 |
 |
 |
| |
Response rates with shorter duration and lower doses of peginterferon alfa-2a are inferior to those with 180 μg for 48 weeks
|
| |
| |
Reported by Jules Levin
Presented at The 21st Conference of the Asian Pacific Association for the Study of the Liver (APASL 2011), 17-20 February 2011, Bangkok, Thailand
Q Xie,1 J Sung,2 T Tanwandee,3 KH Han,4 C Wat,5 C-W Hsu,6 J Jia7
1Department of Infectious Diseases, Ruijin Hospital, Shanghai, China; 2Department of Medicine and Therapeutics, Prince of Wales Hospital, Hong Kong;
3Department of Microbiology, Faculty of Medicine, Siriraj Hospital, Mahidol University, Bangkok, Thailand;
4Department of Internal Medicine, Yonsei University College of Medicine, Seoul, Korea; 5Roche Products Ltd, Welwyn Garden City, UK;
6Liver Research Unit, Chang Gung Memorial Hospital and Chang Gung University College of Medicine Taipei, Taiwan; 7Beijing Friendship Hospital, Capital Medical University, Beijing, China
AUTHOR CONCLUSION
The NEPTUNE study confirms that compared with lower doses and shorter durations, the licensed dose (180 μg/week) and duration (48 weeks) of PEG-IFN alfa-2a is the most efficacious and beneficial treatment regimen for CHB patients
AUTHOR SUMMARY
Rate of HBeAg seroconversion 6 months post-treatment was higher in the 180 μg/48-week arm than in the other treatment arms (36.2% compared with 14.1-25.8% in the other treatment arms). The 90 μg/week dose was inferior to the 180 μg/week dose and 24 weeks of treatment were inferior to 48 weeks of treatment for the primary efficacy endpoint
Response to peginterferon alfa-2 therapy is known to increase post-treatment.1 In the NEPTUNE study, HBeAg seroconversion increased in the 24-week arms between 6 months and 12 months post-treatment (week 72 time point). However, they did not reach the levels achieved with the licensed regimen (180 μg/48 weeks) 6 months post-treatment (the 12-month post-treatment time point had not been reached for the 48-week arms)
Rates of response were consistently higher in the 180 μg/48-week arm for secondary endpoints
High rates of response were achieved in the 180/48-week arm in patients infected with genotype B or genotype C (42.2% and 38.8%, respectively, for HBeAg seroconversion) and a similar trend was observed for all secondary efficacy parameters
BACKGROUND
Peginterferon alfa-2a (40 kDa; PEG-IFN alfa-2a; PEGASYS) has proven efficacy in hepatitis B e antigen (HBeAg)-positive chronic hepatitis B (CHB)1 and is recommended as a first-line therapy for CHB by all international treatment guidelines2-4
Although the licensed dose and duration of PEG-IFN alfa 2a is 180 μg/week for 48 weeks in all countries, there is currently a lack of consensus on the most appropriate dose and duration, which is reflected in varying recommendations in international guidelines2-4
The lack of consensus may result from varying interpretations of the results from the phase 25 and the phase 31 studies of PEG-IFN alfa-2a in HBeAg-positive patients. In the phase 2 study (N=194), patients received PEG-IFN alfa-2a (90, 180 or 270 μg/week) or conventional interferon for 24 weeks.5 Rates of sustained immune control (HBeAg seroconversion 6 months post-treatment) were similar in all three PEG-IFN alfa-2a groups (32%) and significantly higher than in the interferon group (25%). In the phase 3 study, patients (N=271) received a finite 48-week course of PEG-IFN alfa-2a (180 μg/week) and 32% of patients achieved HBeAg seroconversion 6 months post-treatment1
