 |
 |
 |
| |
Long-term Outcomes Following Combination Treatment with Boceprevir plus PegIntron/Ribavirin (P/R) in Patients with Chronic Hepatitis C, Genotype 1 (CHC-G1)
|
| |
| |
Reported by Jules Levin
EASL Apr 14-18 2010
Vienna Austria
J. Vierling1, R. Ralston2, E. Lawitz3, J. McCone4, S.Gordon5, D. Pound6, M. Davis7, J.Galati8, I. Jacobson9, L. Rossaro10, F. Anderson11, J. King12, W. Cassidy13, M. Bourliere14, R. Esteban-Mur15, N. Ravendhran16, G. Galler17, P. Mendez2, C. Brass2, J. Albrecht2
1Advanced Liver Therapies/St. Luke's Episcopal Hospital, Houston, TX; 2Merck Research Laboratories, Kenilworth, NJ; 3Alamo Medical Research, San Antonio, TX; 4Mount Vernon Endoscopy Center, Alexandria, VA; 5Henry Ford Hospital, Detroit, MI; 6Indianapolis Gastroenterology Research Foundation, Indianapolis, IN;
7South Florida Center of Gastroenterology, Wellington, FL; 8Liver Specialists of Texas, Houston, TX; 9Weill Medical College of Cornell University, New York, NY; 10Davis Medical Center, University of California, Sacramento, CA; 11The Liver and Intestinal Research Centre, Vancouver, Canada;
12LSU Medical Center; Louisiana State University, Shreveport, LA; 13Louisiana State University, Baton Rouge, LA; 14Hospital Saint Joseph, Marseille Cedex, France; 15Hospital Vall d'Hebron, Barcelona, Spain; 16Digestive Disease Associates, Baltimore, MD; 17Kelsey Research Foundation, Houston, TX

Abstract
Background: Combination treatment of CHC-G1 with boceprevir, an inhibitor of the HCV NS3 serine protease, leads to high rates of sustained virologic response (SVR). The durability of SVR and the natural history of resistance mutations in patients not responding to therapy are unknown. To characterize long-term outcomes, patients who
participated in boceprevir trials are being monitored for 3 years posttherapy.
Methods: We evaluated samples from 604 patients (treatment-naïve or P/R failures) who received boceprevir plus PegIntron ± ribavirin in phase I/II trials. HCV-RNA was detected using Roche Taqman (LLQ= 29 IU/mL). Resistance mutations were detected by population sequencing of the NS3 protease region (codons 1-181) by Virco BBVA (Belgium) and/or by sponsor. Kaplan-Meier analysis was used to compare the loss
of detectable resistance mutations in the NS3 1-181 sequence.
Results: No late relapse was confirmed in the 290 patients who previously had SVR. A total of 25 serious adverse events (SAEs) occurred, and all were similar to those previously described in longterm follow-up. An additional 172 patients who did not achieve SVR were followed for 32 years, from the naïve trial (38/373; 16%) and from
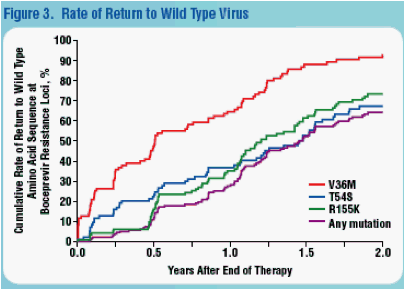
the treatment failure trial (134/226; 59%) in which no patient received the current treatment regimen. We identified 18 boceprevir resistant mutations in the following proportions: R155K, 64%; T54S, 54%; V36M, 54%; T54A, 22%; others <9% each. The rate of return to wild type NS3 1-181 sequence was higher for patients with V36M compared to those with T54S or R155K.
Conclusion: SVR following boceprevir combination therapy was durable. There were no latent SAEs or laboratory abnormalities related to prior treatment. Specific resistance mutations to boceprevir declined at different rates, V36M declined fastest; T54S and R155K declined at similar rates. Variation in the rate of mutational decline likely reflects the relative fitness of the mutants.
Background
· Treatment with boceprevir, an inhibitor of the HCV NS3 serine protease, in combination with peginterferon alfa-2b and ribavirin (RBV) has been shown to be associated with significantly higher SVR rates than peginterferon alfa-2b + RBV alone for patients with chronic hepatitis C infection (CHC-G1).
· Little is known about the durability of the virologic response and long-term safety of this therapeutic regimen.
· A number of resistant mutations have been identified both in vitro and in vivo.1 However, the clinical significance of a greater proportion of such variants, and the effect of stopping therapy upon viral rebound are not well established.
Objectives
· Confirm the durability of the virologic response in CHC-G1 patients with sustained virologic response at Week 24 (SVR24) after end of treatment (EOT)
· Characterize the long-term safety in patients who received at least one
dose of boceprevir
· Characterize the natural history of HCV sequence variants in patients who
received at least one dose of study medication

