 |
 |
 |
| |
Four Year Efficacy and Safety of Tenofovir DF in HBeAg-Negative and HBeAg-Positive Patients with Chronic Hepatitis B (CHB)
|
| |
| |
Reported by Jules Levin
Presented at 21st Conference of the Asian Pacific Association for the Study of the Liver
(APASL)
February 17-20, 2011
Bangkok, Thailand
P Marcellin1, E J Heathcote2, M Buti3, E Gane4, Z Krastev5, R A de Man6, S Gurel7, K Borroto-Esoda8, D H Coombs8, E Mondou8, and J Anderson8
1Hopital Beaujon, University of Paris, Clichy, France; 2University of Toronto, Toronto, ON, Canada; 3Servicio de Medicina Interna Hepatologia, Hospital General Universitari Vall d'Hebron and Ciberehd, Barcelona, Spain; 4Middlemore Hospital, Auckland, New Zealand; 5University Hospital "St. Ivan Rilsky", Sofi a, Bulgaria; 6Erasmus MC, University Medical Center Rotterdam, Rotterdam, Netherlands; 7University of Uludag, Bursa, Turkey; 8Gilead Sciences, Foster City, CA, USA

INTRODUCTION
· Tenofovir DF (TDF) approved for HIV-1 in 2001 and chronic hepatitis B (CHB) in 2008: ~ 3.5 million patient-years
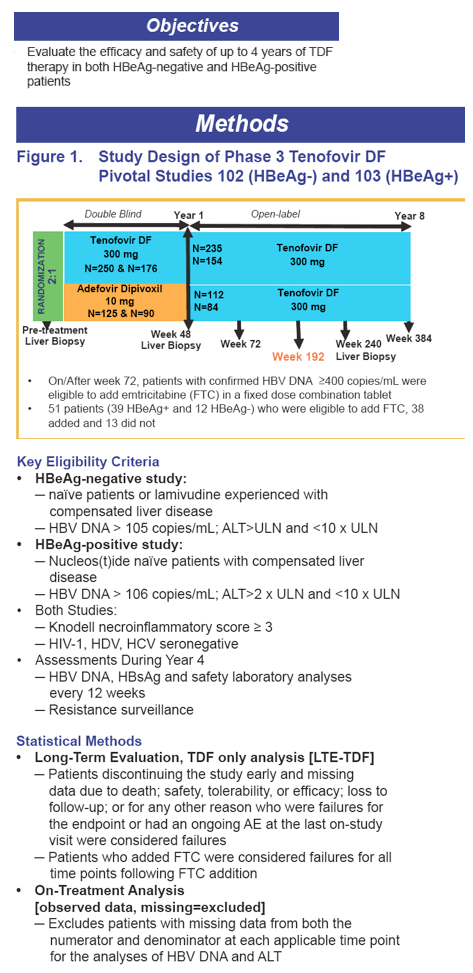
· Week 48 Phase 3 data showed a significantly greater antiviral activity of TDF compared to adefovir dipivoxil (ADV):
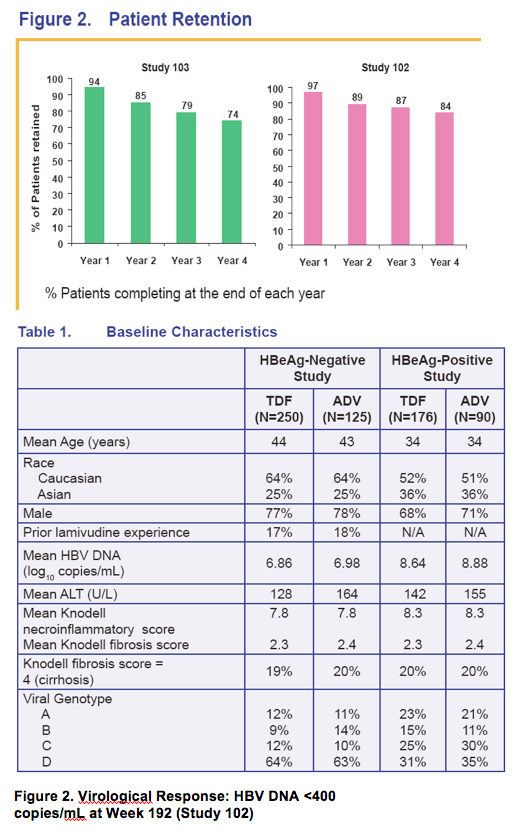
- HBeAg-negative patients: 93% vs 63% HBV DNA <400 copies/mL
- HBeAg-positive patients: 76% vs 13% HBV DNA <400 copies/mL
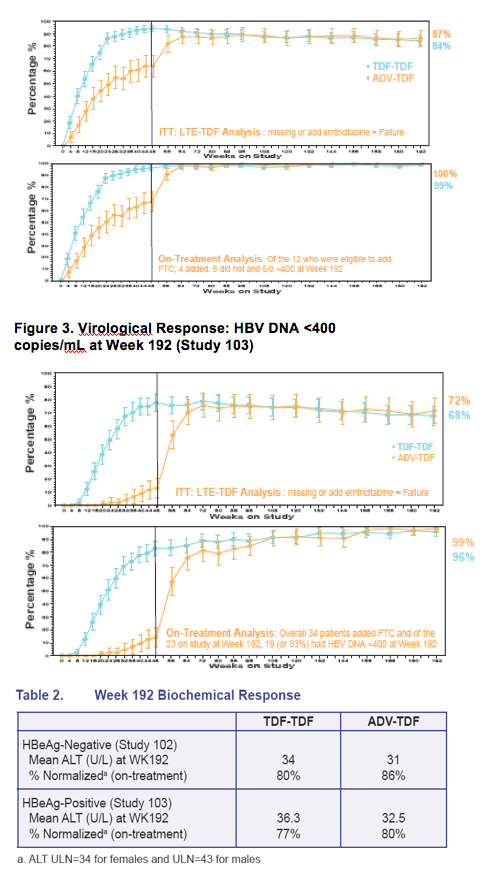
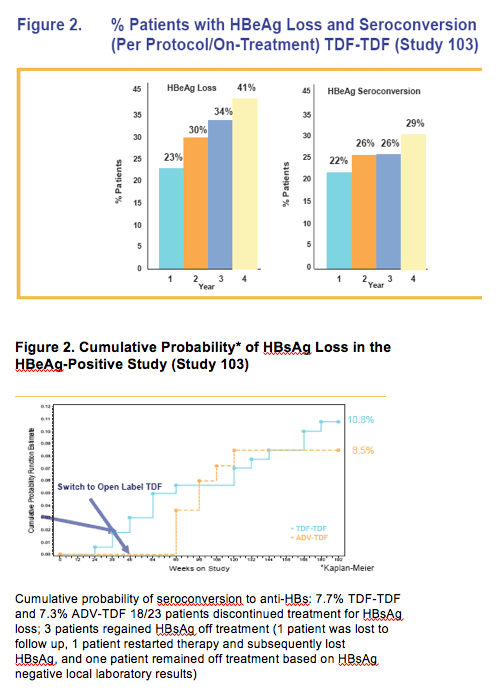
· TDF treatment beyond Week 48 showed
