| |
Interleukin-28B genetic variants and
hepatitis virus infection by different viral genotypes in Spain
|
| |
| |
Hepatology July 2010
Marco Antonio Montes-Cano 1, Jose Raul Garcia-Lozano 1, Cristina Abad-Molina 1, Manuel Romero-Gomez 2, Natalia Barroso 3, Jose Aguilar-Reina 3, Antonio Nunez-Rold‡n 1, Maria Francisca Gonz‡lez-Escribano 1 *
1Servicio de Inmunologia, Hospital Universitario Virgen del Rocio/Instituto de Biomedicina de Sevilla (IBIS), Sevilla, Spain
2UCM de enfermedades digestivas y Ciberehd, Hospital Universitario de Valme, Universidad de Sevilla, Sevilla, Spain
3Servicio de Digestivo Hospital Universitario Virgen del Rocio, Sevilla, Spain
email: Maria Francisca Gonz‡lez-Escribano (mariaf.gonzalez.sspa@juntadeandalucia.es)
*Correspondence to Maria Francisca Gonz‡lez-Escribano, Servicio de Inmunologia, Hospital Universitario Virgen del Rocio, 41013 Sevilla, Spain
Potential conflict of interest: Nothing to report.
fax: (34)-95-501-3221
Funded by:
FIS 07/0061 Fondos FEDER and Plan Andaluz de Investigacion; Grant Number: PAI, grupos CTS-0197, CTS-0102
Abstract
Genetic host factors may modify the course of the hepatitis C virus (HCV) infection. Very recently, a genome-wide scan that reported association of the IL28B locus with response to treatment in HCV infection was published. The aim of the current study was to investigate the relationship of this locus with outcome of HCV infection in a cohort constituted by a total of 731 Spanish individuals. From these, 284 were subjects with persistent infection, 69 were individuals who naturally cleared the virus, and 378 were noninfected subjects. Genotyping of the rs12979860 (C>T) in the IL28B locus was performed using a TaqMan 5 allelic discrimination assay.
The CC genotype was overrepresented among patients infected with viral genotypes non-1 (66.7% versus 39.1% in patients infected with viral genotype-1, P = 8.5 x 10-5, odds ratio [OR] = 0.32, 95% confidence interval [CI] 0.17-0.60); patients with spontaneous resolution of infection (72.5% versus 45.6% of the individuals with persistent infection, P = 6.2 x 10-5, OR = 0.32; 95%CI, 0.18-0.57); and lastly, patients with sustained response (60.2% versus 32.1% found in patients with nonsustained response, P = 3.1 x 10-5, OR = 0.31; 95%CI, 0.17-0.56).
Conclusion: We have found different rates of viral genotype infection depending on the IL28B variant as well as an association of this locus with natural and treatment-mediated response.
Hepatitis C virus (HCV) infection is estimated to affect 170 million people worldwide. This infection results in a chronic active hepatitis in more than 80% of the infected patients, of which 20%-30% develop progressive fibrosis and cirrhosis, whereas only approximately 10%-20% of the infected people spontaneously eliminate the virus.[1] Different factors may influence the ability to spontaneously clear the virus. Ethnic differences in clearance frequency suggest involvement of host genetic variations in spontaneous viral clearance.[2] Currently, the most effective initial therapy for viral clearance is the combination of interferon-alpha (IFN-) and rivabirin (RBV); however, this standard therapy does not produce sustained response in all the patients treated. Different factors have been evaluated as predictors of the sustained response to treatment, with controversial results.[3] Very recently, a genome-wide association study identified a single nucleotide polymorphism (SNP) rs12979860 (C>T) that was strongly associated with sustained virological response to the treatment with pegylated IFN- and RBV in a cohort of more than 1600 American individuals (with different ethnicity) chronically infected with the genotype 1 of the HCV.[4] This SNP is located on chromosome 19q13, 3 kilobases (kb) upstream of the interleukin-28B (IL28B) gene that encodes a type III IFN (IFN-3). Almost simultaneously with this first study, another two genome-wide association studies reporting an association of the response to IFN- and RBV treatment with the IL28B locus (although with different SNPs) have been published in Australian and a Japanese patient cohorts.[5][6] An association of rs12979860 with natural viral clearance in six different cohorts of individuals with diverse ethnic origins has also been reported.[7] The aim of this study was to explore the influence of the rs12979860 variations in the outcome of HCV infection in the Spanish population.
