 |
 |
 |
| |
High Sustained Virologic Response Among Genotype 1 Previous Non-responders and Relapsers to
Peginterferon/Ribavirin when Re-treated With Boceprevir Plus Peginterferon Alfa-2a/Ribavirin
|
| |
| |
Reported by Jules Levin
EASL 2011 Berlin Germany March 30-Apr 2
S. Flamm*1, E. Lawitz2, I. Jacobson3, R. Rubin4, M. Bourliere5, C. Hezode6, J. Vierling7, C. Niederau8, M. Sherman9, V. Goteti10, R. Vilchez10, C. Brass10, J. Albrecht10, and F. Poordad11
1Northwestern Feinberg School of Medicine, Chicago, IL; 2Alamo Medical Research, San Antonio, TX; 3Weill Cornell Medical College, New York, NY; 4Liver Center of Atlanta, Atlanta, GA; 5Fondation Hopital Saint Joseph, Marseille, France; 6A.P.H. Paris, Hopital Henri Mondor,
Creteil Cedex, France; 7Baylor College of Medicine, Houston, TX; 8St. Josef-Hospital Oberhausen, Oberhausen, Germany; 9Toronto General Hospital, Toronto, Canada; 10Merck Sharp & Dohme Corp., Whitehouse Station, NJ, USA; 11Cedars-Sinai Medical Center, Los Angeles, CA; *s-flamm@northwestern.edu
AUTHOR CONCLUSIONS
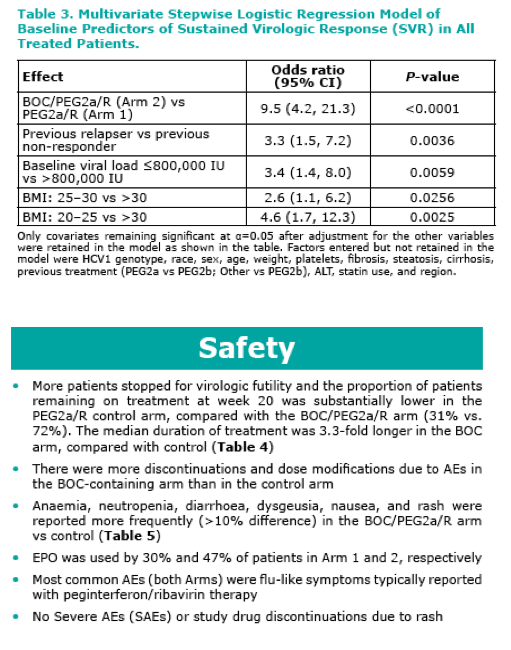
Adding BOC to PEG2a/R for retreatment of patients who have previously failed P/R therapy results in a statistically significant and clinically meaningful increase in efficacy (64% vs 21% for control, P<0.0001)
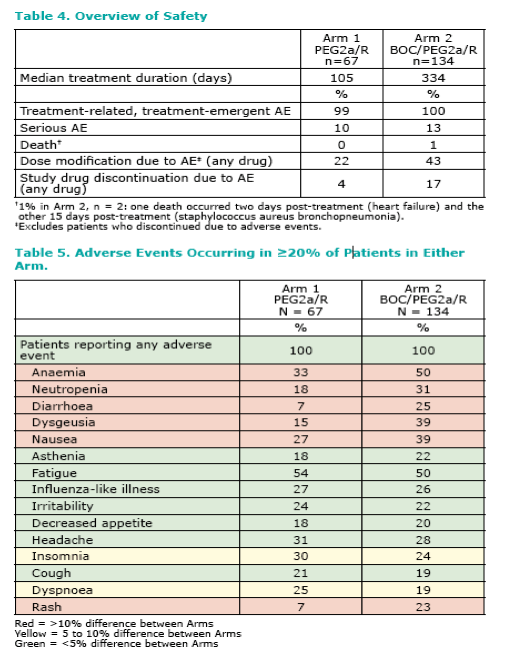
Therapy was well tolerated and most of the safety findings observed with BOC and PEG2a/R in this study were similar to those observed with BOC and PEG2b/R
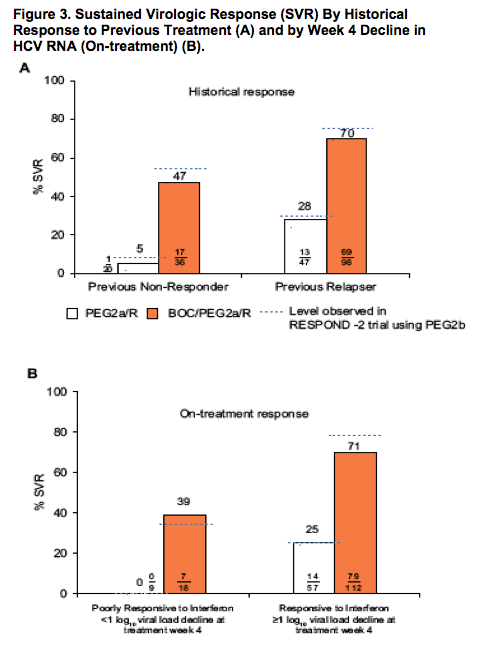
