 |
 |
 |
| |
On-Treatment Response-Guided Therapy with Telaprevir q8h or q12h Combined with Peginterferon Alfa-2a or Alfa-2b and Ribavirin in Treatment-Naïve Genotype 1 Hepatitis C (Study C208)
|
| |
| |
Reported by Jules Levin
EASL Apr 14-18 2010 Vienna Austria
Xavier Forns1, Patrick Marcellin2, Peter Ferenci3, Tobias Goeser4, Frederik Nevens5, Giampiero Carosi6, Joost P Drenth7, Lawrence Serfaty8, Koen De Backer9, Rolf van Heeswijk9, Don Luo10, Gaston Picchio10, Maria Beumont9
1Liver Unit, Hospital Clinic, IDIBAPS, Ciberehd, University of Barcelona, Barcelona, Spain; 2Hopital Beaujon, Clichy, France; 3Department of Internal Medicine 3, Medical University of Vienna, Vienna, Austria; 4Klinikum der Universitat zu Koln, Koln, Germany; 5Department of Hepatology, University Hospital Gasthuisberg, Leuven, Belgium; 6Clinic of Infectious and Tropical Diseases, University of Brescia, Brescia, Italy; 7Radboud University Nijmegen Medical Center, Nijmegen, Netherlands; 8Hopital Saint Antoine, Paris, France, 9Tibotec BVBA, Mechelen, Belgium; 10Tibotec Inc., Titusville, NJ, USA



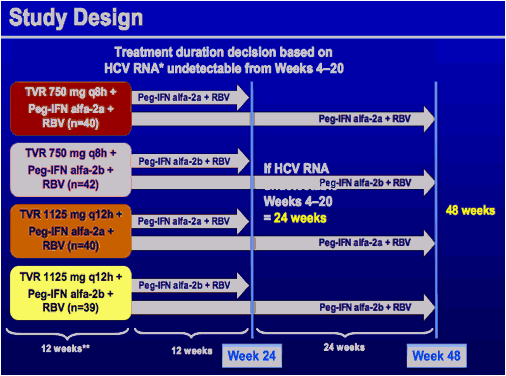
*HCV COBAS TaqMan HPS v2.0 assay (Roche Molecular Systems)
