 |
 |
 |
| |
Impact of Sustained Virological Response After Antiviral Treatment in Chronic Hepatitis C Patients on Life Expectancy and Quality-adjusted Life-years
|
| |
| |
Reported by Jules Levin
EAS Apr 14-18 2010 Vienna Austria
S Cure,1 F Bianic,1 H Cawston,1 L Dartois,1 H Zhang2
1i3 Innovus, UK; 2WWHE&P, Johnson & Johnson Pharmaceutical Services, Raritan, NJ, USA
Huabin Zhang, WWHE&P, Johnson & Johnson Pharmaceutical Services, Raritan, NJ, USA. E-mail: HZhang20@its.jnj.com
This study was supported by Tibotec, a division of Janssen-Cilag. Editing/styling and assistance with incorporating authors' contributions) was provided byGardiner-Caldwell Communications, and was funded by Tibotec/Janssen-Cilag.

ABSTRACT
Background and Aims: Chronic hepatitis C (CHC) is a curable disease. Studies have shown that patients achieving sustained virological response (SVR) can remain virus free. CHC treatment has been evaluated accounting for its efficacy (SVR) and its long-term effects in terms of reducing disease progression to advanced stages including hepatocellular carcinoma, long-term improvement on quality of life, and reduction of medical care costs. The objective of the study was to assess the impact of SVR in CHC patients on lifetime life-years (LY) and quality-adjusted life-years (QALY) based on published cost-utility analyses.
Methods: A systematic literature review identified publications to date reporting the cost-effectiveness of antiviral therapies in CHC. PubMed, the Centre for Reviews and Dissemination (CRD) and Health Technology Assessment (HTA) reports were searched. We included all cost-effectiveness studies based on a Markov model reporting lifetime LYs and QALYs. All published studies assumed that patients in the comparison groups share the same attributes such as starting age, health states, disease progression probabilities, mortality, and utilities associated with each health state, etc. We conducted a back calculation on the LYs and QALYs associated with SVR and non-SVR because SVR is the only difference between the two comparators for the lifetime QALY and LY differences in the final reported incremental cost-effectiveness ratio (ICER) results.
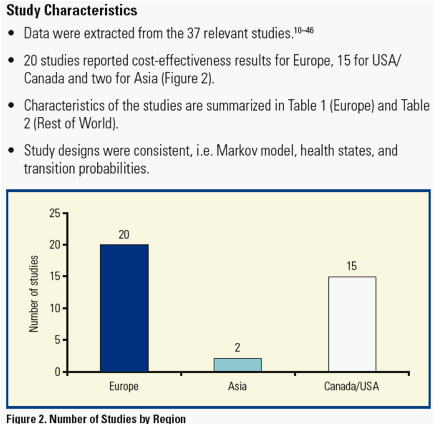
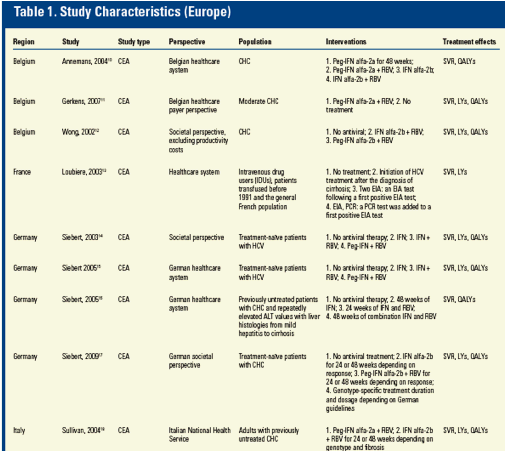
Results: A total of 893 unique references were retrieved and 80 articles met the inclusion criteria. Among the selected studies, 37 reported for each comparator either the SVR rate, LYs and/or QALYs.
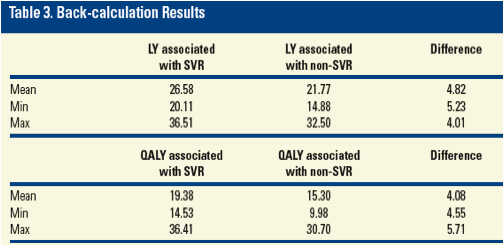
Among all these studies, SVR was consistently associated with more LYs (26.6, range [20.1-36.5]) and QALYs (19.4, range [14.5-36.4]) while non-SVR was associated with a lower number of LYs (21.8, range [14.9-32.5]) and QALYs (15.3, range [10.0-30.7]). This trend was consistent across all studies when this analysis was feasible (n=27).
