 |
 |
 |
| |
Highlights of the Vienna International AIDS Conference by Pablo Tebas, MD, University of Pennsylvania.
|
| |
| |
The International AIDS conference1 took place in Vienna (Austria) between July 18 and the 23 of 2010. On even years this is a massive meeting where more than 20,000 people show up to discuss HIV related research from epidemiology to social aspects of HIV infection, from legal issues that affect patients living with HIV to more scientific topics. On odd years, like last year's Conference in Cape Town, the focus is more on scientific and biological facets of the disease and the meeting is more similar to the Retrovirus conference. The International AIDS Conference meeting is too big for my taste, and it is easy to get lost in the gargantuan conference rooms and the multiple simultaneous lectures, but is always an interesting gathering. My summary will focus around what the conference calls track B, that relates to more clinical issues and on the presentations that caught my eye. Unfortunately I am certain that I am going to miss some important things. I want to apologize beforehand.
Political issues
It is impossible to participate in this conference and not become aware of the political and policy discussions that frequently dominate it. This was supposed to be the year that the conference would focus in drug addicts. 30% of new infections, particularly in Eastern Europe, are driven by IV drug use. Before the conference started, we even signed a big declaration "The Vienna Declaration"2 asking for the decriminalization of drug users, the implementation of methadone programs and other therapeutic programs as a way to curb the epidemic in many regions of the world. The whole conference center and documents were full of signs in Russian to stress this point to the target audience of the message: the Russian government. However, the bold declaration was lost among the worries about funding of AIDS programs both in the US and the developing world and how to sustain the Global Fund to fight AIDS, Tuberculosis and Malaria3 during this period of economic uncertainty and the shifting focus of the US administration policy, particularly in the developing world.
The eternally optimist and charismatic Bill Clinton gave a nice speech at the beginning of the conference. He looked older, thinner and wiser. He mentioned how many improvements managing HIV disease have occurred during the last years, with 5.2 million people at this time receiving antiretroviral therapy.
Unfortunately, that is only a small fraction of what the current need is (10 million more), and an even smaller fraction of the near future needs. He made a strong defense of the Obama administration, which was the target of many activist groups during the meeting, even saying that Dr. Goosby, the Director of the PEPFAR agency deserved a Purple Heart for coming to the Conference. The Obama administration has been criticized for what is seen by many as a lack of commitment to AIDS. PEPFAR funds that used to be channeled directly to ground organizations directly involved in the care of patients in the developing world is been channeled now more towards governments that want to have a bigger say in how that money is spent. The US administration and the local governments seem to want to focus expenses more in Health systems improvements rather than specific diseases like AIDS, and that change has created tension. Clinton warned against the "false choices" that have plagued the debates on AIDS policy for years: treatment vs prevention, AIDS specific funding, vs more generic funding, therapeutic funding vs vaccine funding... He basically asked activists that instead of trying to maintain the status quo, they should push for money in the US Congress for improving health systems, that the improvements in Health systems will lead to the identification of more patients with HIV infection and that the identification of those patients will put political pressure to increase funds for AIDS treatment. He was concerned about domestic (USA) AIDS funding, and the current troubles of some state programs. He chastised drug companies for allowing the swelling of the waiting list of ADAP programs in the US, asking them directly to fund the 1700 patients that are currently waiting for treatment, particularly in some Southern states. He even said that it would cost pharmaceutical companies next to nothing to fix that problem. He is absolutely right on that one. He also reminded developing country governments that they promised to decrease waste in their HIV programs and increase accountability of how the AIDS money is used, and they have failed in many cases to do so.
The Obama administration was the "punch bag" during much of the meeting and also took hits from Desmond Tutu, who even wrote a very critical editorial in the New York Times during the Conference 5. Although it is always easy to criticize the US and the Obama administration for not increasing our contributions to the global fight on AIDS the problem of contributions to the global fund is particularly appalling among some countries of the European Union. The UNAIDS/Kaiser Permanente report4 is a document worth reading as it provides data about the growing gap of US$7.7 billion between available resources and current needs and also about how much of the global contribution to AIDS expenses each country makes. I find particularly eye opening the chart about how much money each one of the big economies have contributed per each million $ of their GDP (putting everybody on the same scale). This is the ranking of the countries:
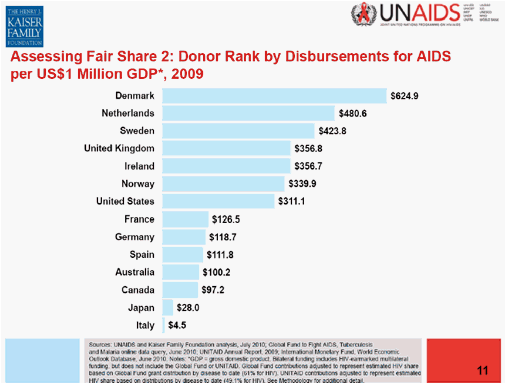
The US is in the middle of the graph by its contributions (which are the largest in size by far, because of the size of its economy) when compared to the northern Europeans countries, but the USA contributes 3 times more than France, Germany or Spain, 10 times more than Japan or 80 more than Italy. Austria contributions, the Conference host, are pathetic; however not much of the criticism during the meeting was directed at any of those governments.
