 |
 |
 |
| |
Early Viral Response Of Controlled-release Interferon Alpha2b And Ribavirin Vs. Pegylated-interferon Alpha2b And Ribavirin In Treatment-naïve Genotype-1 Hepatitis C: 12 Week Results (SELECT-2 Trial)
|
| |
| |
Reported by jules Levin
EASL Apr 14-18 2010 Vienna Austria
E. Lawitz1, Z.M. Younossi2, P. Mehri3, A. Rigney4, Z. Krastev5, K. Tchernev6, D. Takov7, W. A. Long4
1Alamo Medical Research, San Antonio,Texas, 2Inova Health System, Falls Church, Virginia, 3eStudy, Chula Vista, California, 4Biolex Therapeutics, Pittsboro, North Carolina,
5UMHAT "St. Ivan Rilski", Sofia, Bulgaria, 6UMHAT "Alexandrovska", Sofia, Bulgaria, 7Military Medical Academy, Sofia, Bulgaria

ABSTRACT
Background and Aim: A microsphere-based controlled-release formulation of
interferon alpha2b (CR2b) designed to reduce dosing frequency and side effects is being investigated. The aim of this study was to compare efficacy and safety of CR2b dosed q2weeks to pegylated interferon alpha2b (PEG2b) dosed weekly in treatment-naïve genotype1 (G1) HCV patients treated with weight-based ribavirin.
Methods: In this 72-week trial, 116 G1 treatment-naïve patients were randomized to CR2b [Locteron, Biolex Therapeutics, Pittsboro, NC, USA] 640 ug, 480 ug, 320 ug q2weeks (blinded to CR2b dose) or weekly PEG2b [PegIntron, Schering Plough, Kenilworth, NJ, USA] all in combination with weight-based ribavirin. 86 subjects received CR2b (640 ug, n=29; 480 ug, n=29; or 320 ug, n=28); 30 subjects received PEG2b. HCV RNA was measured weekly for five weeks, every other week for another seven weeks, and every three months thereafter. Adverse events including flu-like symptoms (FluSxs) were collected during weekly clinic visits for 12 weeks and monthly thereafter. FluSxs were also collected daily for 12 weeks by patient selfreport using the internet (ePRO).
Results: Results from a planned interim analysis through Week 12 are available. RVR rates (%) were 14,3,4,13 respectively for q2week 640 ug, 480 ug, 320 ug CR2b and for weekly PEG2b. EVR rates (%) were 59,62,68,60 respectively for q2week 640 ug, 480 ug, 320 ug CR2b and for weekly PEG2b.
FluSxs (arthralgia, chills, fever, headache, or myalgia reported at weekly clinic visits) occurred 64,65,56,126 times respectively on q2week 640 ug, 480 ug, 320 ug CR2b and weekly PEG2b respectively. (Daily ePRO data for FluSxs remain blinded.) Neutrophil counts < 750 occurred (%) in 14,21,11,10 respectively for q2week 640 ug, 480 ug, 320 ug CR2b and for weekly PEG2b; no neutrophil counts < 500 were reported in the first 12 weeks. Subjects with neutropenia had the same overall rate of EVR as subjects without neutropenia.
Conclusions: CR2b dosed q2weeks provided antiviral efficacy equivalent to weekly PEG2b. FluSxs were half as frequent on CR2b; modest neutropenia occurred more commonly on CR2b but the neutropenia did not interfere with achievement of EVR. Final 12-week results including ePRO will be presented at the meeting.
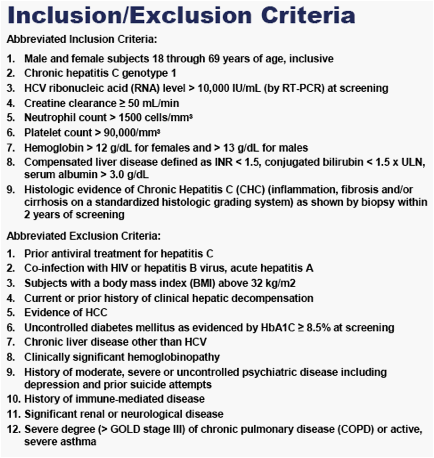
INTRODUCTION & BACKGROUND
Successful treatment of chronic genotype 1 hepatitis C, defined as sustained viral response (SVR) 24 weeks after completion of active treatment, remains challenging. Standard of care (SOC) currently a) requires 48 weeks of treatment with daily oral weight-based ribavirin and weekly subcutaneous pegylated interferon alpha (IFNa) 2a or 2b, b) carries significant side effects, and c) provides sub-optimal SVR rates (<50%).