Abbreviations:
CHC, chronic hepatitis C; CI, confidence interval; G1, viral genotype 1; HCV, hepatitis C virus; IFN- interferon alpha; IL28B, interleukin-28B; NIS, noninfected subjects; NSR, nonsustained response; OR, odds ratio; RBV, ribavirin; SNP, single-nucleotide polymorphism.
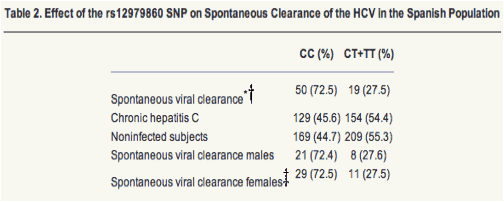
Data of distribution of rs12979860 genotypes in males and females with spontaneous viral clearance are also displayed.
* Comparison spontaneous viral clearance versus chronic hepatitis; P = 6.2 x 10-5; OR = 0.32; 95% CI, 0.18-0.57.
Comparison spontaneous viral clearance versus noninfected subjects; P = 2.2 x 10-5; OR = 0.31; 95%CI, 0.17-0.56.
Comparison spontaneous viral clearance males versus females; P = 1.0.
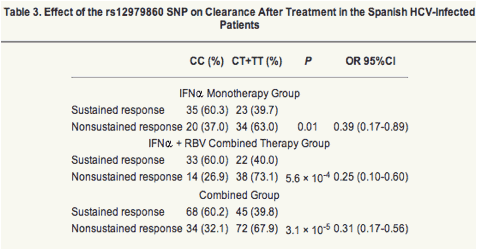
Data of distribution of rs12979860 genotypes in patients with different treatment schedules are also displayed.
OR, Odds ratio; CI, confidence interval.
Results
Genotypes of the rs12979860 were unequivocally assigned in all the cases, except in one of the individuals with persistent infection, who was eliminated from the analysis (CHC genotypes n = 283), and they were in Hardy-Weinberg equilibrium in the NIS control group (CC: 44.7%, CT: 42.1%, and TT: 13.2%; C 0.66 and T 0.34). Demographic data, viral genotype and viral load, route of infection, fibrosis stage, and biochemical features of our CHC cohort are displayed in Table 1. With regard to the viral genotypes, 74.2% of the CHC patients were infected with viral genotype 1 (G1), 23.3% were infected with non-1 viral genotype (non-G1) (4.2% with genotype 2, 15.2% with genotype 3, and 3.9% with genotype 4), and the rest (2.5%) had co-infections with different HCV genotypes. When patients were stratified according to their rs12979860 genotypes (CC versus CT+TT), the CC genotype was overrepresented among non-G1 (66.7%) on comparing with both G1-infected patients (39.1%, P = 8.5 x 10-5, OR = 0.32, 95%CI = 0.17-0.60) and NIS (44.7%, P = 0.001, OR = 0.40, 95%CI = 0.22-0.72), whereas frequency of the CC genotype among G1-infected patients was similar to that of the NIS group (P = 0.18). Moreover, we found higher median levels of alanine aminotransferase (106.0 IU/L versus 85.5 IU/L, P = 0.006) and lower median levels of gamma-glutamyltransferase (35.5 IU/L versus 41.0 IU/L, P = 0.002) in patients CC when compared with patients CT+TT. No statistical significant differences between individuals CC and CT+TT were observed in any other cases (Table 1).
