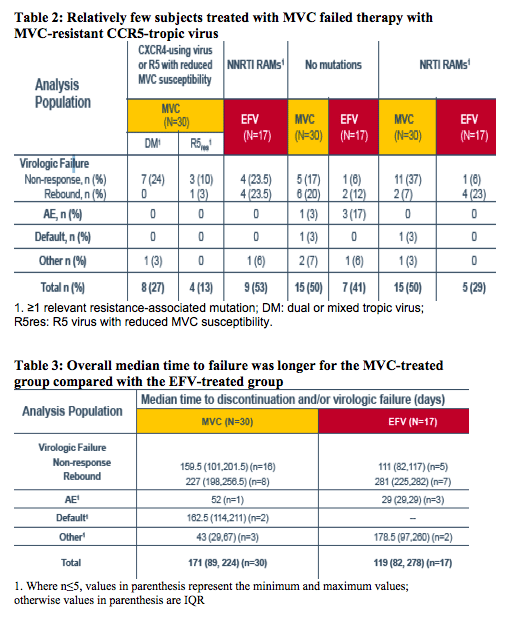
In the current virologic analysis, plasma samples from subjects experiencing failure, as assessed using the time to loss of virologic response (TLOVR50) algorithm through Week 48, were analyzed at the time of failure for resistance to any component of their regimens
METHODS
The 721 screening samples from MERIT patients enrolled on the basis of an R5 Trofile result (original assay) were re-tested with the ESTA
- The Monogram Biosciences' Clinical Reference Laboratory performed the enhanced Trofile assay using stored env expression vector pools from the original amplifications
- No clinical outcome information from the MERIT study was available to Monogram Biosciences
Failure samples were selected for analysis according to the TLOVR50 algorithm, the duration of therapy (≥28 days), and the presence of plasma HIV-1 RNA >50 copies/mL at 48 weeks or at the time of the last active dose
- Failures for non-virologic reasons were included
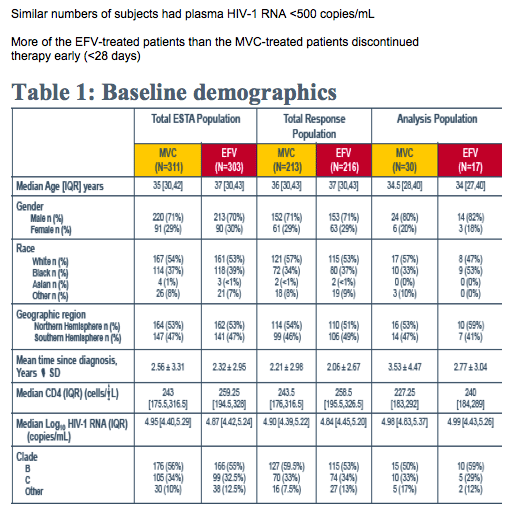
Analysis was performed at Baseline and at the time of virologic failure:
-- PhenoSenseGT® was performed to assess the genotype and phenotype of EFV and of the background regimen (ZDV and 3TC) copies/mL) resistance-associated mutations described in the IAS USA tables3were used.
-- The original Trofile assay result was used to assign tropism to the Baseline timepoint and at the latest on-treatment assessment on or before time of failure
-- Susceptibility to MVC in virologic failure with CCR5-tropic virus was assessed using the PhenoSense Entry® assay
-- Analysis could be performed when plasma HIV-1 RNA concentration was >500 copies/mL

|
| |
|
 |
 |
|
|