Recent advances including identification of the importance of the IL28b gene in
treatment response, and the ongoing developments of direct acting antiviral agents as add-ons to SOC, show much promise for achieving both shorter (response-guided) treatment durations and higher SVR rates in the near future in chronic genotype 1 hepatitis C.
Regardless of when the anticipated improvements in the current treatment paradigm for chronic genotype-1 hepatitis C occur, and regardless of what form those improvements take, IFNa will remain part of the backbone of chronic HCV treatment for the foreseeable future. As a result, improvements in both the dosing schedule and side-effect profile of IFNa remain worthwhile goals.
We have been investigating a microsphere-based controlled-release formulation of interferon alpha2b (CR2b) (Locteron, Biolex Therapeutics, Pittsboro, N. Carolina, USA) designed to provide an improved pharmacokinetic profile that is anticipated to permit a) reduced dosing frequency (q2weeks), b) the same efficacy, and c) reduced flu-like symptoms.
The primary objective of this study (SELECT-2) was to assess the virologic response to three dose levels of CR2b, dosed every two weeks, in comparison with pegylated IFNa2b (PEG2b) (PEG-Intron, Schering Plough, Kenilworth, New Jersey, USA) dose weekly, in treatment-naïve, genotype-1subjects with chronic hepatitis C receiving weight-based doses of ribavirin.
Secondary objectives of SELECT-2 included assessments of:
- the safety, tolerability and immunogenicity of three dose levels of CR2b
compared to that of PEG2b including the intensity and duration of flu-like
symptoms (i.e. fever, chills, myalgia, arthralgia and headache) as measured by
ePRO and by clinic visit assessments
- the rates of dose reductions and of study drug discontinuations for tolerability
reasons of three dose levels of CR2b compared to that of PEG2b
- the pharmacokinetics of three dose levels of CR2b (IFNa2b level) and of PEG2b (pegylated IFNa2b levels)
- the impact on general health-related quality of life (HRQOL), as measured by SF-36 and HQLQ of three dose levels of CR2b and of PEG2b
- the impact on the onset of depression and depressive symptoms, as measured
by the Becks Depression Inventory (BDI) of three dose levels of CR2b and of
PEG2b
- the impact on days missed from work during treatment with each of three dose
levels of CR2b and of PEG2b
- the incidence of binding and neutralizing antibodies
Another important aim of SELECT-2 was to provide data (from the patients
randomized to 480 ug CR2b and to PEG2b in the trial) to support formal testing of the null hypothesis that flu-like symptoms are not different on the 480 ug dose of CR2b and on standard doses of PEG2b. To achieve the overall sample size of 122 patients (61 on CR2b, 61 on PEG2b) required to provide 85% power in testing this null hypothesis, another trial "480 STUDY" (also reported at this meeting) was also initiated. Together the two clinical studies (SELECT-2 and 480 STUDY) provide a prospectively-created combined population called EMPOWER (also reported at this meeting) designed specifically for testing of this hypothesis.
DESIGN
SELECT-2 is a randomized 72-week Phase 2b study designed to compare three doses of CR2b to the standard dose of PEG2b. The design of SELECT-2 is found in Figure 1 below. The dose of CR2b was double-blind for the first 12 weeks, but investigators and patients knew whether they were randomized to CFR2b or PEG2b throughout the trial.
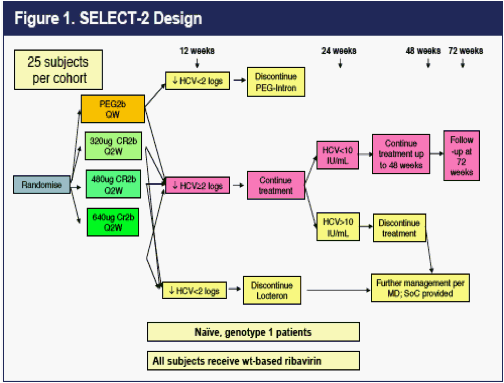
RESULTS
A total of 116 patients were randomized and dosed in SELECT-2, 57 at 14 sites in the US, and 59 at 10 sites in Bulgaria and Romania. The first patient was dosed in April 2009, and the last patient received his first dose in July 2009.