Results obtained regarding the effect of the rs12979860 variations in the spontaneous HCV clearance in our study are displayed in Table 2. The CC genotype was associated with the spontaneous resolution of infection; 72.5% of those individuals who cleared the virus had the CC genotype versus 44.7% of the NIS group (P = 2.2 x 10-5, OR = 0.31, 95%CI = 0.17- 0.56) and 45.6% of the CHC group (P = 6.2 x 10-5, OR = 0.32, 95%CI = 0.17-0.59), whereas individuals with persistent infection had a frequency of this genotype similar to that of the NIS group (CHC versus NIS, P = 0.82). Next, we tested whether the effect of this polymorphism was the same in both sexes, because this factor had been the most consistently associated with natural elimination of the virus. The rs12979860 CC genotype was associated with spontaneous clearance in both men and women. Regarding viral clearance after treatment, data of response were available in 219 patients; those 65 subjects without data of response were excluded from this part of the study. Viral clearance after treatment was associated with the IL28B locus, because frequency of rs12979860CC among patients with SR (n = 113) was 60.2% versus 32.1% found in patients with NSR (n = 106) (P = 3.1 x 10-5, OR = 0.31, 95%CI = 0.17-0.56). We found an association of this polymorphism with SR in the monotherapy as well as in the combined therapy groups. In the monotherapy group, frequency of CC patients with SR was 35 of 58 (60.3%) versus 20 of 54 (37.0%) among patients with NSR (P = 0.01, OR = 0.39, 95%CI = 0.17-0.89), and in the combined therapy group, frequency of CC patients with SR was 33 of 55 (60.0%) versus 14 of 52 (26.9%) among patients with NSR (P = 5.6 x 10-4, OR = 0.25, 95%CI = 0.10-0.60) (Table 3).Therefore, according to our data, distribution of rs12979860 genotypes relating to the response was the same in both treatment schedules, and consequently, we combined both therapy groups for analysis. Finally, when patients were stratified by their viral genotypes, the rate of SR in CC patients infected by non-G1 was 87.2% (34/39) and 84.2% in CT+TT patients infected by non-G1 (16/19, P = 0.76); whereas in patients CC infected with G1 was 53.9% (34/63) and in patients CT+TT infected with G1 was 29.6% (29/98, P = 1.98 x 10-3, OR = 0.36, 95%CI = 0.18-0.73).
Discussion
In this study, we found a preference of the HCV genotypes to infect individuals with a determinate rs12979860 genotype and association of the IL28B locus with spontaneous viral clearance as well as with the response to treatment in the Spanish population. Very recently, three genome-wide association studies have reported an association between the IL28B locus and the response to IFN-a and RBV therapy in HCV-infected patients.[4-6] In our study, the rs12979860CC genotype was overrepresented in SR patients. The association was detected in both patients treated with only IFN-a and patients treated with the combined therapy IFN-a and RBV. The group of patients bearing the genotype associated with a better response had lower pretreatment median levels of gamma glutamyltransferase, which is the aminotransferase more consistently associated with the SR, with OR comparable to viral genotype.[9-11] Levels of alanine aminotransferase were higher in our group of CC patients with CHC; although this aminotransferase has not been clearly associated with SR, some authors described association of higher levels in the baseline with SR in patients infected with non-G1.[12] Except for frequency of the viral genotypes, we did not find differences between both rs12979860 genotype groups and the rest of the factors analyzed previously described as related to SR. All three previous studies support a robust association of the IL28B locus with the response to the antiviral therapy across different population groups, including only viral genotype 1-infected patients. This is the most common viral genotype in developed countries and the poorest responder to therapy (40%-50% of responder versus 75% of patients infected with others genotypes). The current study included 23.3% of non-G1-infected patients, and, surprisingly, determinate HCV genotypes had preference by individuals with a particular rs12979860 genotype because the frequency of subjects bearing CC was overrepresented among non-G1-infected patients (66.7%). Although these results need confirmation in other cohorts, taking into account frequency of rs12979860 CC genotype in our noninfected population (44.7%), we could speculate with a possible positive selection of individuals rs12979860 CC by the non-G1 virus or, conversely, a negative selection of these subjects by the G1 (39.1%). In this sense, both the highest rs12979860 C allelic frequency and the greatest rate of infection by non-G1 viral HCV genotypes have been described in Asian populations, whereas the lowest frequency of C allele and the highest rates of G1 infection have been described in African populations.[4][13] Some studies support the idea that elements of both innate and adaptive immune response could be under selective pressure in viral infections, and this fact could determine the final picture found in observational studies.[14-16] There exists no systematic explanation for the viral genotype-specific differences found in response to treatment; therefore, if non-G1 viral genotypes had a preference to infect patients with a determinate IL28B genotype, influence currently attributed to the virus could be caused, at least partially, by the host genetic background. Although the individuals included in some combinations of viral and host genotypes did not permit statistical analysis, our results suggest an influence of both host and virus factors in the SR. In this sense, the highest rate of SR was found in CC patients infected by non-G1 (87.2%) and the lowest among individuals CT+TT infected with G1 (29.6%). The influence of the host genotype could be stronger among individuals infected by G1 (rate of response of CC 53.9% versus 29.6% in CT+TT) than among those infected by non-G1 (rate of response of CC 87.2% versus 84.2% in CT+TT).