Bill Gates, also a frequent guest in this conference, described what he thought is necessary to curb the epidemic in a speech that was both technical and passionate. Hackers usually talk about Microsoft as the "evil empire", which has definitely not been the case in Bill Gates response to AIDS…
Next month the big economies of the world will meet to discuss the next round of funding for the Global Fund. That, and subsequent commitments, will be the time to evaluate some of the political speeches that many governments made at Vienna. I would be insincere if did not tell you that I am very concerned about the future.
New antiretrovirals and strategies
The meat of this conference regarding new antiretrovirals was presented in a long late breaker session on Thursday. I will highlight the studies that caught my eye that I think they have the potential to affect care.
Joe Eron presented a very interesting study looking at the perennial question about when to start therapy, using data from the CASCADE cooperative6. The good thing about this cohort is that the time of infection is mostly known, what prevents some of the biases of other cohorts. What I liked more about this presentation is that in spite of the statistical complexities, at the end it was easy to understand the message and the study provided some numbers that are easy to remember. The endpoints they looked at in their study were AIDS defining events and all cause mortality. They did not look at other events that we used to think that were not HIV related, and now know that are increased in HIV patients like myocardial infarctions, kidney or liver failure. The benefit to treat HIV infection early is statistically significantly better than delaying therapy for anybody with CD4 cell counts below 500, but the benefits of antiretroviral therapy are smaller the higher the CD4 count is. Above 500 the benefits of starting treatment early become less clear. I think this is one of the first studies that gave the number of patients that need to be treated to prevent one of those clinical events. For example, if the CD4 is between 350 and 500 in order to prevent an AIDS event in 3 years you need to treat 34 patients , and in order to prevent one death (AIDS or not) you need to treat 71 patients. If your CD4 cell count is between 0 and 50 you only need to treat 3 patents to prevent and AIDS event. This type of analysis does not take into account the additional benefits of therapy, mainly preventing secondary cases of transmission of HIV, but provides important information for the individual patient that is trying to make a decision, and also for policy makers that want to know what the expenses are in exchange for the benefits of expanding antiretroviral programs. I thought this was a well done study that supports, at least partially, the current antiretroviral guidelines in the US.
Among the rest of the presentations regarding new antiretroviral treatment I will highlight 3 of them:
1. The analysis of the TMC-278 trials7. Rilpivirine (TMC278) is the most recent addition to the non-nucleoside reverse transcriptase inhibitor class. Cal Cohen presented the results of the two pivotal phase III trials of rilpivirine (ECHO and THRIVE) in antiretroviral naïve patients. These were large studies with more than 600 patients a piece. In these non inferiority studies patients received either rilpivirine 25mg qd or efavirenz 600mg qd, with Truvada ( tenofovir (TDF)/emtricitabine) in the ECHO and either TDF/emtricitabine, lamivudine/zidovudine or abacavir/lamivudine in THRIVE. 1368 patients were randomized to the study with a median baseline viral load of 5.00 log10 copies/mL and a median CD4 256 cells/mm3. Rilpivirine was non-inferior to efavirenz. At week 48, the proportion of patients with less than 50 copies/mL was 84.3% in the rilpivirine groups and 82.3 % in the Efavirenz groups. Somewhat surprisingly, the virologic failure rate was greater in the TMC-278 arms (9.0% vs 4.8% in the efavirenz group). This difference was compensated by an improved tolerability of rilpivirine with a lower rate of discontinuations due to side effects. The lipid profile was slightly better with TMC-278.