As neither the phase 2 study nor the phase 3 study was designed to identify both the most appropriate dose and duration of PEG-IFN alfa-2a in HBeAg-positive patients and cross-study comparisons are not appropriate, a single study comparing different treatment regimens was required. The NEPTUNE study was designed to establish the most appropriate PEG-IFN alfa-2a treatment regimen for HBeAg-positive patients
Objective
The aim of the NEPTUNE study was to compare the efficacy and safety of PEG-IFN alfa-2a given for either 24 or 48 weeks and at doses of either 90 μg/week or 180 μg/week
Methods
This was a phase 4, double-blind, randomized, multicenter trial with a 2 x 2 factorial design (Figure 1). HBeAg-positive patients were randomized (1:1:1:1) to PEG-IFN alfa-2a (90 μg/week or 180 μg/week for 24 or 48 weeks) by subcutaneous injection once weekly. Patients were stratified by baseline alanine aminotransferase (ALT) levels: > upper limit of normal (ULN) - 2 x ULN; 2-5 x ULN; 5-10 x ULN and viral genotype (A, B, D or C plus other genotypes)
Figure 1. NEPTUNE Study: a 2 x 2 factorial design non-inferiority study in HBeAg-positive patients
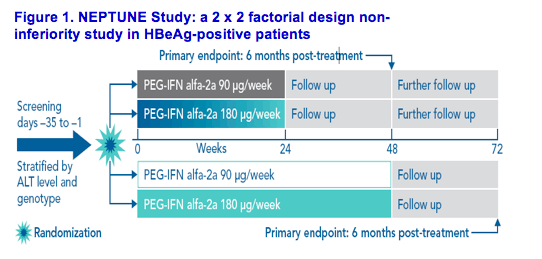
The primary efficacy parameter was HBeAg seroconversion 6 months post-treatment. Secondary endpoints included HBeAg loss, HBsAg seroconversion, HBsAg clearance, ALT normalization, HBV DNA <2,000 IU/mL and HBV DNA <20,000 IU/mL and a triple endpoint of HBeAg seroconversion, ALT normalization and HBV DNA <20,000 IU/mL. All secondary endpoints were assessed 6 months post-treatment. As rates of response to PEG-IFN alfa-2a are known to increase post-treatment, HBeAg seroconversion was also assessed at week 72 (6 months post-treatment for the 48-week arms and 12 months post-treatment for the 24-week arms)
Statistical Analysis
The null hypotheses were that 24 weeks is inferior to 48 weeks and 90 μg/week is inferior to 180 μg/week. The primary test statistic was the odds ratio (OR) with a non inferiority margin pre-specified as 1.88. OR 48/24 or 180/90 was significant if the upper 95% confidence interval (CI) was <1.88 at the significance level of 0.025; the null hypothesis would be rejected
RESULTS
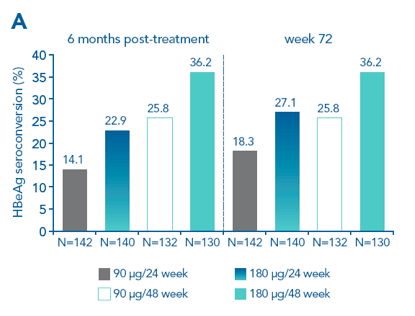
Highest rates of HBeAg seroconversion 6 months post-treatment were achieved in the 180 μg/48-week arm (36.2%; Figure 2), which is the licensed dose and duration for PEG-IFN alfa-2a
At week 72, response rates in the 24-week arm (12 months post-treatment) had increased to 18.3% in the 90 ug/24 week-arm and 27.1% in the 180 ug/24-week arm, but did not reach the levels achieved in the 180 ug/48-week arm 6 months post-treatment
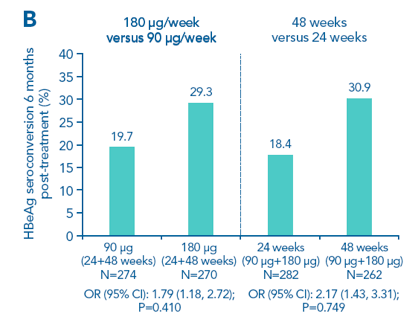
