 |
 |
 |
| |
Multicenter Evaluation of the Elecsys® HBsAg II Quant Assay
|
| |
| |
Reported by Jules Levin
Presented at The 21st Conference of the Asian Pacific Association for the Study of the Liver (APASL 2011), 17-20 February 2011, Bangkok, Thailand
F Bonino1, S Bowden2, M Brunetto3, S Louisirirotchanakul4, T Tanwandee5, P Phisalprapa4, B Zacher6, K Wursthorn6, H Wedemeyer6
1. Scientific Direction and Central Laboratory, Fondazione IRCCS
Orspedale Maggiore Policlinico Mangiagalli e Regina Elena di Milano,
University of Pisa, Italy
2. Melbourne Health, Victorian Infectious Disease Reference Laboratory
(VIDRL), Melbourne, Victoria, Australia
3. Laboratory of Molecular Genetics of Hepatitis Viruses, Hepatology Unit,
University Hospital of Pisa, Italy
4. Division of Gastroenterology, Department of Medicine, Faculty of
Medicine, Siriraj Hospital, Mahidol University, Bangkok, Thailand
5. Department of Gastroenterology, Department of Medicine, Bangkok Siriraj
Hospital, Mahidol University, Bangkok, Thailand
6. Hanover Medical School (MHH), Department of Gastroenterology,
Hepatology and Endocrinology, Hanover, Germany
Author Conclusions
The Elecsys HBsAg II quant assay reliably quantified HBsAg in routine clinical samples across all major HBV genotypes encountered globally, and at all stages of infection tested, including in sera with high HBsAg levels (up to 873,300 IU/mL).
The assay gave exact results over a broad linear range (0.05-52,000 IU/mL) reflecting clinically relevant HBsAg titers, and is optimized to reliably quantify HBsAg in samples encountered in clinical practice.
The defined onboard dilution step reduces the need for retesting; more than 70% of the samples gave a final result on first analysis.
Onboard dilution reduces the potential for pipetting errors, lowers "hands-on" time and saves costs. Onboard reagents are stable for up to 8 weeks, improving convenience and reducing waste.
The assay showed high precision across the entire measuring range. The high precision of the assay is particularly important if serum HBsAg titers are to be compared between different samples from the same patient, for example, during on-treatment monitoring.
The Elecsys HBsAg II quant assay is standardized against the WHO Second International Standard for HBsAg, ensuring the reliability of HBsAg values. This is important when comparing values between different laboratories.
The Elecsys HBsAg II quant assay is highly suitable for the quantification of HBsAg levels in routine clinical samples, providing accurate, standardized results for clinical decision making.
Background:
· Clearance of hepatitis B surface antigen (HBsAg) during the natural course of chronic hepatitis B infection (CHB) is associated with improved long-term clinical outcome, including reduced incidence of cirrhosis and hepatocellular carcinoma, and longer survival1,2.
· HBsAg clearance is, therefore, considered to be the best definition of virologic response to treatment and the closest outcome to clinical cure in CHB3.
· Studies have shown that the degree and timing of reductions in HBsAg levels during pegylated interferon (PEG-IFN)-based therapy may predict sustained response in both hepatitis B 'e' antigen (HBeAg)-positive and HBeAg-negative CHB4-9 and may also predict response to treatment with some nucleos(t)ide analogs10,11.
· HBsAg levels may also indicate disease state, and so differentiate between patients with inactive and active disease12,13
.
· Based on these findings, there is increasing interest in the role of quantitative HBsAg in the individualization of CHB management and therapy14, and therefore a need for accurate, simple, standardized and widely available quantitative HBsAg assays to ensure optimal clinical decision making.
· The Elecsys® HBsAg II quant assay is a new quantitative electrochemiluminescence immunoassay which uses onboard dilution and has a broad linear range reflecting clinically relevant HBsAg titers.
· The assay is standardized against the WHO Second International Standard for HBsAg.
Aim:
This international multicenter study aimed to evaluate the performance of the Elecsys HBsAg II quant assay using routine serum samples from treated
and untreated patients with CHB, in different phases of infection, representing all major hepatitis B virus (HBV) genotypes.
Methods:
Description of the assay
· The automated Elecsys HBsAg II quant assay procedure is as follows: two biotinylated monoclonal anti-HBsAg antibodies and a mixture of ruthenium-labeled monoclonal anti-HBsAg antibodies and polyclonal anti-HBsAg antibodies form a sandwich complex with HBsAg in the sample.
Streptavidin-coated microparticles are added and the complex immobilized via interaction with biotin and streptavidin. The microparticles are magnetically captured and a voltage applied to induce chemiluminescence that is measured by a photomultiplier. The total assay time is 18 minutes.