Interim efficacy and safety data as of April 4, 2010 from this ongoing trial are
presented in these analyses. As of that date, all patients continuing randomized treatment had completed the clinic visit required at 36 weeks treatment. The last patient will complete his final end of treatment visit at Week 48 in early June 2010.
The last Week 72 visit will occur in December 2010.
Pharmacokinetic data, data on immunogenicity, and ePRO data are not yet available.
DEMOGRAPHICS
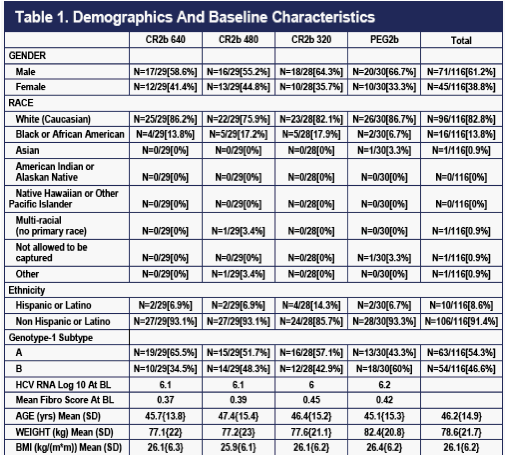
The four treatment groups were fairly well-balanced at baseline (Table 1). The mild imbalances present that may impact efficacy, such as those in race, sub-genotype, or viral load at baseline, appear to balance each other out.
EFFICACY
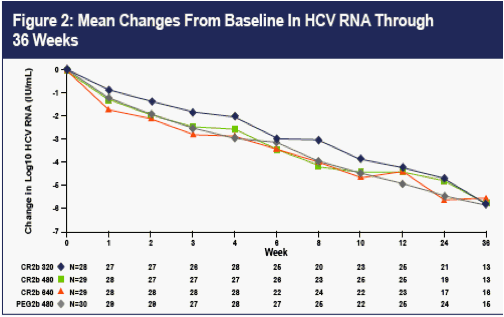
As can be seen from the graph of mean drops in viral load from baseline shown in Figure 2, the 480 ug and 640 ug doses of CR2b appeared to provide reductions in viral load equivalent to PEG2b; 320 ug CR2b appeared to be somewhat less effective.
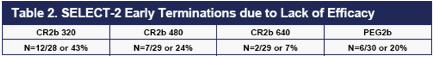
Consistent with these results, the 320 ug dose of CR2b had a higher rate of
discontinuations for lack of efficacy than the other three doses (Table 2).
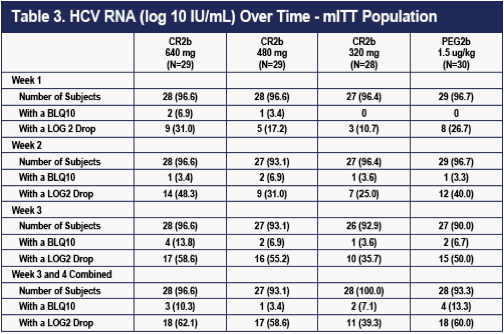
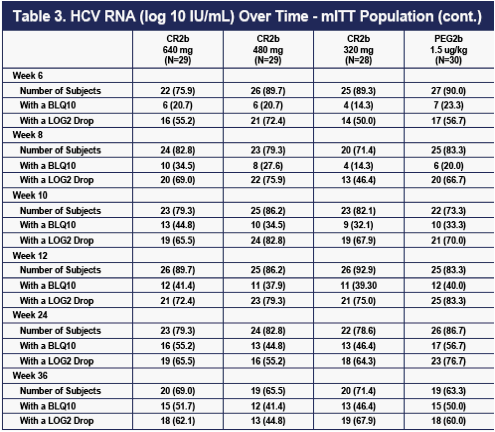
Serial rates of >2 log drops and of undetectable HCV RNA for the four groups are shown in Table 3 below. Despite the higher dropout rate for lack of efficacy on the 320 ug dose of CR2b shown in Table 2, all three doses of CR2b provided what appeared to be equivalent efficacy to that of PEG2b at Weeks 24 and 36.
