 |
 |
 |
| |
Q2week Controlled Release Interferon Alpha2b + Ribavrin Reduces Flu-like Symptoms >50% And Provides Equivalent Efficacy In Comparison To Weekly Pegylated Interferon Alpha2b + Ribavirin In Treatment-naïve Genotype-1 Chronic Hepatitis C: Results From EMPOWER, A Randomized Open-label 12-week Comparison In 133 Patients
|
| |
| |
Reported by Jules Levin
EASL Apr 14-18 2010 Vienna Austria
W.A. Long1*, D. Takov2, K. Tchernev3, I. Kotzev4, A. Rigney1, Z. Krastev5, S. Stoynov6, R. Balabanska7, E. Lawitz8, Z. Younossi9, R. Ghalib10, E. Zuckerman11, R. Safadi12, R. Tur-Kaspa13, N. Assy14, Y. Lurie15
1Biolex Therapeutics, Pittsboro, NC, USA, 2Military Medical Academy, 3UMHAT "Alexandrovska", Sofia, 4UMHAT "St. Marina", Varna, 5UMHAT "St. Ivan Rilski", 6UMHAT 'Queen Giovanna - ISUL' EAD, 7Tokuda Hospital, Sofia, Bulgaria, 8Alamo Medical Research, San Antonio, TX, 9Inova Health Systems, Falls Church, VA,
10The Liver Institute at Methodist Dallas, Dallas, TX, USA, 11Carmel Medical Center,, Haifa, 12Holy Family Hospital Nazareth, Nazareth, 13Rabin Medical Center, Petah-Tikva, 14Rebekah Ziv Medical Center, Zefat, 15Sourasky Medical Center, Tel Aviv, Israel. *wlong@biolex.com

ABSTRACT
Background and Aim: The aim of this study was to test the hypothesis that 480ug controlled-release-interferon-alpha2b (CR2b) dosed q2weeks reduces flu-like symptoms (FluSxs) but retains equal efficacy compared to pegylated interferon alpha2b (PEG2b) dosed weekly in treatment-naïve-genotype-1 (G1) chronic HCV patients treated with weight-based ribavirin.
Methods: EMPOWER is a 12-week, randomized, open-label Phase 2b trial designed with 85% power to detect a 50% reduction in flu-like symptoms in patients treated with 480ug CR2b [Locteron®, Biolex Therapeutics, Pittsboro, NC, USA] versus PEG2b [PegIntron®, Schering Plough, Kenilworth, NJ, USA], both in combination with weightbased ribavirin. Patients were enrolled in two contributing trials (SELECT-2 & 480STUDY) specifically designed to provide jointly the sample sizes required for the hypothesis test in EMPOWER. HCV RNA was measured weekly for three weeks and then every other week. Safety labs were measured weekly for three weeks and monthly thereafter. Adverse events including a pre-specified cluster of flu-like symptoms (FluSxs) were collected during weekly clinic visits for 12 weeks. FluSxs were also collected daily for 12 weeks by patient self-report using the internet (ePRO). Other measurements include serial measurements of BDI, HQLQ, SF-36, and days
missed from work.
Results: Preliminary results from 101/133 enrolled patients are available (n=48 on q2week-480ug-CR2b, n=53 on PEG2b). Mean declines in Log10 HCV RNA from baseline were equivalent. FluSxs (arthralgia, chills, fever, headache, or myalgia reported at weekly clinic visits) counts during the first week were 50 versus 109 on q2week-480ug-CR2b vs. PEG2b, and total FluSxs counts during the 12 weeks were 109 versus 314 on q2week-480ug-CR2b vs. PEG2b. Total days missed from work wer 82 versus 109 for q2week-480ug-CR2b versus PEG2b. Neutrophil counts < 750 occurred in 13% (6/48) and 8% (4/53) respectively for q2week-480ug-CR2b and PEG2b; no neutrophil counts < 500 were reported in the first 12 weeks.
Conclusions: In these 101 patients, q2week-480ug-CR2b over 12-weeks provided a) >50% reduction in FluSxs that preceded the possible impact of the q2week dosing interval, and b) equivalent antiviral efficacy. Days missed from work may also be reduced. Final 12-week results from all 133 patients (including ePRO) will be presented at the meeting.
EMPOWER:
EUROPEAN, ISRAELI, & US MULTICENTER, RANDOMIZED, PARALLEL, OPENLABEL STUDY WITH COMPARISON OF THE EFFECTS OF CONTROLLED-RELEASE INFa2b VERSUS PEGYLATED IFNa2b ON FLU-LIKE SYMPTOMS IN TREATMENTNAïVE, GENOTYPE-1 PATIENTS WITH CHRONIC HCV
INTRODUCTION & BACKGROUND
Successful treatment of chronic genotype-1 hepatitis C, defined as sustained viral response (SVR) 24 weeks after completion of active treatment, remains challenging. Standard of care (SOC) currently a) requires 48 weeks of treatment with daily oral weight-based ribavirin and weekly subcutaneous pegylated interferon alpha (IFNa) 2a or 2b, b) carries significant side effects, and c) provides sub-optimal SVR rates (<50%).
