 |
 |
 |
| |
Raltegravir Pharmacokinetics during Pregnancy
|
| |
| |
Reported by Jules Levin
ICAAC Sept 14 2010 Boston
B. M. Best1, E. V. Capparelli1, A. Stek2, S. K. Burchett3, Y. Huo4, F. Aweeka5, E. Smith6, J. S. Read7, M. Mirochnick81Univ. of California San Diego, CA, USA; 2Univ. of Southern California, Los Angeles, CA, USA; 3Children's Hosp Boston, MA, USA; 4Harvard Sch. of Publ. Health, Boston, MA, USA; 5Univ. of California, San Francisco, CA, USA; 6NIAID, Bethesda, MD, USA; 7NICHD, Bethesda, MD, USA; 8Boston Univ., Boston, MA, USA
CONCLUSIONS
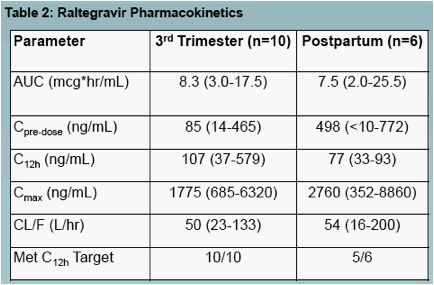
Consistent with previous reports, raltegravir pharmacokinetics showed extensive variability
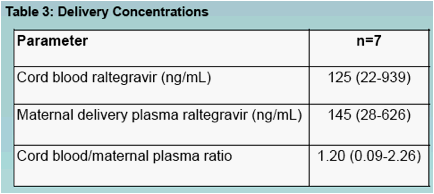
Raltegravir readily crossed the placenta
Raltegravir exposure was not consistently altered during 3rd trimester compared to postpartum and historical data, and the standard dose appears appropriate during pregnancy.



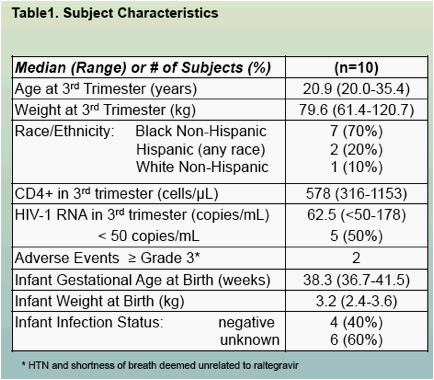
Ten pregnant women completed 3rd trimester visit, six of these have completed postpartum visits (table 1)
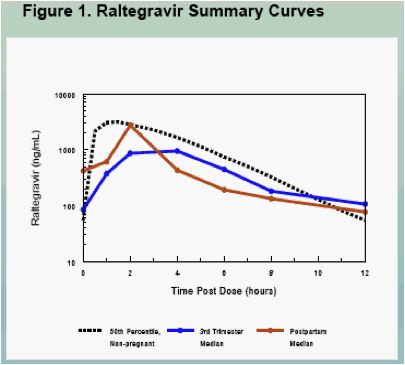
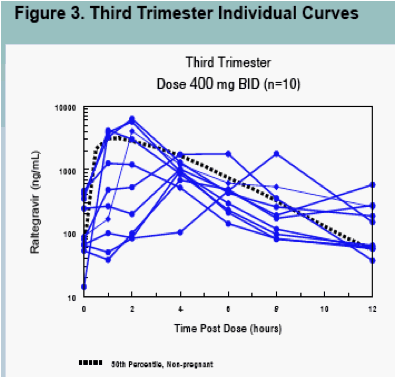
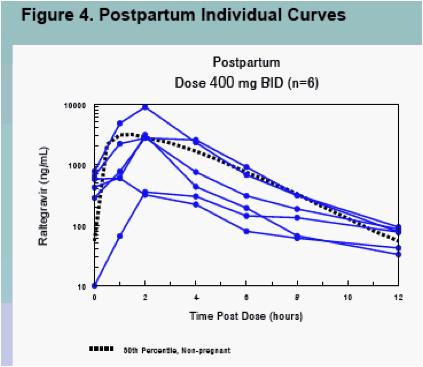
Raltegravir pharmacokinetics were highly variable, but were not significantly different between pregnancy and postpartum (Figures 1, 3 and 4, Table 2)
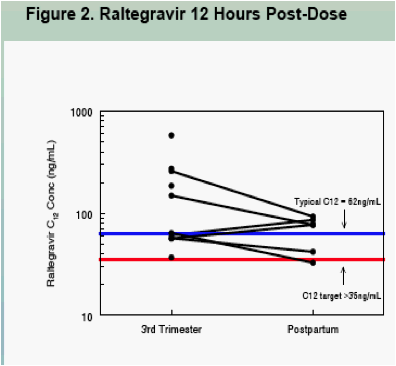
All 3rd trimester concentrations drawn 12 hours after a witnessed dose were above the minimum cut-off value of 35 ng/mL (figure 2)
Cord blood and maternal plasma raltegravir concentrations were similar (Table 3)

|
| |
|
 |
 |
|
|