Efficacy data from the end of treatment (Week 48) and 24 weeks post treatment (SVR) will be important in confirming the relative efficacy of 320 ug CR2b, 480 ug CR2b, 640 ug CR2b in combination with weight-based oral ribavirin versus PEG2b in chronic genotype-1 HCV in combination with weight-based oral ribavirin.
SAFETY
The overall safety of the four dose groups is summarized in Tables 4, 5, 6 and 7. No deaths occurred. Dropouts for any reason were somewhat higher on 320 ug CR2b; this difference was largely accounted for by an excess of dropouts for lack of efficacy on this dose. Dropouts due to AEs were equivalent in the four groups. Nearly all patients experienced at least one AE.
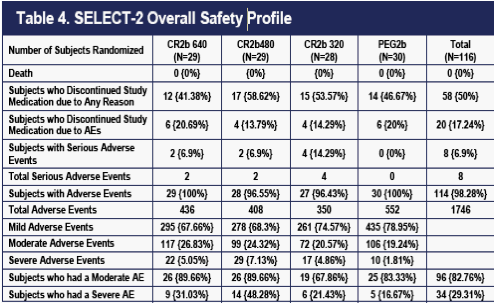
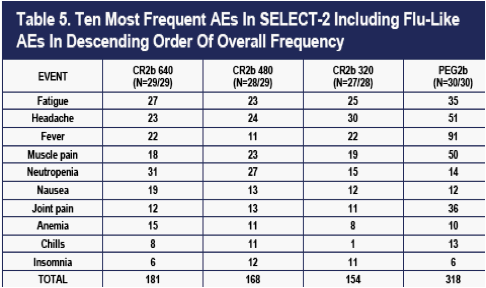
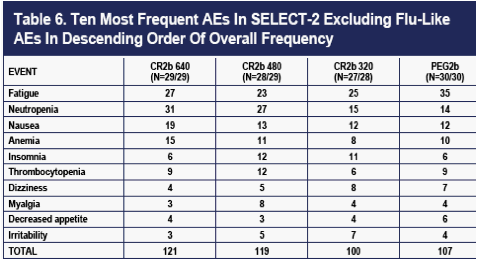
The overall number of AEs was highest in the PEG2b group (Table 4 and 5). The numbers of moderate AEs were equivalent in the four groups. Severe AEs occurred more frequently on CR2b, but the pattern was not dose-related. An excess of bone marrow effects comprised mostly of neutropenia accounted for the difference in severe AEs (data not shown). Patients with neutropenia had better viral outcomes (higher rates of EVR) than any of the four dose groups (data not shown), despite more frequent dose-reductions.
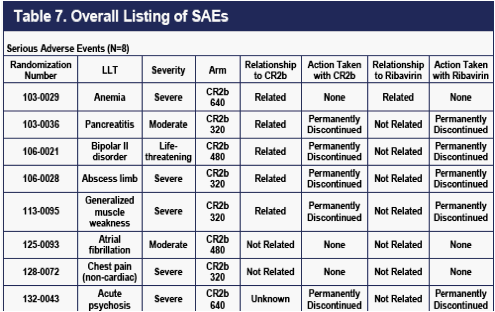
As of April 4, 2010, eight SAEs had occurred on CR2b. The absence of SAEs on
PEG2b is probably a chance finding, because the eight SAEs observed on CR2b are all different from each other (Table 7), are all labeled for IFNa2b, and did not occur in a dose-related pattern.
The one SAE characterized as life-threatening was what appeared to be an overdose gesture in a patient with suicidal ideation and a history of bipolar disease. Since the April 4, 2010 data cut, an additional SAE (dehydration) has been reported in a patient on PEG2b.
FLU-LIKE SYMPTOMS
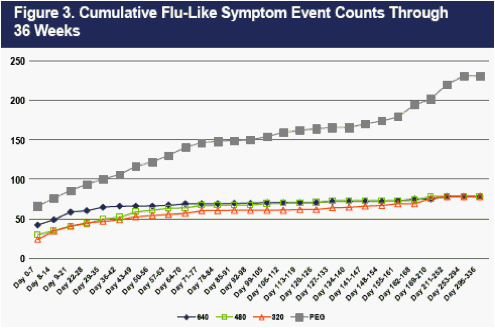
As shown in Figure 3, flu-like symptom events recorded at weekly clinic visits were less frequent on CR2b than on PEG2b, and the reductions were dose-related. Differences in flu-like symptom event counts were evident at Day 7- immediately prior to the second dose of IFNa in the PEG2b group, and well-prior to the second dose of IFNa in the CR2b groups (which occurred on Day 14).