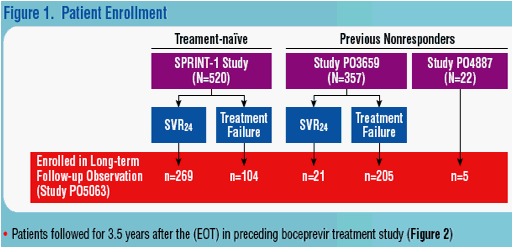

RESULTS
Patients
· Demographics of patients at their initial enrollment in previous treatment studies are listed in Table 1
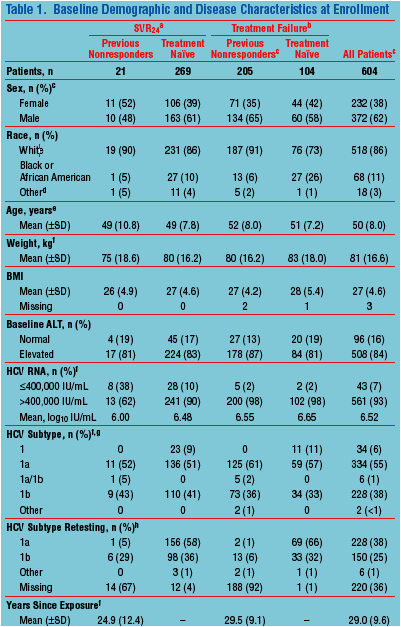
BMI, body mass index.
a SVR24 in the previous treatment protocol.
b Treatment failure in the previous treatment protocol.
c Five patients who previously participated in P04487 are counted in the "All Patients" column but not in other columns.
d American Indian, Alaskan Native, Asian, Pacific Islander, or Multiracial.
e On entry into long term follow-up (P05063).
f On entry into the previous treatment protocol.
g Determined by sequencing of the 5'UTR for P03659 and TRUGENE HCV 5'NC assay for P03523.
h Determined by Virco assay based on sequencing of domain 329bp in the NS5B polymerase gene; all samples unavailable for retesting were classified as missing.
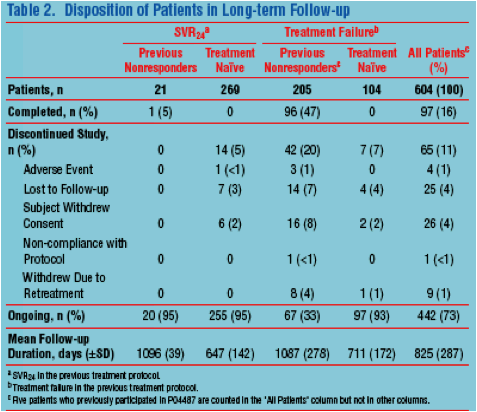

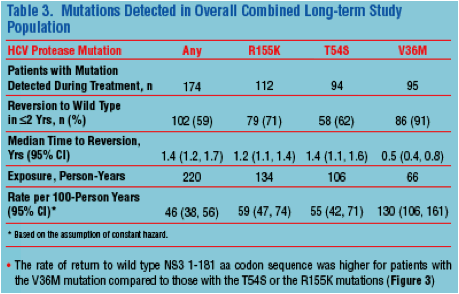


|
| |
|
 |
 |
|
|