The main conclusion of these studies is that TMC_278 (rilpivirine) is pretty much equivalent to efavirenz, and maybe a little bit better tolerated. When the drug is approved by the FDA, something that certainly will happen soon, it will be another addition to the "preferred" list of antiertrovirals. Its main advantage over efavirenz is precisely the improved tolerability. For those patients with difficulties tolerating efavirenz the choice will be clear….for the rest, I think there will be questions, and we will need more long term data. In the future, cost will be an important consideration in the antiretroviral selection, and it will be interesting to see what happens when efavirenz becomes generic. That will hopefully be a strong force driving the cost of antiretrovirals down in the developed world.
An additional advantage of rilpivirine is that this drug is theoretically easy to combine with other drugs, so we could have another once a day tablet, but the problem in that regard is that the only "preferred" nucleoside combination to combine rilpivirine with is Tenofovir/FTC (Truvada) and we already have a fixed dose combination with efavirenz (Atripla).
2. New nevirapine data8 was presented evaluating a new long lived formulation (nevirapine XR), that can be administered once daily, and has a nicer pharmacokinetic profile than the old nevirapine, with lower peaks and only slightly lower troughs. The efficacy of this new formulation is similar to the twice daily formulation, and it is obviously easier to use. In the VERxVE study, there was a trend to lower frequency of side effects with the long acting formulation, both hepatic and dermatological, although the data was presented only orally in one of the answers to questions. Nevirapine is a good drug, a little bit tricky to use during the first few weeks, when most of the potential toxicities occur. Having a once daily formulation is a clear improvement for this "classic" antiretroviral that reminds me of the Phoenix, always coming out of its own ashes.
3. Arribas et al. presented the results of the Phase IIb, dose finding study of the new integrase inhibitor by Glaxo. GSK1349572 (ING572), the SPRING Study9 . In this important study, all patients received a two NRTI backbone and in addition they were randomized to 3 different doses of ING572 10 mg QD, ING572 25 mg QD, ING572 50 mg QD or EFV 600 mg QD. Mean baseline viral load was 4.46 log copies/mL and mean baseline CD4 cell count was 324 cells/µL. At 16 weeks, the proportion of patients with less than 50 copies/mL (TLOVR) were 90-96% in the three ING572 groups and 60% in the EFV group (this was not a surprise, as it is a well known fact that integrase inhibitors "clear" the virus faster than other antiretroviral regimens. There were no serious adverse events in the study. The lipid profile of the new drug looks good. Glaxo has made the decision to continue the development of this drug at a dose of 50 mg daily. I though it was a very impressive result for this promising new drug.
Then, a series of underpowered studies were presented during this and other sessions. Sometimes I wonder why some of these studies are accepted before they have reached final conclusions. My summary will be brief as I think these studies, although interesting, should not affect clinical practice. The SUPPORT study (N=101)10 reported similar efficacy of fosamprenavir/ritonavir when compared to efavirenz based regimens. SHIELD presented data on 31 patients that received Abacavir with 3TC and raltegravir -it looks ok-11. PROGRESS12 compared lopinavir/ritonavir/TDF/FTC to raltegravir/LPV/r (n=205) , showing similar efficacy. The SPARTAN13 study compared ATV/r + raltegravir to the usual Truvada+ atazanavir/ritonavir(n=94). The raltegravir/ATV/r combination looked good. The unusual results of the study were the surprising poor results (at week 24) of the more classic arm (only 30 patients in that arm) and the significant amount of resistance to raltegravir in patients failing the novel regimen. Early results of maraviroc and atazanavir/r combination compared to truvada atazanavir/r, also showed similar efficacy (80 vs 89%)14. Unfortunately all these studies are underpowered to prove biological equivalence in the usually accepted, clinically relevant, sense of the word (plus minus 10% efficacy). Due to the small sample size of these studies it is easy to make an error assuming that there are no differences between the different combinations when in fact there may be.
Some strategy trials were also presented: A step down strategy similar to the old ACTG 343 study was evaluated in the ATLAS study15 (n=40). Patients with undetectable viral loads "stepped down" from TDF/FTC/ATV/r to 3TC/ATV/r. Only 36 patients have reached week 24 and all of them maintained undetectability. Interestingly there was an increase in lipids after the switch, confirming the fact that tenofovir has lipid-lowering properties.