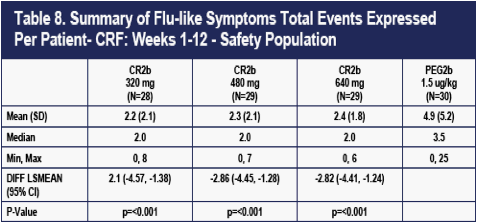
The differences in flu-like symptom event counts on the CR2b arms versus PEG2b present at Day 7 progressively increased thereafter during the 48-week dosing period, largely because flu-like events continued to accumulate on PEG2b with each dose. Flu-like events largely ceased in the three CR2b groups after about 6 weeks. By Week 36, 78, 78, 78 and 221 flu events had occurred in the 320 ug CR2b, 480 ug CR2b, 640 ug CR2b, and PEG2b groups respectively.
The differences in flu-like symptom counts in the first 12 weeks between the three CR2b arms and PEG2b were all highly statistically significant (Table 8).
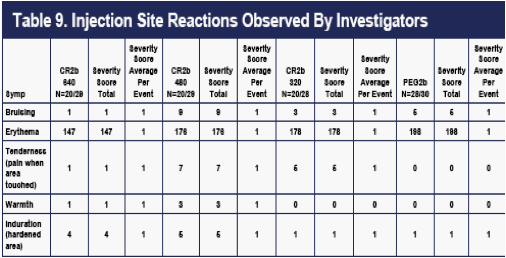
INJECTION-SITE REACTIONS
Injection site reactions (ISRs) as observed by the investigative staffs (Table 9), and as reported by the patients (Table 10), were also recorded at weekly clinic visits. ISRs as observed by investigators occurred in approximately 2/3 of the patients and were generally less frequent on CR2b than on PEG2b. An exception was induration (rare in all groups), which appeared to occur at higher rates on the 480 ug and 640 ug CR2b doses.
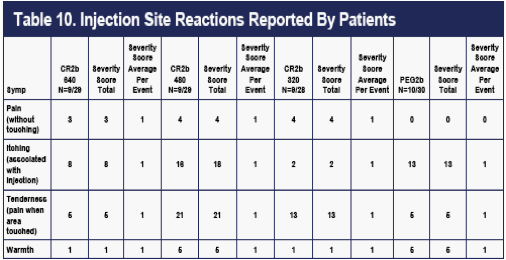
ISRs as reported by patients occurred in approximately 1/3 of the patients and were fairly evenly distributed among the dose groups.
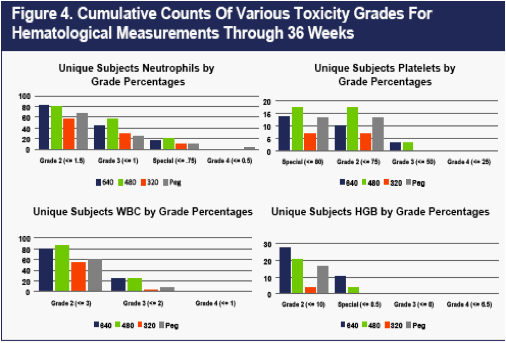
Laboratory Findings
The only noteworthy findings in the safety laboratory measurements occurred in the hematological assessments. As shown in Figure 4, higher rates of mild or moderate (CTCAEv3 Grade 2 and Grade 3) reductions in WBC, platelets, hemoglobin, and particularly neutrophils were observed on the two higher doses of CR2b in comparison to PEG2b, but Grade 4 reductions were not observed, with the exception of one neutrophil count <500 on PEG2b after Week 12.

Neutropenia during IFNa and ribavirin treatment of chronic HCV has recently been shown not only to predict a good viral response to treatment, but also to carry no increased risk of infection (1-3). In this study, the patients who experienced neutropenia had an EVR rate higher than that observed in any of the four treatment groups (data not shown).
|
| |
|
 |
 |
|
|