 |
 |
 |
| |
IMPACT OF LOW-DOSE RITONAVIR BOOSTING ON THE PHARMACOKINETICS OF DANOPREVIR (RG7227; ITMN-191), A HIGHLY POTENT AND SELECTIVE INHIBITOR OF THE HCV NS3/4A PROTEASE
|
| |
| |
Reported by Jules Levin
EASL Apr 14-18 2010 Vienna Austria
Joshua Haznedar,1 Jennifer Fretland,2 Gilbert Leong,2 Steven Blotner,3 Todd Hill,4 Patrick Smith,1 and Jonathan Tran1
1Roche, Clinical Pharmacology, South San Francisco, CA, USA; 2Roche Palo Alto LLC, Department of Drug Metabolism and Pharmacokinetics, Palo Alto, CA, USA; 3Hoffmann-La Roche Inc., Pharma Development Innovation Biomathematics, Nutley, NJ, USA; 4Hoffmann-La Roche Inc., Clinical Research and Exploratory Development Operations, Nutley, NJ, USA
Author Discussion
CYP3A-mediated inhibition by low-dose RTV has been successfully utilised in the HIV treatment paradigm to simplify HIV protease inhibitor regimens.
The main objective of this pilot PK study in healthy volunteers was to determine if low-dose RTV could 'boost' the PK of danoprevir. Low-dose RTV significantly enhances danoprevir C12h (representative of Cmin for a q12h dosing regimen),
which appears to be the key parameter driving efficacy and prevention of resistance. Less of an effect was observed on AUC and Cmax, which are key parameters often attributed to safety.
The effect of multiple doses of RTV 100 mg q12h x 10 days on danoprevir C12h is about 2-fold less than the effect of a single dose of RTV 100 mg. This is likely due to the mixed inhibition and induction effects of RTV on CYP enzymes.[4]
Based on the PK results, a phase Ib study of RTV-boosted danoprevir in combination with standard of care in HCV-infected patients was conducted.[5] Low doses and dosing frequency of danoprevir (100 mg and 200 mg; q12h and q24h) were chosen for co-administration with RTV 100 mg to achieve lower danoprevir Cmax and AUC than unboosted high doses of danoprevir while maintaining optimal Cmin for efficacy.
Author Conclusions
PK boosting of danoprevir by low-dose RTV appears feasible. Danoprevir dose and dosing frequency may be reduced when co-administered with low-dose RTV.
The co-administration of danoprevir single dose and low-dose RTV was safe and well tolerated in healthy volunteers.
Introduction
RG7227 (ITMN-191; danoprevir) is a potent inhibitor of the HCV serine protease enzyme encoded by the nonstructural gene 3/4A (NS3/4A).
Danoprevir is currently being evaluated in phase II trials (300 mg q8h and 600 mg q12h) in combination with standard of care in patients with chronic hepatitis C genotype 1.
Ritonavir (RTV), a potent inhibitor of cytochrome P450 3A (CYP3A), is an HIV protease inhibitor that is mostly used at low, sub-therapeutic doses (i.e. 100-400 mg/day) to enhance or 'boost' the exposures of other HIV protease inhibitors that are extensively metabolised by the CYP3A isoenzyme (e.g. lopinavir/RTV, tipranavir/RTV, darunavir/RTV.[1]
Enhancement of HIV protease inhibitor pharmacokinetics (PK) via RTV boosting enables regimen simplification, improves treatment response and reduces the emergence of resistance.[2]
Danoprevir metabolism is mediated primarily by CYP3A and is likely to be inhibited by RTV.[3] Therefore, this pharmacological strategy to simplify the dosing regimen for danoprevir using low-dose RTV is being explored.
Objective
To evaluate the effects of single and multiple doses of RTV on the PK of danoprevir in healthy subjects
Methods
The study was designed as a single-centre, open-label, fixed-sequence PK study.
Subjects were screened for participation within 21 days of dosing. Subjects were admitted to the study unit on Day -1 (the day before dosing initiation) and discharged on Day 14.
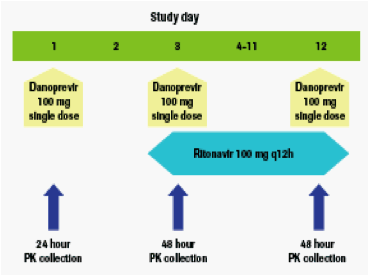
All subjects received a single dose of danoprevir 100 mg at three separate occasions: alone (Day 1), with the first dose of RTV 100 mg q12h (Day 3), and with the Day 12 dose of RTV 100 mg q12h. All study medications were administered with a meal. The dosing scheme and PK collections are illustrated in Figure 1.
Figure 1. Dosing scheme
Non-compartmental analysis using WinNonlin (Version 5.2.1; Pharsight Inc., Mountain View, CA, USA) was used to estimate danoprevir PK parameters. Analysis of variance (ANOVA) was used to analyse primary PK parameters.
Primary inclusion/exclusion criteria
· Healthy, non-smoking males or females aged between 18 and 45 years with a body mass index (BMI) between 18 and 30 kg/m2.