The strategy trial (MONET) presented 96 weeks data16. The 48 week data of this study had been presented during the Cape Town conference last year. Patients with an undetectable viral load were randomized to receive darunavir/ritonavir QD monotherapy or Darunavir/ritonavir QD plus two nucleosides. At 48 weeks darunavir/ritonavir monotherapy was non inferior. At 96 week (switch = failure analysis (TLOVR) 80.6% of the patients randomized to triple therapy had less than 50 copies/mL compared to 74.8% of the patients randomized to monotherapy. The monotherapy arm did not achieve non-inferiority as the lower limit of the 95% CI was -16%. However, when patients that failed monotherapy were intensified with nucleosides the efficacy of the 2 strategies was similar. The strategy of stepping down to boosted protease inhibitor monotherapy has the potential to save a significant amount of resources, but it has never caught on in the US. HCV co infection was a predictor of failure of this strategy.
Timing of antiretroviral in patients with Tuberculosis
The results of the CAMELIA study were presented17. This is probably one of most important studies presented in Vienna and one of the highlights of the Conference. This study, together with similar studies already published, have the potential to change clinical practice immediately. This was a study cosponsored by the National Institute of Health and the French equivalent (ANRS). In this study patients with very advanced HIV disease (median CD4 cell count 25) that presented with tuberculosis were randomized to receive antiretroviral treatment within 2 weeks of the diagnosis of tuberculosis or to delay the initiation of antiretroviral therapy for a period of 6-8 weeks. Patients that started antiretroviral therapy earlier had a significant decrease in mortality. Both the mortality and the benefit derived from treatment were greater the lower the baseline CD4 count was. The study was consistent with the results of ACTG5164 from the ACTG published by Andy Zolopa19, and the recent NEJM paper from Haiti20. The main difference with those studies is that the patients that participated in the CAMELIA study had more advanced disease so the benefits were immediate and much more dramatic than in other studies. The immune reconstitution syndrome (IRIS) was more common in patients that were started on antiretorivral therapy earlier; however IRIS was easy to manage and did not contribute significantly to bad outcomes. I think the question of when to start antiretroviral in patients with active tuberculosis is pretty much over, maybe with the exception of patients with TB meningitis, that were not included in this trial. The answer simple: as soon as you can, particularly in patients with a very low CD4 cell counts. Delaying antiretroviral therapy will only increase mortality. How to implement this new recommendation in the developing world is the next big challenge in this area.
The CAPRISA study
It would be impossible to talk about this conference without mentioning the results of the CAPRISA study that was presented and published in Science during the conference18. This study evaluated the use of a topical 1% tenofovir gel microbicide that decreases the risk of transmission of HIV by 39%. The product was well tolerated and also decreased the risk of herpes infection. For those women that were consistently adherent to the microbicide the efficacy preventing HIV infection was 54%. After a decade of microbicide studies that did not show any efficacy and in some cases even showed an increased risk of HIV transmission, the positive results of this study were welcomed. Certainly 39% efficacy is clearly insufficient, but is better than nothing. However, I don't think this product will make it to large field trials, particularly because after only 2 years more than 12% of the individuals using it had become infected with HIV. On the other hand, what this study shows is a path forward and the potential utility of microbicides as a tool to curb the epidemic. Future trials will evaluate the use of combinations of antiretrovirals in microbicides or the use medicated rings that only need to be changed once a month and will simplify the use of these products. It is going to be very interesting to see how microbicides compare to the use of oral PrEP in preventing new cases of HIV infection.
Metabolic complications
I co-chaired a session on the complications of antiretroviral treatment. During this session the bone results of ACTG5142 were presented21. After the initiation of antiretroviral therapy, and pretty much independently of the regimen selected, there is a decrease of bone mass of around 2-4% and this decrease is greater in those patients that start a regimen that contains tenofovir. The results of this study were similar to the ones reported in the Retrovirus Conference in San Francisco in February by another ACTG study (A5224)22. The next big question in the bone area is going to be the evaluation of interventions that can prevent this initial bone loss after the initiation of treatment, which has been universally seen in trials that have looked at this particular issue. Roger Bedimo presented data about the fracture risk in patients with HIV infection in the large VA database23. The study confirmed the high prevalence of fractures in patients with HIV, and also showed that hepatitis C co-infection is an independent risk factor for this. I think the most important finding of this study was to show, for the first time, that fractures in HIV patients are independently associated with mortality. I think that should settle the question about the clinical relevance of the low bone mineral density that we see so frequently in our patients.
Another substudy of A5142 was presented 24showing the individuals the start tenofovir have a more significant decline in creatinine clearance than individuals the start other regimens, and that the decrease in creatinine clearance is greater when tenofovir is combined with boosted protease inhibitors. The good news in the study was that the cases of frank kidney failure were very rare. Similar results were also presented during the Retrovirus Conference in San Francisco25. I think the next big question in this area is going to be the long-term implications of these small decreases in creatinine clearance that are observed in patients receiving tenofovir. So far the data doesn't look too worrisome, but we need to keep an eye on it.