- Both viremic and nonviremic patients on ADV can effectively switch to TDF and achieve or maintain viral suppression (HBV DNA < 400 copies/mL) and normal ALT at week 192
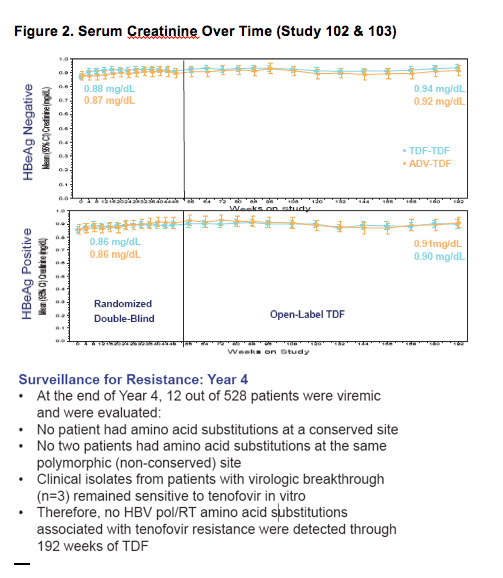
- TDF patients treated for 192 weeks maintained HBV DNA < 400 copies/mL and normal ALT levels
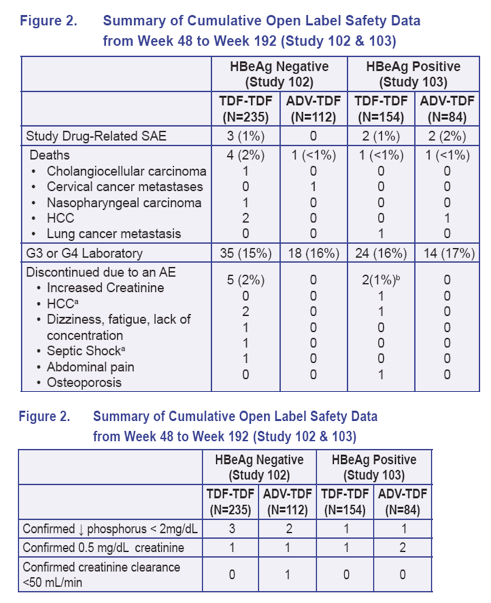
102 & 103: Decreases in phosphorus were transient and resolved on treatment without intervention
103: TDF-TDF confirmed creatinine increase, peak creatinine 1.5 mg/dL at week
192; patient remains on treatment at full dose
103: ADV-TDF patients had an initial ≥0.5 mg/dL increase in creatinine on ADV that was confirmed after switching to TDF. One patient had an increase (grade 1) to a peak of 1.8 mg/dL (nadir creatinine clearance 44 mL/ mIn); patient was dose adjusted and remains on treatment at week 192 (creatinine=1.3 mg/dL). The other
patient had a peak creatinine of 1.7 mg/dL (grade 1), patient was dose reduced, and creatinine improved to 1.4 mg/dL at week 144/last available time point
One additional ADV-TDF patient who had a grade 1 creatinine on ADV had a transient grade 1 increase to 1.6 mg/dL at week 96 (0.1 increase from baseline). Patient remains stable and on study without interruption or modification
102: TDF-TDF patient w/creatinine increase was associated with advanced HCC/death; ADV-TDF patient, creatinine increase at week 180 to peak of 1.3 mg/dL (and concurrent creatinine clearance of 48 mL/min) remains on study with a dose reduction to every other day
|
| |
|
 |
 |
|
|