Recent advances including identification of the importance of the IL28b gene in
treatment response, and the ongoing developments of direct acting antiviral agents as add-ons to SOC, show much promise for achieving both shorter (response-guided) treatment durations and higher SVR rates in the near future in chronic genotype-1 hepatitis C.
Regardless of when the anticipated improvements in the current treatment paradigm for chronic genotype-1 hepatitis C occur, and regardless of what form those improvements take, IFNa will remain part of the backbone of chronic HCV treatment for the foreseeable future. As a result, improvements in both the dosing schedule and side-effect profile of IFNa remain worthwhile goals.
We have been investigating a microsphere-based controlled-release formulation of interferon alpha2b (CR2b) (Locteron®, Biolex Therapeutics, Pittsboro, N. Carolina, USA) designed to provide an improved pharmacokinetic profile that is anticipated to permit a) reduced dosing frequency (q2weeks), b) the same efficacy, and c) reduced flu-like symptoms.
OBJECTIVES
The primary objective of this combined analysis is to test the null hypothesis that flulike symptom severity scores are not different in treatment-naïve patients with genotype-1 chronic HCV randomized to 480 ug CR2b every two weeks versus those randomized to 1.5 ug PEG2b weekly, when both are given in combination with oral weight-based ribavirin. The hypothesis test will be useful in designing the Phase 3 program.
The secondary objective of this combined analysis is to characterize and compare the efficacy and safety profiles of 480 ug CR2b every two weeks versus 1.5 ug PEG2b weekly, both for 12 weeks, and both in combination with oral weight-based ribavirin in treatment-naïve patients with chronic genotype-1 HCV. These comparisons will also be useful in designing the Phase 3 program for Locteron in chronic HCV.
The tertiary objective of this combined analysis is to compare the practicality, utility and power of collecting of flu-like symptoms using daily hand-held ePRO devices versus the standard method of using patient recall at weekly clinic visits. This comparison, which has just begun, will also be useful in designing the Phase 3 program for CR2b in chronic HCV.
The specified minimum sample size of 122 patients required for EMPOWER was acquired in two trials (SELECT-2 and 480 STUDY) rather than a single trial because several other questions (including dose selection for Phase 3 and PK/PD relationships) needed to be addressed at the same time, and only finite time and resources were available. A minimum of 122 patients provides 85% power. Deriving the sample size required for the hypothesis test from two separate studies (SELECT-2 and 480 STUDY) permitted us to answer these other questions while we were accumulating the required patients for EMPOWER.
DESIGN
EMPOWER was prospectively designed as a combined analysis built from patients randomized to 480 ug CR2b or PEG2b in two different trials, SELECT-2 and 480 STUDY.
SELECT-2 is a randomized, multicenter, 72-week Phase 2b study designed to
compare three doses of CR2b to the standard dose of PEG2b. The design of SELECT- 2 is found in Figure 1 below. The dose of CR2b was double-blind for the first 12 weeks of SELECT-2, but investigators and patients knew whether they were randomized to CR2b or PEG2b throughout the trial.
Figure 1. SELECT-2 Design
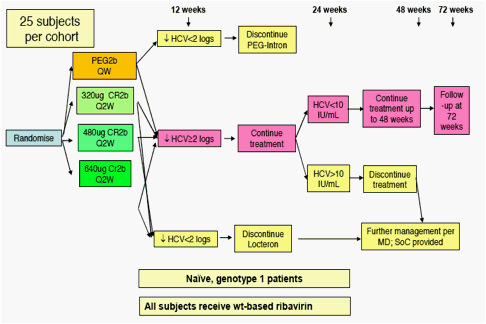
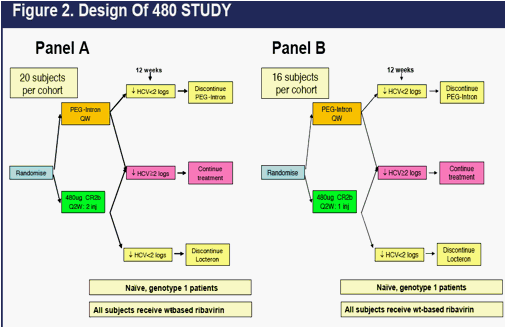
480 STUDY is a randomized, multicenter, 12-week, open-label, Phase 2b study
composed of two panels comparing the 480 ug dose of CR2b to the standard dose of PEG2b. The designs of 480 STUDY Panel A and 480 STUDY Panel B are found in Figure 2 below.