There was a presentation of a new clinical entity, fortunately very rare, a T8 infiltrative encephalitis in patients with HIV infection that was almost universally fatal26 (a total of 10 cases). We need more data about how frequent this process really is. It is something to think about when a clinician evaluates a patient with HIV infection and unexplained encephalitis.
A whole session27 was devoted to epidemiologic studies showing the high prevalence of low vitamin D. levels among patients with HIV infection. There was not much new information in this session beyond what is already known. We're going to need studies about how to best correct these deficiencies and what is the role that different antiretroviral regimens play in the development of low vitamin D levels.
Eradication and inflammation
As it was in the retrovirus conference these two topics dominated plenaries and symposia. Not much new data was presented. The big challenge in HIV research for the next decade is to find out if it is possible to eliminate all HIV from the body, basically "curing" infected individuals, or if not, if it is possible to find a "functional cure" that would maintain viral suppression in patients with HIV without the need for antiretroviral therapy.
References:
1. http://www.aids2010.org/
2. http://www.viennadeclaration.com/
3. http://www.theglobalfund.org
4. http://www.kff.org/hivaids/upload/7347-06.pdf
5. http://www.nytimes.com/2010/07/21/opinion/21tutu.html
6. Funk MJ, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. THLBB201
7. Cohen C, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. THLBB206.
8. Gathe J, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. THLBB202
9. Arribas J, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. THLBB205.
10. Kumar P, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. THPE0111
11. Young B, et al. 18th IAC; Vienna, July 18-23, 2010; Absts. THPE0112 and THPE0163.
12. Reynes J, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. MOAB0101
13. Kozal M, et al. 18th IAC; Vienna, July 18-23, 2010; Abst.. THLBB204
14. Mills A, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. THLBB203.
15. De Luca A, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. THLBB207
16. Rieger A, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. THLBB209.
17. Blanc F, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. THLBB106.
18. Abdool Karim Q, et al. 18th IAC; Vienna, July 18-23, 2010; Abst TUSS0502.
Also Karim QA, Karim SSA, Frohlich JA, et al, on behalf of the CAPRISA 004 Trial Group. Effectiveness and safety of tenofovir gel, an antiretroviral microbicide, for the prevention of HIV infection in women. Science Express. Published online July 19 2010. Available at: http://www.sciencemag.org/cgi/rapidpdf/science.1193748.pdf Accessed August 5, 2010.
19. Zolopa AR et al. Early antiretroviral therapy reduces AIDS progression/death in individuals with acute opportunistic infections: A multicenter randomized strategy trial. PLoS One2009 May 18; 4:e5575.
20. Severe P, Jean Juste MA, Ambroise A, et al. Early versus Standard Antiretroviral Therapy for HIV-Infected Adults in Haiti. N Engl J Med 2010; 363:257-265
21. Huang, J et al. 18th IAC; Vienna, July 18-23, 2010; Abst. WEAB0304.
22. Bone and Limb Fat Outcomes of ACTG A5224s, a Substudy of ACTG A5202: A Prospective, Randomized, Partially Blinded Phase III Trial of ABC/3TC or TDF/ FTC with EFV or ATV/r for Initial Treatment of HIV-1 Infection. Grace McComsey , D Kitch , E Daar , C Tierney , N Jahed , P Tebas , L Myers , P Sax , and AACTG Study A5224. Tien10 Program of the Conference of Retrovirus and Opportunistic Infections. San Francisco February 16-19, 2010. Oral 106LB
23. Bedimo R, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. TUAB0104.
24. Goicochea M, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. WEAB0305.
25. Chronic Kidney Disease and Exposure to ART in a Large Cohort with Long-term Follow-up: The EuroSIDA Study. Ole Kirk , A Mocroft , P Reiss , S De Wit , D Sedlacek , M Beniowski , J Gatell , A Phillips , B Ledergerber , J Lundgren , , for the EuroSIDA Study Group Program of the Conference of Retrovirus and Opportunistic Infections. San Francisco February 16-19, 2010. Oral LB 107
26. Lescure FX, et al. 18th IAC; Vienna, July 18-23, 2010; Abst. WEAB0303.
27. Poster discussion. Shining Light on Vitamin D deficiency. Vienna, July 18-23, 2010; Session. WEPDB1.
|
| |
|
 |
 |
|
|