To test the null hypotheses, the duration groups and dose groups were pooled. For both comparisons, the null hypotheses were retained: the upper 95% CI was >1.88 and the p values for non inferiority were not significant. This demonstrates that 90 μg/week is inferior to 180 μg/week and that 24 weeks of therapy are inferior to 48 weeks of therapy
Figure 2. Highest rates of HBeAg seroconversion in the 180 μg/48-week arm. A. Response rates in the four treatment arms; B. Non-inferiority analyses of 90 μg/week versus 180 μg/week and 24 weeks versus 48 weeks
Highest rates of all secondary endpoint parameters were achieve in the 180 ug/48-week arm (Table 1). In total, three patients in each of the 48-week treatment arms achieved HBsAg clearance compared with only one patient in the 90 μg/24-week arm and no patients in the 180 μg/24-week arm
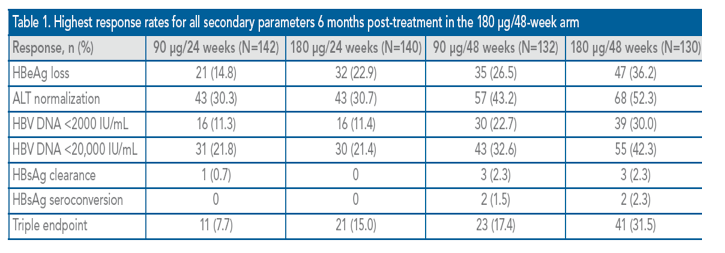
In the majority of cases where hypothesis testing was conducted for secondary endpoints, 90 μg/week was inferior to 180 μg/week and 24 weeks of therapy were inferior to 48 weeks of therapy (Table 2). For ALT normalization, the null hypothesis of inferiority of 90 μg/week compared with 180 μg/week showed borderline non-inferiority
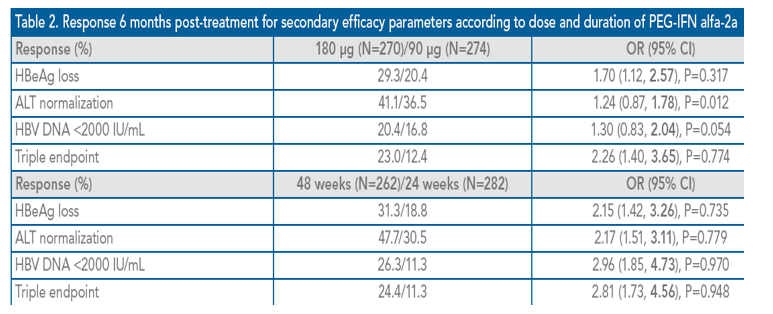
Genotype Subanalysis
A small number of patients were infected with genotype A (N=22) or genotype D (N=38). Rates of HBeAg seroconversion in the 90 μg/24-week, 180 μg/24-week, 90 μg/48-week and 180 μg/48-week arms were 0%, 12.5%, 33.3% and 25.0%, respectively, in genotype A-infected patients and 9.1%, 33.3%, 18.2% and 0%, respectively, in genotype D-infected patients. As a result of the small numbers of patients infected with genotypes A and D, comparisons between treatment arms are not appropriate
The majority of patients included in the analysis were infected with either genotype B (N=186; 34.2%) or genotype C (N=281, 51.7%)
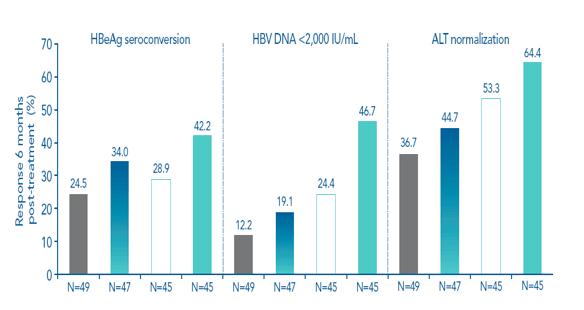
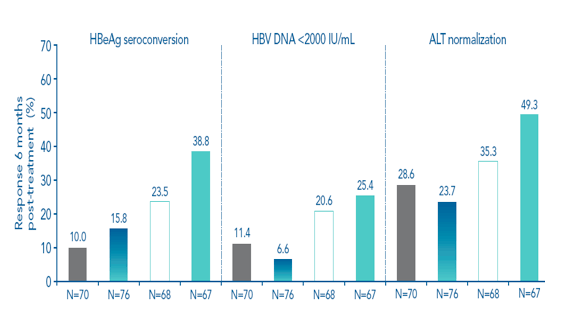
The highest response rates for the primary and secondary efficacy endpoints were achieved in the 180 μg/48-week arm for both genotype B- and genotype C-infected patients (Figure 3). Rates of HBeAg seroconversion 6 months post treatment were similar in genotypes B and C
Figure 3. High rates of response were achieved in the 180 μg/48-week arm in patients infected with genotype B or genotype C
Genotype B
Genotype C
References
1. Lau GK et al. Peginterferon alfa-2a, lamivudine and the combination for HBeAg positive chronic hepatitis B. N Engl J Med 2005;352:2682-2695
v
2. European Association for the Study of the Liver. EASL clinical practice guidelines: management of chronic hepatitis B. J Hepatol 2009;50:227-242
3. Liaw YF et al. Asian-Pacific consensus statement on the management of chronic hepatitis B: a 2008 update. Hepatol Int 2008;2:263-283
4. Lok A, McMahon B. Chronic hepatitis B: update 2009. http://www.aasld.org/practiceguidelines/Documents/Bookmarked%20Practice
%20Guidelines/Chronic_Hep_B_Update_2009%208_24_2009.pdf
5. Cooksley WGE et al. Peginterferon alfa-2a (40 kDa): an advance in the treatment of hepatitis B e antigen-positive chronic hepatitis B. J Viral Hepatitis 2003;10:298-305
Disclosure information: Editorial support for the development of this poster was funded by F. Hoffmann-La Roche, Basel, Switzerland
|
| |
|
 |
 |
|
|