Further studies are needed to clarify the weight of these factors in the response. Another question is whether this gene is involved in natural viral clearance. During the preparation of this manuscript, a paper describing an association of this SNP with natural viral clearance in six different cohorts of individuals with different ethnic origins was published.[7] This study reported association of this polymorphism with natural viral clearance among American individuals with both European and African ancestry. The current study shows a similar association between rs12979860 and natural viral clearance, because frequency of the CC genotype was overrepresented in individuals with spontaneous viral clearance. Therefore, our results support the same conclusion as the previous study in a Spanish cohort. This association seems to be independent of sex, which is a factor consistently associated with natural clearance.
In conclusion, we have found different rates of viral genotype infection depending on IL28B variant as well as an association of this locus with natural and treatment-mediated response.
Patients and Methods
A total cohort consisting of 731 Spanish individuals were included in this study. They were selected by using surnames and by having grandparents born in Spain. This cohort included 284 subjects with persistent infection, 69 individuals who naturally cleared the virus, and 378 noninfected subjects. The persistent infection group included 166 males and 118 females suffering from biopsy-proven chronic hepatitis C (CHC) with compensated liver disease followed in the outpatient clinic of the Hospital Universitario Virgen del Rocio and Hospital Universitario de Valme (Sevilla, Spain) from 2001 to 2004. All CHC patients were hepatitis B surface antigen and human immune deficiency virus negative, anti-HCV positive, and HCV RNA positive in serum. Anti-HCV, HbsAg, and human immune deficiency virus were determined by commercially available methods (HCV 3.0 test, ORTHO, and Enzygnost hepatitis B surface antigen 5.0 and anti-human immune deficiency virus-1/2 plus; DADE, Behring, respectively). Percutaneous liver biopsies were performed under ultrasonographic control. A portion of the biopsy specimen was used for the histology diagnosis. Disease staging was defined according to Scheuer,[8] with ranking from F0 (absence of fibrosis) to F4 (cirrhosis stage). Patients were stratified into two groups: F0-F2, absence of fibrosis to moderate fibrosis; and group F3-F4, with advanced fibrosis-cirrhosis. Data of response to treatment (51.4% received IFN-a, and 48.6% IFN-a plus RVB) were available in 219 patients; 113 of them had a sustained response (SR), HCV RNA levels remained undetectable during 6 months after therapy discontinuation) and 106 had a nonsustained response (NSR), including nonresponder patients (HCV RNA levels detectable during the completed period of the treatment) and relapsed responder patients (undetectable HCV RNA during the therapy but detectable after discontinuation).
The group with spontaneous viral clearance comprised 29 men and 40 women who were anti-HCV positive and HCV-RNA negative. Most of these subjects were blood donors with anti-HCV positive in the routine screening of viral antibodies; these subjects are referred to the hepatology unit and, according to the established protocol, HCV-RNA detection is performed. Lastly, a group of 223 male and 155 female blood and bone marrow donors (noninfected subjects [NIS]) were considered as representative of the normal frequencies of the SNP studied in the Spanish population. Patients and controls agreed to a blood examination according to the guidelines of the Hospital Bioethic Committee.
Genotyping.
DNA from patients and controls was extracted from peripheral blood using standard methods. Genotyping of the rs12979860 was performed using a TaqMan 5 allelic discrimination assay (Applied Biosystems, Foster City, CA). The primers used were 5 GCCTGTCGTGTACTGAACCA 3 (forward) and 5 GCGCGGAGTGCAATTCAAC 3 (reverse), and the TaqMan MGB probe sequences were 5 TGGTTCGCGCCTTC 3 and 5 CTGGTTCACGCCTTC 3. The probes were labeled with the fluorescent dyes VIC and FAM, respectively. Polymerase chain reaction reactions were carried out in a total volume of 8 uL with the following amplification protocol: denaturation at 95ˇC for 10 minutes, followed by 40 cycles of denaturation at 92ˇC for 15 seconds, and finished with annealing and extension at 60ˇC for 1 minute. Genotyping of each sample was automatically attributed by the SDS 1.3 software for allelic discrimination.
