 |
 |
 |
| |
Exposure-Viral Response Analyses of the Non-nucleoside Polymerase Inhibitor ABT-333, Following Monotherapy and ABT-333 Plus Pegylated Interferon and Ribavirin Therapy
|
| |
| |
Reported by Jules Levin
EASL 2010, Vienna, Austria, 14-18 April 2010
Sven Mensing, Rajeev Menon, Cheri Klein, Paul Diderichsen, Aksana Kaefer, Peter Noertersheuser, Walid Awni
Corresponding author: Rajeev Menon, Abbott, 100 Abbott Park Road, Dept. R4PK, AP13A, Abbott Park, IL 60064, USA

INTRODUCTION
ABT-333 is a novel non-nucleoside sulfonamide palm site NS5B polymerase inhibitor being developed for the treatment of HCV genotype 1 infection.
ABT-333 exhibits highly potent and selective activity against genotypes 1a and 1b HCV polymerases.
ABT-333 was safe and well tolerated in single and multiple ascending doses up to the highest doses studied in healthy volunteers: 2000 mg in single doses and 1600 mg BID in multiple doses.
In naïve genotype 1 HCV+ patients, ABT-333 has been administered in 2 blinded randomized studies (Figure 1).
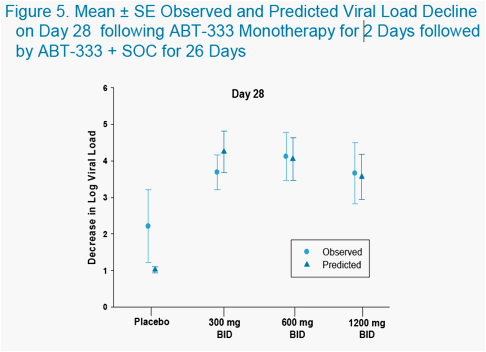
-- ABT-333 or Placebo was administered as monotherapy for 2 days in a Phase 1b study in a 18 subjects. The maximum least square (LS) mean change in viral load from baseline was -1.52 log at the 600 mg BID dose.1 Following the Phase 1b study, ABT-333 or Placebo was administered to HCV patients in a Phase 2a study as monotherapy for 2 days followed by ABT-333 or Placebo plus Pegylated Interferon (PegIFN) and Ribavirin for 26 days in 30 subjects.2 The viral load profile from the Phase 2a study is shown in Figure 2.
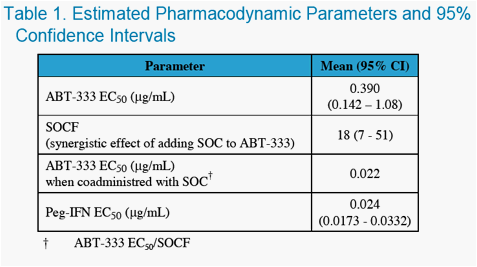
Pharmacokinetic and viral load data from these 2 studies were combined and used to characterize the exposure-viral response relationship between ABT-333 concentrations and viral load decline.
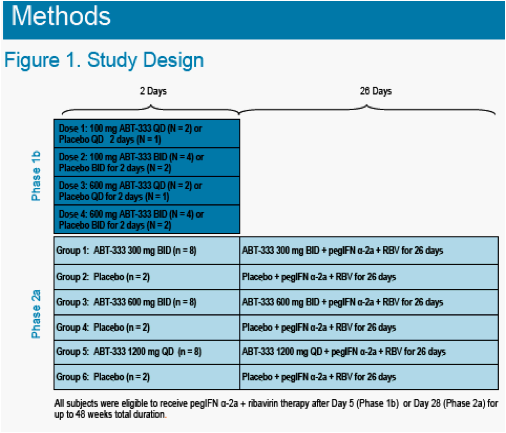




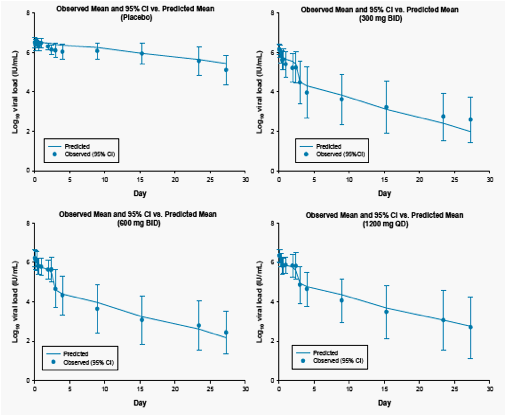
Figure 4. Mean Observed and Predicted Viral Load Data



|
| |
|
 |
 |
|
|