**TVR + Peg-IFN alfa-2a (180 µg/wk) + RBV (1000-1200 mg/d), or TVR + Peg-IFN alfa-2b (1.5 µg/kg/wk) + RBV (800-1200 mg/d). All patients were followed during 24 weeks after end of treatment to assess SVR

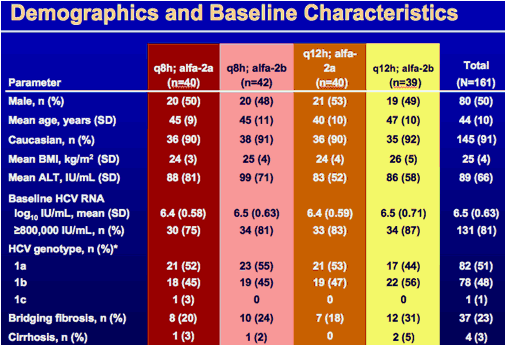
*Determined by the HCV NS3 genotyping assay
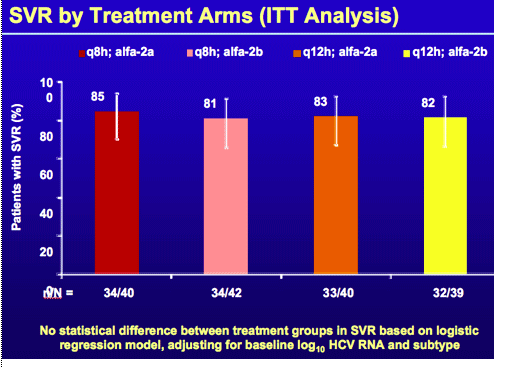
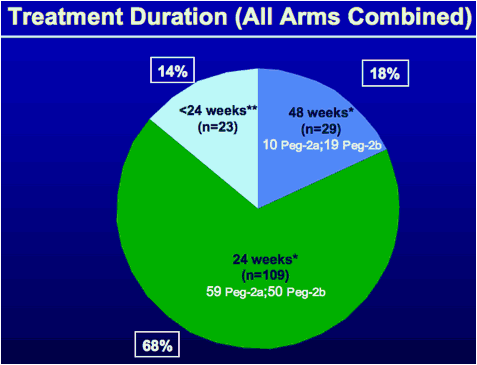
*Based on the assigned intention to treat. Some patients discontinued earlier
**23 pts discontinued <24 weeks: 8 for virological reasons,11 due to adverse event (AE), 4 due to other reasons
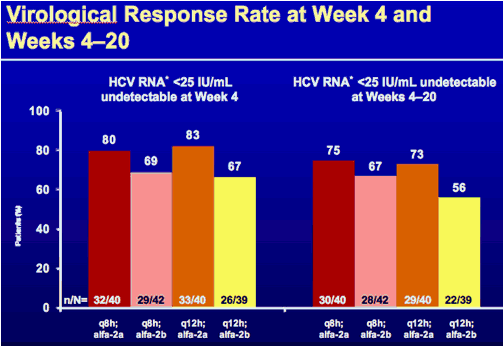
*HCV RNA was measured with the HCV COBAS TaqMan HPS v2.0 assay (Roche Molecular Systems) with a limit of quantification (LOQ) of 25 IU/mL. If no HCV RNA signal was detected below the LOQ, results are reported as < 25 IU/mL undetectable
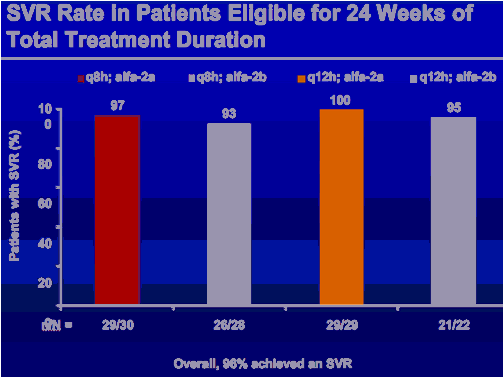
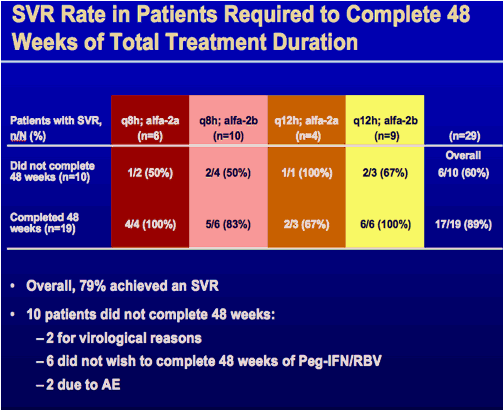
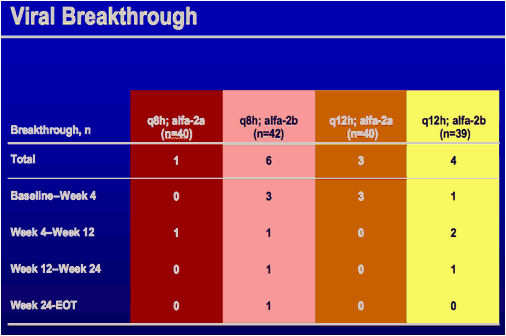
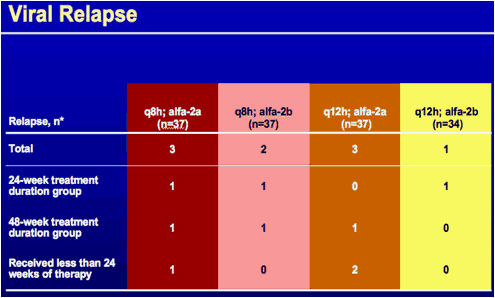
*Subjects with undetectable HCV RNA at end of treatment
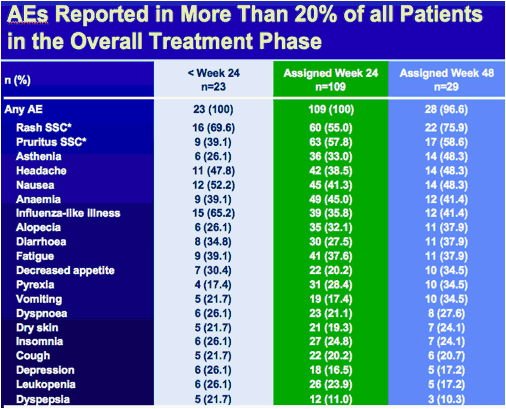
*SSC: special search category
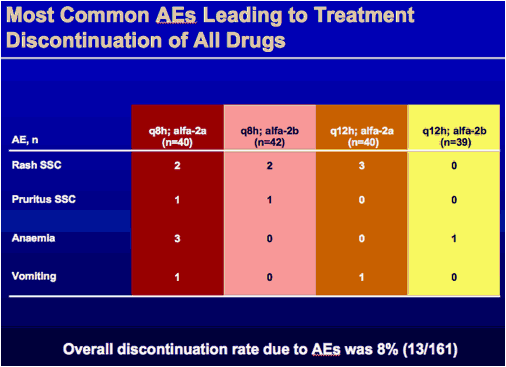
|
| |
|
 |
 |
|
|