The addition of BOC substantially increased SVR in patients who were poorly responsive to interferon, as determined by the 4-week lead-in
BOC is safe and effective with both peginterferon alfa2a and 2b
BACKGROUND
· More than 170 million people are chronically infected with hepatitis C virus (HCV) worldwide
-- HCV genotype 1 is most common and least responsive to current therapies
-- Leading indication for liver transplantation in Europe and United States
-- Major etiologic factor in hepatocellular carcinoma
· Addition of boceprevir (BOC), an oral inhibitor of HCV-NS3 protease, to peginterferon alfa-2b and ribavirin (R) in the RESPOND-2 trial resulted in a sustained virologic response (SVR) rate of 66% (vs. 21% for control,
P<0.0001) in a population of previous virologic relapsers and non-responders (see Poster 484)
· In a large Phase 3 study in patients infected with HCV genotype 1, the rates of SVR and tolerability did not differ significantly between peginterferon alfa-2a (PEG2a) and peginterferon alfa-2b-ribavirin (PEG2b/R)
regimens (McHutchison et al, N Engl J Med. 2009;361:580-593)
· There have been no studies that have examined the efficacy and safety of BOC in combination with peginterferon alfa-2a (Pegasys®; PEG2a) plus (R)
Objective
To investigate the efficacy and safety of BOC when added to PEG2a/R backbone therapy in previous treatment failure patients with HCV genotype 1

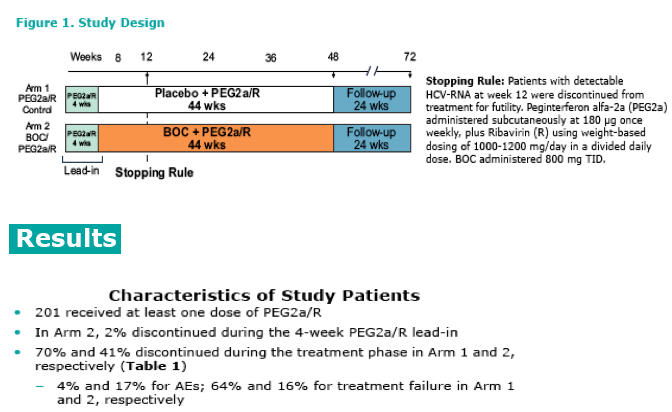
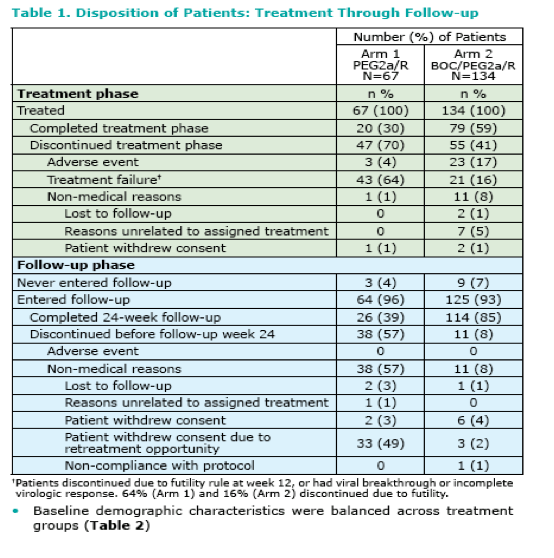
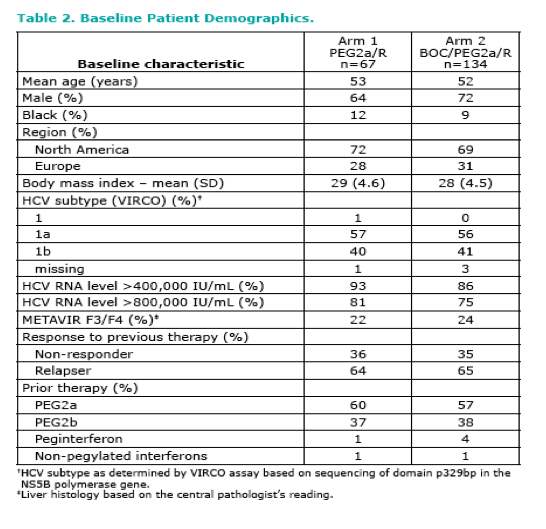
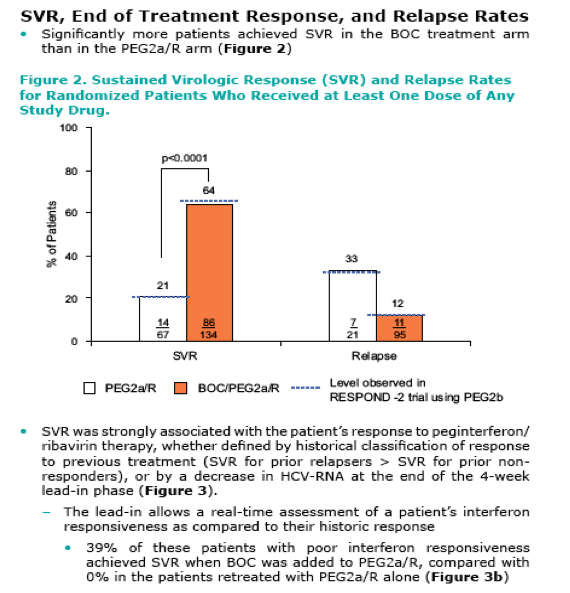
|
| |
|
 |
 |
|
|