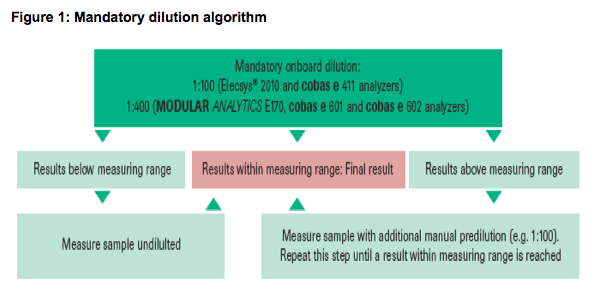
· Every sample has to be diluted; onboard dilution is performed by the analyzer according to the algorithm shown in figure 1.
Samples
· Samples from patients with confirmed HBeAg-positive or -negative CHB (HBV DNA positive and HBsAg positive for >6 months), including follow-up
samples from patients with or without antiviral treatment, were collected independently by five centers: three in Europe (Hanover Medical School (MHH),
Germany [e 411 analyzer]; University Hospital of Pisa, Italy [e 411 analyzer]; Roche Diagnostics GmbH, Penzberg, Germany [Elecsys 2010 analyzer]), one in Asia
(Siriraj Hospital, Bangkok, Thailand [E170 analyzer]) and one in Australia (Victorian Infectious Diseases Reference Laboratory, Australia [e 411 analyzer]).
Analyses
· Dilution analyses (undiluted to 1:1,000,000) of all sera were performed to assess the mandatory dilution algorithm recommended by the manufacturer,
determine the assay range free of high-dose hook effect, and investigate any effect of HBV genotype or disease stage.
· Intra- and inter-assay precision were established by a single center (Penzberg) using Elecsys HBsAg II quant reagents (Roche Diagnostics), human sera
and controls (Elecsys PreciControl HBsAg II, Roche Diagnostics) in a protocol (EP5-A2) of the Clinical and Laboratory Standards Institute
(CLSI; http://www.clsi.org/). Two runs were performed per day in duplicate for each of 21 days
Results
A total of 611 serum samples from 345 patients were included in the analyses (table 1).
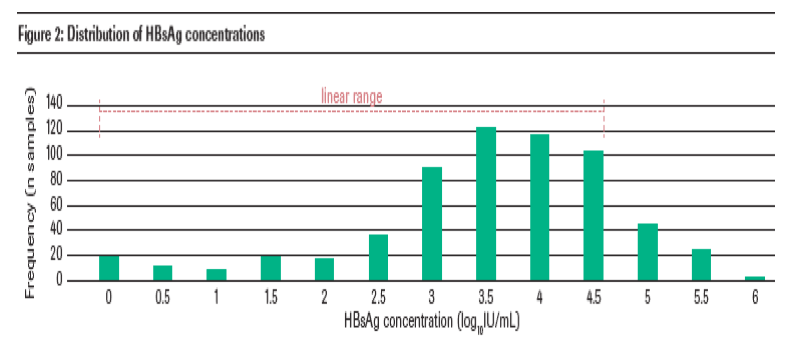
The overall distribution of HBsAg concentrations found in the samples is shown in figure 2. The median HBsAg concentration in the 611 samples was 2,763 IU/mL (range 0.11-873,300 IU/mL). The majority of samples had HBsAg levels within the linear range of the Elecsys HBsAg II quant assay (0.05-13,000 IU/mL for the Elecsys 2010 and cobas e 411 analyzers and 0.05-52,000 IU/mL for the MODULAR ANALYTICS E170, cobas e 601 and cobas e 602 modules).
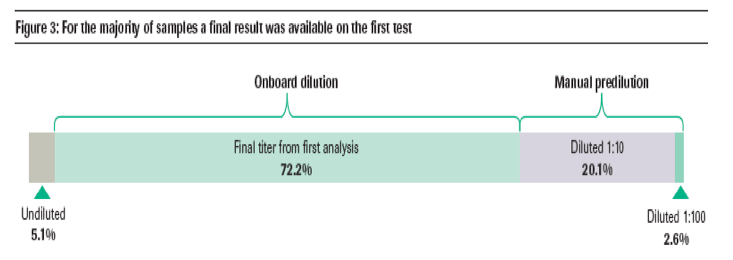
70.0% and 85.6% of samples tested on analyzers incorporating 1:100 (Elecsys 2010; cobas e 411) and 1:400 (MODULAR ANALYTICS E170) onboard dilution, respectively, provided a final result on first test.
Across all platforms, 72.2% of samples had a final titer which was measurable using the initial onboard dilution.
- The majority of samples above the measuring range on first analysis required only a single reanalysis using a manual predilution step of 1:10.
-- ~2% of samples required a second manual dilution step of 1:100 (figure 3).
No high-dose hook effect was seen when following the mandatory dilution algorithm up to the maximum HBsAg serum level tested (873,300 IU/mL).
-- The deviation of the final titer (lowest dilution step of a sample resulting in a value within the measuring range) to the titer obtained by the previous/subsequent dilution steps was very low (mean -0.464% [standard deviation 4.83%]; 95% percentile: -7.9% to +12.1%).