Conclusions: SVR is associated with longer life expectancy and more QALYs than non-SVR. It is important to account for these lifetime benefits when the values of an antiviral treatment in CHC are being evaluated.
Background & Objectives
Hepatitis C primarily affects the liver, resulting in significant inflammation and tissue damage. Asymptomatic in nature, the majority of individuals are unaware of their infection status for many years. After acute infection, 85% of individuals develop chronic infection.1
Disease progression is variable, occurring over a period of 20-50 years, and may lead to liver cirrhosis, hepatocellular carcinoma (HCC) and death in a significant proportion of patients.2-4 Hepatitis C virus (HCV) is the leading cause of chronic liver disease and HCC.2,3 Furthermore, mortality associated with HCV is expected to increase dramatically over the next two decades.4
Treatment of hepatitis C usually consists of a course of either mono- or combination therapy of pegylated interferon and/or ribavirin, for periods of 24 or 48 weeks, with three treatment goals: 1) prevention of cirrhosis and its complications; 2) reduction of extra-hepatic manifestations; and 3) prevention of disease transmission.5
Treatment is successful in approximately half of all patients infected with genotype 1 and in >80% of patients with genotypes 2 and 3.6 Successful treatment is defined as a SVR in the majority of patients, i.e. serum HCV RNA negativity 24 weeks after the end of therapy.7
SVR is generally considered to indicate permanent resolution of infection.8,9 Therefore it is usually thought that patients with mild-to-moderate liver disease who achieve SVR are at lower risk of further disease progression.8,9
The aim of the study was to assess the impact of SVR in chronic hepatitis C (CHC) patients on lifetime life-years (LY) and quality-adjusted life-years (QALYs) based on published cost-utility studies.
Methods
Search Strategy
· A systematic literature review of all published economic evaluations was carried out for all antiviral therapies in CHC in November 2009.
· The electronic databases searched were PubMed/Medline, the CRD, and HTA.
· Search criteria included terms related to:
- The disease (e.g. 'hepatitis C' or 'hepativirus' or 'CHC').
- The type of study, i.e. economic evaluations (e.g. 'cost*', 'costs and cost analysis', or 'cost benefit')