· Able to participate and willing to give written informed consent and to comply with the study restrictions.
· Use of any medications (prescription or over the counter), within 2 weeks (4 weeks for metabolic enzyme inducers) or five half-lives (whichever is longer) before the fi rst dose of study medication was excluded.
· Consumption of any other nutrients known to modulate CYP enzyme activity (e.g. products containing grapefruit or Seville orange) within 7 days prior to administration of study medication was excluded.
Results
Fourteen subjects were enrolled and received at least one dose of danoprevir and were included in the safety analysis.
Twelve of the 14 subjects enrolled completed the study and provided PK data for analysis. Two subjects were prematurely withdrawn due to inability to comply with the study requirements (meal restrictions).
Baseline characteristics of the 14 subjects enrolled and included in the safety analysis are described in Table 1.
Table 1. Baseline characteristics

Pharmacokinetics
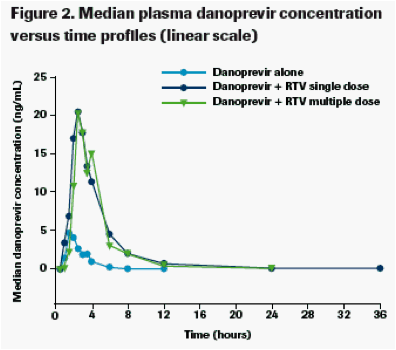
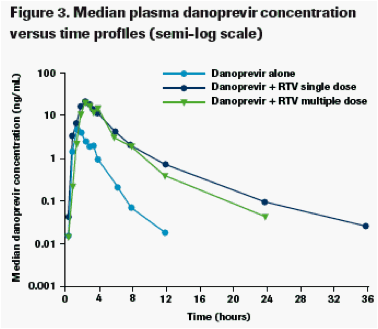
Single-dose (Day 3) and multiple-dose (Day 12) RTV 100 mg q12h substantially increased danoprevir exposure as shown by the median plasma concentration versus time profiles (Figures 2 and 3).
Geometric least squares mean (GLSM) of AUC0-∞, Cmax and C12h together with the 90% confidence intervals (CI) are summarised in Table 2.
Co-administration with single-dose RTV (100 mg) resulted in approximately 6-fold increase in danoprevir AUC0-∞,
3-fold increase in Cmax and more than a 50-fold increase in C12h (Table 2).
Multiple dosing of RTV 100 mg q12h x 10 days resulted in approximately 5-fold increase in danoprevir AUC0-∞, 3-fold
increase in Cmax and a less pronounced 27-fold increase in C12h (Table 2).
Results were similar, when data from four subjects with unquantifiable C12h values on Day 1 were included in the
analysis using extrapolated danoprevir C12h values (Table 2).
Median danoprevir Tmax values were comparable between treatments. Mean danoprevir terminal half-life (t1/2) was
approximately 2- to 3-fold longer in presence of RTV than when dosed alone.
Table 2. Statistical analysis of danoprevir PK parameters after single-dose administration alone (Day 1), with single-dose RTV (100 mg) (Day 3), and with multiple-dose RTV (100 mg q12h x 10 days) (Day 12)
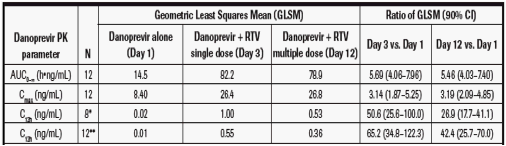
AUC0-∞ = area under the plasma concentration versus time curve from time zero extrapolated to time infi nity; Cmax = observed maximum plasma
concentration; C12h = observed plasma concentration at 12 hours post-dose
* Includes data only from eight subjects who had quantifi able C12h in all treatments
** Includes extrapolated C12h for four subjects on Day 1
Safety
Overall danoprevir plus RTV was well tolerated. The most frequently reported adverse events among subjects receiving danoprevir plus RTV were diarrhoea (4/14) and headache (3/14), which were of mild intensity.
There were no clinically significant changes in laboratory safety tests, vital signs or echocardiograms and there were no deaths, no reports of serious adverse events and no adverse events that led to trial treatment withdrawal.
References
1. Hammer SM, Eron JJ, Reiss P, et al. Antiretroviral treatment of adult HIV infection 2008 recommendations of the international AIDS society-USA panel. JAMA 2008;300:555-570.
2. Scott JD. Simplifying the treatment of HIV infection with ritonavir-boosted protease inhibitors in antiretroviral-experienced patients. Am J Health Syst Pharm 2005;62:805-15
3. Roche data on fi le.
4. Foisy MM, Yakiwchuk EM, and Hughes CA. Induction Effects of Ritonavir: Implications for Drug Interactions. Ann Pharmacother 2008;42:1048-59
5. Gane E, Rouzier R, Stedman C, et al. Ritonavir boosting of low dose RG7227/ITMN-191, HCV NS3/4A protease inhibitor, results in robust reduction in HCV RNA at lower exposures than provided by unboosted regimens. EASL April 14-18 2010 Vienna, Austria
|
| |
|
 |
 |
|
|