HBsAg levels were reliably determined across all genotypes (HBV genotypes A-G).
- Dilution analyses showed a linear relationship for all genotypes tested, with no evidence of high-dose hook effect when following the mandatory dilution algorithm.
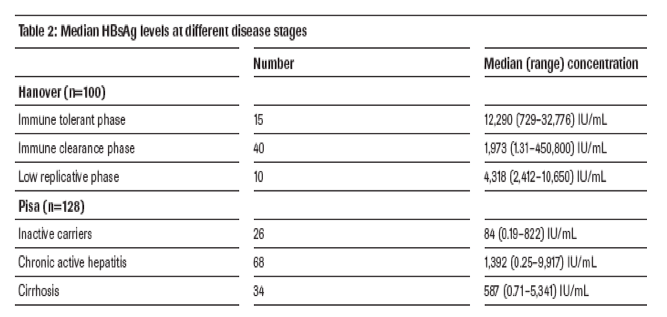
Median HBsAg levels at different disease stages are shown in table 2.
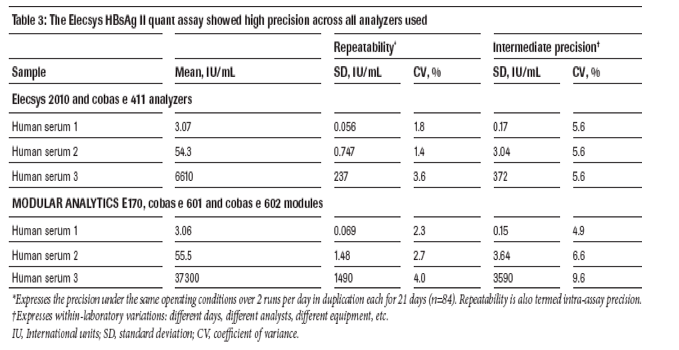
The Elecsys HBsAg II quant assay showed high precision across all analyzers used (CV%: 1.4-9.6; HBsAg concentrations 0.1-37,300 IU/mL) (table 3).
References
1. AASLD guidelines. Chronic hepatitis B: update 2009. Lok AS and McMahon BJ. Hepatology 2009;50:1-36
2. EASL Clinical Practice Guidelines: Management of chronic hepatitis B.
European Association for the Study of the Liver. J Hepatol 2009;50:227-242
3. Diagnostic markers of chronic hepatitis B infection and disease. Bonino F et al. Antivir Ther 2010;15(Suppl 3):35-44
4. On-treatment HBsAg decline during peginterferon alpha-2a (40KD) + lamivudine in patients with HBeAg-positive CHB as a potential predictor of durable off-treatment response. Lau GK et al. Hepatolology 2008;48:A910
5. In patients with HBeAg-negative chronic hepatitis B HBsAg serum levels early during treatment with peginterferon alfa-2a predict HBsAg clearance 4 years post-treatment. Marcellin P et al. Hepatolology 2008;48:A919
6. Hepatitis B surface antigen serum levels help to distinguish active from inactive hepatitis B virus genotype D carriers. Brunetto MR et al. Gastroenterology 2010;139:483-490
7. High rates of HBsAg seroconversion in HBeAg-positive chronic hepatitis B patients responding to interferon: A long-term follow-up study. Moucari R et al. Hepatology 2009;49:1151-1157
8. On-treatment decline in serum HBsAg levels predicts sustained immune control 1 year post-treatment and subsequent HBsAg clearance in HBeAg-negative hepatitis B virus-infected patients treated with peginterferon alfa-2a [40KD] (PEGASYS). Marcellin P et al. Hepatol Int 2010;4:151, abstract PP209
9. On-treatment decline in serum HBsAg levels predicts sustained immune control 6 months post-treatment and subsequent HBsAg clearance in HBeAg-positive hepatitis B virus-infected patients treated with
peginterferon alfa-2a [40KD] (PEGASYS). Piratvisuth T et al. Hepatol Int 2010;4:152, abstract PP211
10. Three-year efficacy and safety of tenofovir disoproxil fumarate treatment for chronic hepatitis B. Heathcote EJ et al. Gastroenterology 2011;140:132-143
11. Kinetics of hepatitis B surface antigen decline during 3 years of telbivudine treatment in hepatitis B e antigen-positive patients. Wursthorn K et al. Hepatology 2010;52:1611-1620
12. Hepatitis B surface antigen (HBsAg) levels in the natural history of hepatitis B virus (HBV)-infection: a European perspective. Jaroszewicz J et al. J Hepatol 2010;52:514-522
13. Quantitative HBsAg: A new specific marker for the diagnosis of HBsAg inactive carriage. Martinot-Peignoux M et al. J Hepatol 2010;52(Suppl 1):S282
14. A new role for an old marker, HBsAg. Brunetto MR. J Hepatol 2010;52:475-477
|
| |
|
 |
 |
|
|