- Inclusion criteria: all cost-effectiveness and cost-utility studies based on a Markov model developed in CHC and reporting at least one of the outcomes of interest.
· Endpoints of interest: LYs, QALYs and total costs.
Data Extraction
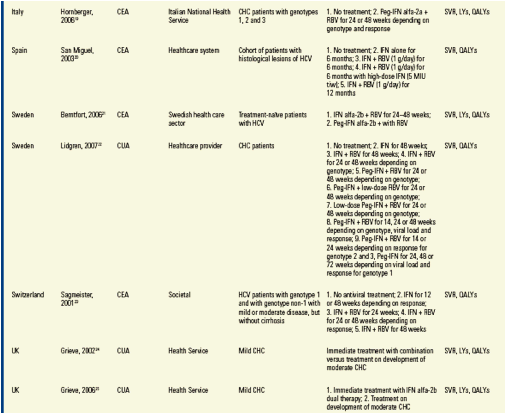
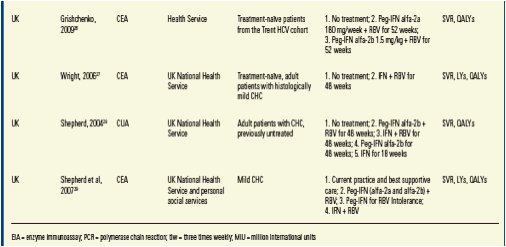
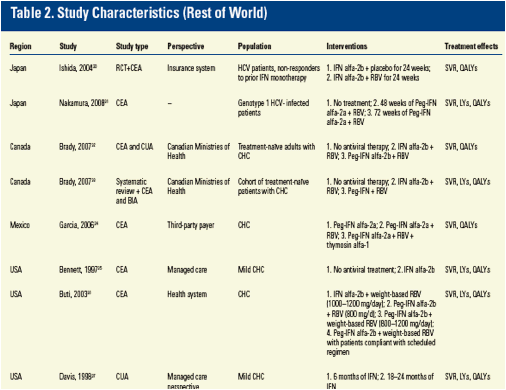
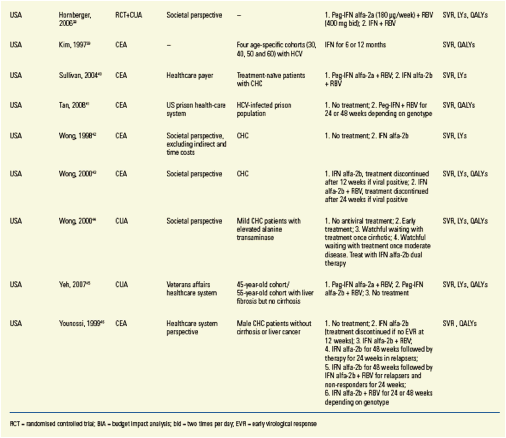
· The following data were recorded for each study:
- Study type, e.g. cost-effectiveness analysis (CEA), cost-utility analysis (CUA)
- Cohort characteristics, e.g. age, gender, HCV severity, HCV genotype
- Perspective, e.g. third-party payer or societal perspective
- Markov structure, e.g. health states
- Treatments
- Inputs, e.g. transition probabilities, utilities, discount rates
- Outcomes, e.g. SVR, LYs, QALYs, total costs.
Back-calculation
· The calculations assumed that SVR difference is the only difference between the two comparators (A and B) for the lifetime QALY and LY differences in the final reported ICER results.
· Therefore the following equations apply:Treatment A: SVRA * LYSVR + (1-SVRA) * LYnon-SVR = total LYATreatment B: SVRB * LYSVR + (1-SVRB) * LYnon-SVR = total LYB
· Thus considering that LY or QALY for SVR patients and for non-SVR patients are the same for both treatments:
LYSVR = [total LYA - (1-SVRA) * LYnon-SVR]/SVRA
LYnon-SVR = [total LYB - (SVRB * total LYA)/SVRA] / [(1-SVRB)-SVRB* (1-SVRA)/SVRA]
· The back calculation equations for QALY are identical to those for LY
RESULTS
Systematic Literature Review
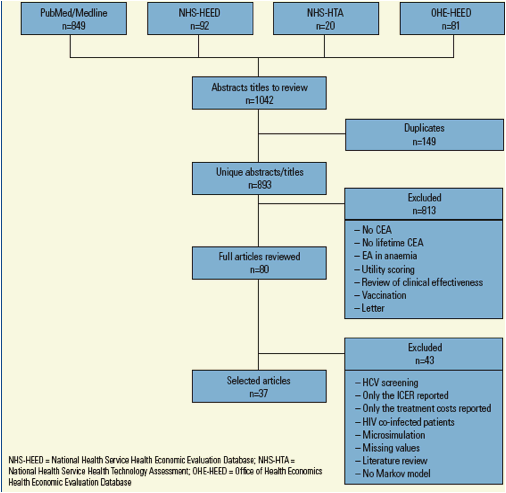
· Figure 1 shows a flow diagram of the literature review and data extraction.
· 1042 citations were screened, and 149 duplicates were excluded.
· 893 abstracts/titles were evaluated and 80 articles were identified as potentially relevant. The reasons for exclusion of the 813 articles were:
- No CEA
- No lifetime CEA
- Economic analysis in anaemia
- Utility scoring
- Review of clinical effectiveness
- Vaccination
- Letter.
· The full papers were screened from the 80 identified as potentially relevant, 37 were selected and data were extracted. The reasons for exclusion of the 43 non-selected articles were:
- HCV screening
- Only the ICER reported
- Only the treatment costs reported
- HIV co-infected patients
- Microsimulation
- Missing values
- Literature review
-- No Markov model.
Figure 1. Flow Diagram of Literature Review and Data Extraction (1st and 2nd Screening)

|
| |
|
 |
 |
|
|