Statistical Analysis.
Genotypic frequencies were obtained by direct counting, and statistical analysis was performed by the chi-squared test calculated on 2 x 2 contingency tables assuming a recessive model (CC versus CT+TT) using the Statcalc program (Epi Info version 6.0; Centers for Disease Control and Prevention, Atlanta, GA). Odds ratio (OR) with 95% confidence intervals (95% CI) was calculated using the same program. Median values of quantitative variables were compared using a nonparametric test (Mann-Whitney two-tailed test). P-values less than 0.05 were considered statistically significant.
References
1 Thomas DL, Seeff LB. Natural history of hepatitis C. Clin Liver Dis 2005; 9: 383-398.
2 Kenny-Walsh E. Clinical outcomes after hepatitis C infection from contaminated anti-D immunoglobulin: Irish Hepatology Research Group. N Engl J Med 1999; 340: 1228-1233.
3 Kau A, Vermehren J, Sarrazin C. Treatment predictors of a sustained virologic response in hepatitis B and C. J Hepatol 2008; 49: 634-651.
4 Ge D, Fellay J, Thompson AJ, Simon JS, Shianna KV, Urban TJ, et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature 2009; 46: 399-401.
5 Suppiah V, Moldovan M, Ahlenstiel G, Berg T, Weltman M, Abate ML, et al. IL28B is associated with response to chronic hepatitis C interferon- and ribavirin therapy. Nat Genet 2009; 41: 1100-1104
6 Tanaka Y, Nishida N, Sugiyama M, Kurosaki M, Matsuura K, Sakamoto N, et al. Genome-wide association of IL28B with response to pegylated interferon- and ribavirin therapy for chronic hepatitis C. Nat Genet 2009; 41: 1105-1109.
7 Thomas DL, Thio CL, Martin MP, Qi Y, Ge D, O'Huigin C, et al. Genetic variation in IL28B and spontaneous clearance of hepatitis C virus. Nature 2009; 461: 798-801.
8 Scheuer PJ. Classification of chronic viral hepatitis: a need for reassessment. J Hepatol 1991; 13: 372-373.
9 Berg T, von Wagner M, Nasser S, Sarrazin C, Heintges T, Gerlach T, et al. Extended treatment duration for hepatitis C virus type 1: comparing 48 versus 72 weeks of peginterferon-alfa-2a plus ribavirin. Gastroenterology 2006; 130: 1086-1097.
10 Berg T, Sarrazin C, Herrmann E, Hinrichsen H, Gerlach T, Zachoval R, el al. Prediction of treatment outcome in patients with chronic hepatitis C: significance of baseline parameters and viral dynamics during therapy. HEPATOLOGY 2003; 37: 600-609.
11 von Wagner M, Huber M, Berg T, Hinrichsen H, Rasenack J, Heintges T, et al. Peginterferon-alpha-2a (40KD) and ribavirin for 16 or 24 weeks in patients with genotype 2 or 3 chronic hepatitis C. Gastroenterology 2005; 129: 522-527.
12 Shiffman ML, Suter F, Bacon BR, Nelson D, Harley H, Sol‡ R, et al. Peginterferon alfa-2a and ribavirin for 16 or 24 weeks in HCV genotype 2 or 3. N Engl J Med 2007; 357: 124-134.
13 Yu ML, Chuang WL. Treatment of chronic hepatitis C in Asia: when East meets West. J Gastroenterol Hepatol 2009; 24: 336-345.
14 Pang PS, Planet PJ, Glenn JS. The evolution of the major hepatitis C genotypes correlates with clinical response to interferon therapy. PLoS One 2009; 11: e6579.
15 Telenti A. Adaptation, co-evolution, and human susceptibility to HIV-1 infection. Infect Genet Evol 2005; 5: 327-334. Links
16 Kawashima Y, Pfafferott K, Frater J, Matthews P, Payne R, Addo M, et al. Adaptation of HIV-1 to human leukocyte antigen class I. Nature 2009; 458: 641-645.
|
|
| |
| |
|
|
|