RESULTS
A total of 133 patients were randomized and dosed with 480 ug CR2b or PEG2b in SELECT-2 and 480 STUDY. A total of 30 sites participated in the two trials: 14 sites in the US, eight sites in Bulgaria, three sites in Romania, and five sites in Israel. The first patient was dosed in April 2009 in the United States, and the last patient received his first dose on February 9, 2010 in Israel.
Interim efficacy and safety data as of April 4, 2010 from this ongoing combined
analysis are presented below. As of that date, all patients from SELECT-2 and all patients from 480 STUDY Panel A had completed the Week 12 visit, and all patients from 480 STUDY Panel B had completed at least the Week 7 visit. The last patient from Panel B will complete his Week 12 visit on May 4, 2010.
Pharmacokinetic data, data on immunogenicity, data from days missed from work, and data from ePRO are not yet available. The results of the hypothesis test await completion of Panel B data and will be reported later.
DEMOGRAPHICS
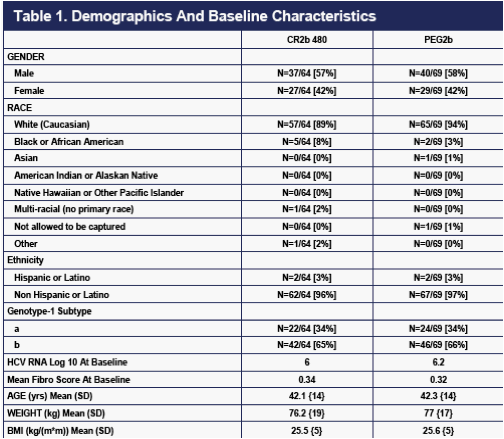
The two treatment groups were fairly well-balanced at baseline (Table 1). The mild imbalances present that may impact efficacy, such as those in race, genotype-1 subtype, or viral load at baseline, appear to balance each other out.
EFFICACY
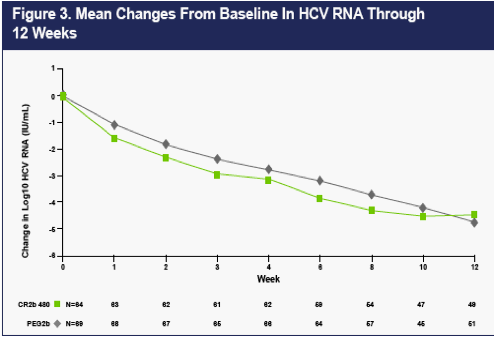
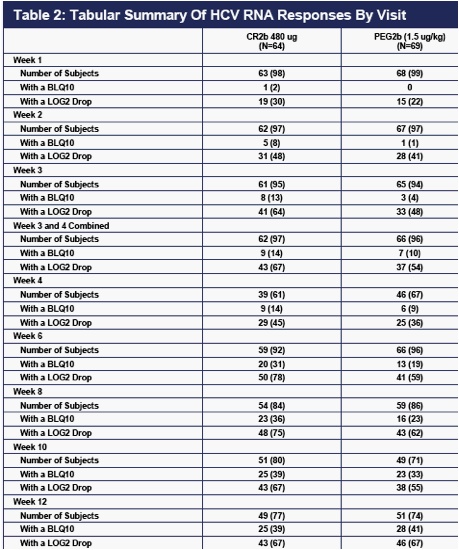
As can be seen from the graph of mean drops in viral load from baseline shown in Figure 3, the 480 ug dose of CR2b appears to provide reductions in viral load modestly higher than PEG2b in the first several weeks of treatment. Note that HCV RNA data are still pending on a few patients in 480 STUDY Panel B at Weeks 8, 10, and 12.
The rates of >2 log drops and the rates of undetectable HCV RNA through 12 weeks are shown in Table 2 below. The data available also suggest that CR2b provides somewhat more rapid reductions in HCV RNA during the first several weeks, and that PEG2b catches up by Week 12. Again, some HCV RNA values for patients in 480 STUDY Panel B are still pending at Weeks 8, 10, and 12.
SAFETY
The overall safety of the two dose groups is summarized in Table 3. No deaths
occurred. There were five more dropouts for any reason on CR2b, but one less
dropout due to AEs on CR2b. Nearly all patients experienced at least one AE.
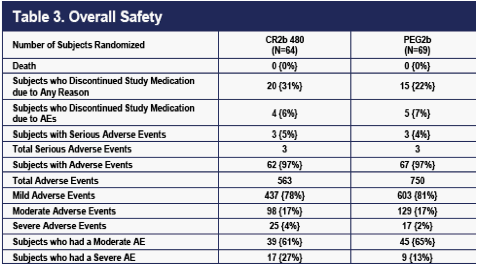
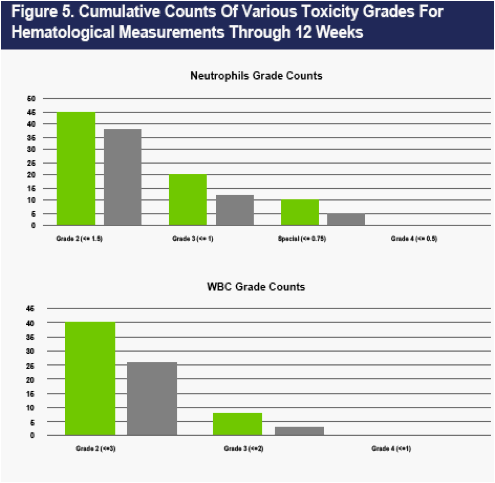
The overall number of AEs was higher in the PEG2b group (Table 3). The number of moderate AEs was also higher in the PEG2b group (Table 3). Eight more severe AEs occurred in the CR2b than in the PEG2b group (25 vs 16, Table 3). An excess of bone marrow effects (comprised mostly of neutropenia) accounted for the difference in severe AEs (data not shown). No patient experienced a neutrophil count <500.
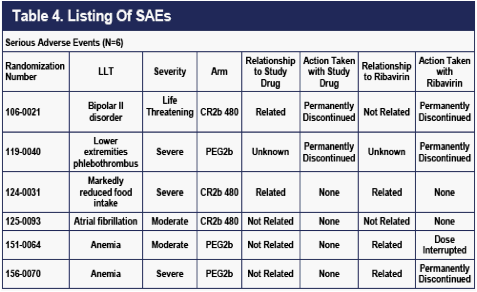
As of April 4, 2010, six SAEs had occurred, three on CR2b, and three on PEG2b. All SAEs observed to date are labeled for IFNa2b (Table 4).
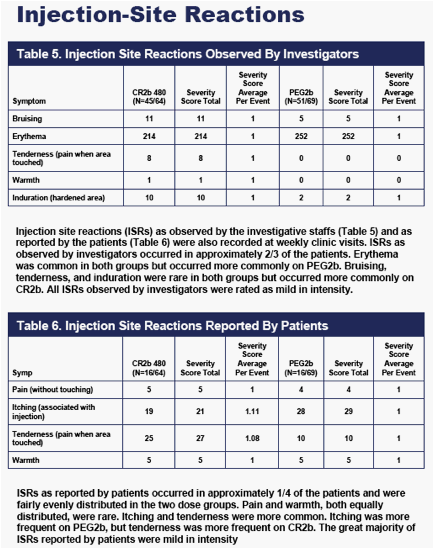
FLU-LIKE SYMPTOMS
As shown in Figure 4, flu-like symptom events recorded at weekly clinic visits were less frequent on CR2b than on PEG2b. Differences in flu-like symptom event counts were evident at Day 7- immediately prior to the second dose of IFNa2b in the PEG2b group, and well-prior to the second dose of IFNa2b in the CR2b group (which occurred on Day 14).
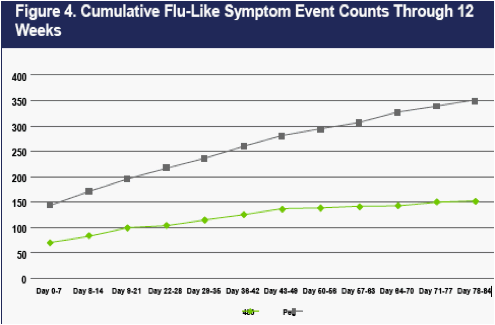
The differences in flu-like symptom event counts on CR2b versus PEG2b present at Day 7 progressively increased thereafter during the 12-week dosing period, largely because flu-like events continued to accumulate at a higher rate on PEG2b. By Week 12, 350 flu-events had occurred in the PEG2b group, and 152 flu-events had occurred in the 480 ug CR2b group.

references
1. Alvarez-Uria G, Day JN, Nair AJ, Russell SK, Vilar FJ. Reduction in neutrophil count during hepatitis C treatment: drug toxicity or predictor of good response? Dig Dis Sci 2009. Published on line 16 September 2009.
2. Lindsay KL, Morishima C, Wright EC, Shiffman ML, Everson GT, Lok AS, Bonkovsky JL, Lee WM, Morgan TR, Ghany MG, HALT-C Trial. Blunted cytopenias and weight loss: new correlates of virologic null response to re-treatment of chronic hepatitis C.
3. Roomer R, Hansen BE, Janssen HL, deKnegt RJ. Infections during treatment with peginterferon alfa and ribavirin for chronic hepatitis C are not associated with neutropenia. Hepatology 50:317A, 2009.
|
| |
|
 |
 |
|
|